Off-patent drugs represent one of the most systematically underexploited categories in the pharmaceutical IP landscape. Their pharmacology is known. Their safety records span decades. Their manufacturing is mature. Yet translating a new indication for a generic molecule into a licensed, reimbursed, commercially viable therapy remains genuinely hard, hard in ways that are distinct from the challenges facing novel drug development, and hard in ways that most pharma IP teams and portfolio managers have not fully mapped.
This analysis goes through every layer of that challenge: the regulatory mechanics in the U.S. and EU that currently block or complicate generic repurposing, the IP tools that do and do not apply, the economic structures that make investment rational or irrational, the specific drugs and companies where these dynamics have played out in real litigation and real deals, and the strategies available to academic institutions, biotech companies, and larger pharma organizations trying to extract value from molecules that nobody owns anymore.
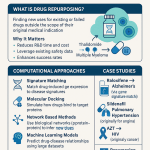
Part I: What Drug Repurposing Actually Is, and Why Generics Are the Hard Case
Defining the Categories: Repurposing, Repositioning, Reformulation
Terminology in this space is imprecise and worth anchoring before going further, because regulatory strategy depends on which category a program falls into.
Drug repurposing, sometimes called drug repositioning, refers to the development of an already-approved drug for a new therapeutic indication. The core asset, the active pharmaceutical ingredient (API) with its existing pharmacokinetics, safety database, and manufacturing process, remains unchanged. What changes is the clinical use. Sildenafil (Pfizer’s Viagra, approved by the FDA in 1998 for erectile dysfunction) was repurposed from its original development program targeting angina pectoris, where it underperformed as a vasodilator but produced a persistent and unexplained side effect that became the basis for a separate NDA. That transition happened while the compound was still on patent, giving Pfizer full commercial control.
Drug reformulation is adjacent but distinct. A reformulation involves changing the delivery mechanism, dosage form, or release profile of an existing drug without necessarily changing its indication. Depakote ER (divalproex sodium extended-release, AbbVie) is a reformulation of immediate-release Depakote. The active molecule is the same; the pharmacokinetic profile differs enough to support separate Orange Book listings and additional patent claims. Reformulation frequently supports evergreening strategies, discussed in detail in Part IV.
The genuinely difficult problem, the one that this analysis focuses on, is the third category: repurposing a drug that has already gone off-patent. By the time a researcher or small biotech identifies a credible new indication for metformin, nitisinone, or fluvoxamine, the original manufacturer has typically left the market or has no commercial interest in the new use. Generic versions are being sold at commodity prices by dozens of manufacturers. Nobody owns the molecule. That commercial vacuum is both the opportunity and the structural problem.
Why Generic Repurposing Looks Attractive on Paper
The economic argument for repurposing off-patent drugs is straightforward. Traditional de novo drug development costs roughly $2.5 billion and takes 10-15 years from initial synthesis to first approval, including the cost of failures. A repurposed generic bypasses the entire preclinical safety characterization phase because the drug has an established human safety record. Depending on whether existing clinical data addresses the new indication, phase I and part of phase II may be compressible or avoidable. That translates to a potential reduction in development cost of 50-60% and timeline compression of 3-5 years, at minimum.
The examples that circulate in scientific literature make the case vivid. Nitisinone, originally developed as an agricultural herbicide and repurposed for hereditary tyrosinemia type 1, was subsequently found to slow the progression of alkaptonuria (AKU), an ultra-rare genetic metabolic disorder affecting connective tissue. The SONIA-2 trial, which ran in the UK, Spain, France, the Czech Republic, and Slovakia, demonstrated a statistically significant reduction in AKU Severity Score Index in patients treated with nitisinone 10mg daily versus placebo. The EMA granted a positive opinion for this indication in 2020. The drug is now marketed as Orfadin by Swedish Orphan Biovitrum (Sobi), which benefited from orphan drug designation providing 10 years of market exclusivity in the EU and 7 years in the U.S.
Sirolimus (rapamycin), a generic immunosuppressant originally approved for renal transplant rejection, was repurposed for autoimmune lymphoproliferative syndrome (ALPS) after researchers observed that its mTOR inhibition mechanism could suppress the lymphocyte accumulation driving the disease. The National Institutes of Health conducted the key clinical work. Formal licensing of this use in the U.S. has moved through a complicated process precisely because the NIH, not a commercial manufacturer, drove the development.
Dexamethasone’s role during COVID-19 is the clearest recent example of rapid generic repurposing at scale. The RECOVERY trial (Randomized Evaluation of COVID-19 Therapy), run by Oxford University, demonstrated a one-third reduction in mortality for hospitalized COVID-19 patients on mechanical ventilation. The cost per course of treatment was approximately $5. The drug was generic, cheap, and globally available. Yet the formal label for COVID-19 was not updated in many jurisdictions for months after the trial reported, precisely because no commercial entity had the incentive to file a supplemental NDA.
The gap between ‘we know this works’ and ‘patients can reliably access this with label support and reimbursement coverage’ is where generic repurposing gets stuck. Every part of that gap involves either regulatory mechanics, IP economics, or commercial infrastructure that currently favors novel drug development over off-patent drug repurposing.
The Polypharmacology Basis for Repurposing
The scientific reason generic repurposing is possible at all is that most approved drugs have pharmacological effects beyond their primary mechanism. Targets interact with multiple downstream pathways. Off-target binding produces secondary effects, some adverse, some potentially therapeutic. The degree of polypharmacology varies by drug class: kinase inhibitors, for instance, are notoriously promiscuous binders, while highly selective biologics have narrower secondary pharmacology.
Systematic identification of repurposing opportunities has moved from serendipity toward rational, data-driven approaches. Network-based methods map drug targets onto disease-gene interaction networks to identify mechanistic overlaps between approved indications and new disease states. Chemoproteomics uses activity-based protein profiling to identify all protein targets that a compound binds at physiologically relevant concentrations, not just the primary target. Machine learning models trained on genomic, proteomic, and clinical datasets generate ranked lists of candidate drug-indication pairs for experimental validation.
The output of these computational approaches is a long list of candidate repurposing hypotheses. Most will not survive even early clinical testing. The bottleneck is not identifying candidates. It is moving validated candidates through development and regulatory review when no commercial entity owns the compound and has an economic reason to invest.
Part II: The Regulatory Mechanics, and Where They Break Down
The U.S. Framework: NDAs, ANDAs, the 505(b)(2) Pathway, and the Gap for Non-Manufacturers
Generic drug approval in the U.S. runs through the ANDA (Abbreviated New Drug Application) pathway under Section 505(j) of the Federal Food, Drug, and Cosmetic Act. An ANDA filer certifies that its drug product is bioequivalent to a reference listed drug (RLD) and either that the RLD’s patents are expired, invalid, or not infringed (a Paragraph IV certification), or that it is not seeking approval for patent-protected indications (a Section viii carve-out). The ANDA pathway does not involve new clinical trials. It relies entirely on the RLD’s safety and efficacy record.
The consequence is structural: an ANDA does not support adding a new indication. If someone wants to add a new approved indication to a generic drug’s label, the applicable pathway is a supplemental NDA (sNDA) under Section 505(b)(1), which requires full clinical trial data, chemistry, manufacturing, and controls documentation, and a complete regulatory submission. That requirement exists because the FDA’s approval process requires a sponsor who is legally responsible for the drug product and can supply the drug samples required for manufacturing review.
That legal responsibility is the problem for academic institutions and non-profits. An academic research group at Michigan Medicine might run a fully powered randomized controlled trial demonstrating that a generic drug works for a new indication. They have the clinical data. They do not have a drug manufacturing facility. They cannot supply CMC data. They cannot provide the product samples the FDA requires. Without those elements, they cannot file an sNDA, which means they cannot get the label updated, which means clinicians and payers rely on off-label use with no standardized dosing guidance and often no insurance coverage.
A Federation of American Scientists analysis published in 2024 proposed a specific legislative solution: a ‘labeling-only’ 505(b)(2) pathway for non-manufacturers. A 505(b)(2) application allows an applicant to rely on previously published studies or on the FDA’s prior findings of safety and efficacy for an existing approved product, supplemented by the applicant’s own data for the new use. The proposed labeling-only variant would go further, allowing a non-manufacturer to reference existing FDA determinations on CMC data, use commercially available drug product samples for any required analytical testing, and submit only the clinical data supporting the new indication. This would let a research institution file for a label update without needing a manufacturing relationship.
As of 2025, that pathway does not exist. It requires Congressional action to create. The FDA has indicated interest in the concept but has not implemented it through guidance alone because the current statutory framework does not support it. That means the regulatory gap for non-manufacturer sponsors is real and will remain real until Congress acts.
The EU Framework: No Pathway, a Pilot Program, and the EMA’s Limits
In the European Union, the situation is if anything less structured. The EMA operates under a centralized procedure, mutual recognition procedure, and decentralized procedure for marketing authorizations. None of these has a dedicated pathway for repurposing off-patent generic drugs for new indications when the applicant is not the original marketing authorization holder and is not a commercial manufacturer.
A generic company can theoretically file for a new indication using the full dossier process, but that requires clinical trial data, full CMC documentation, and a marketing authorization holder capable of supplying the drug. For an academic institution, the process is practically inaccessible. The EMA has acknowledged this gap repeatedly in policy documents and stakeholder consultations.
The EMA’s response has been a pilot program, launched in collaboration with the European network for Health Technology Assessment (EUnetHTA) and supported by the IMI (Innovative Medicines Initiative) REPO-TRIAL project, to support non-commercial sponsors in generating evidence for repurposing authorization. The pilot has supported programs including the nitisinone-AKU case and a fluvoxamine program for obsessive-compulsive disorder in pediatric patients. The structural limitation of the pilot is that it provides scientific advice and procedural support, but not funding. The clinical trials still need to be financed. Academic groups running on grant funding often cannot absorb the cost of a regulatory-standard clinical trial even with EMA scientific support.
European academic consortia have proposed a dedicated EU regulatory procedure, tentatively called a ‘Repurposed Medicine Marketing Authorization’ or similar, that would apply specifically to off-patent drugs with new indications, allow marketing authorization based on non-commercial clinical evidence, and include a government-regulated pricing mechanism to prevent commercial exploitation of a publicly financed development process. That proposal has not advanced to formal legislative action as of 2025.
Off-Label Prescribing: The Workaround and Its Limits
The practical reality for most successfully repurposed generics is that they are prescribed off-label, meaning physicians prescribe them for unapproved indications based on published evidence. Off-label prescribing is legal in both the U.S. and EU, and it is common. An estimated 20% of all prescriptions in the U.S. are off-label, with higher rates in oncology (estimated 60%) and psychiatry.
Off-label use has specific structural weaknesses for repurposed generics. Insurance reimbursement is unreliable. Many payers require an on-label indication for routine reimbursement; off-label use often triggers prior authorization requirements that create access barriers. Dosing guidance may be inconsistent because without a formal approved indication, there is no FDA-reviewed label specifying dose, administration, and patient selection criteria. Prescribers make individual judgment calls rather than following a standardized protocol, which creates variability in clinical practice.
The post-COVID fluvoxamine situation illustrates this precisely. The TOGETHER trial (a platform adaptive trial conducted in Brazil and led by McMaster University) showed statistically significant reduction in COVID-19-related emergency room visits and hospitalization for patients receiving fluvoxamine 100mg twice daily. The evidence base was credible and published in The Lancet. Fluvoxamine is a generic SSRI, available from multiple manufacturers at low cost. But with no sponsor filing for an emergency use authorization or supplemental NDA, formal regulatory status in the U.S. never materialized. Prescribers who wanted to use fluvoxamine for COVID-19 were prescribing off-label with a variable evidence base and minimal insurance coverage.
Key Takeaways, Part II
The ANDA pathway cannot support new indication filings. The sNDA process requires a manufacturing sponsor that academic institutions and non-profits cannot satisfy. The proposed ‘labeling-only’ 505(b)(2) pathway would solve the non-manufacturer problem but requires Congressional action. The EU has no dedicated pathway for repurposed generic marketing authorization, and the EMA pilot provides procedural support but not funding. Off-label prescribing allows clinical use but produces reimbursement gaps, dosing inconsistency, and lack of regulatory validation.
Part III: Intellectual Property Mechanics for Off-Patent Drugs
What Cannot Be Patented Once a Drug Goes Generic
Composition-of-matter patents, the strongest form of pharmaceutical IP because they claim the chemical compound itself and block all uses, cannot be obtained for an already-known and publicly disclosed molecule. The novelty requirement under 35 U.S.C. Section 102 bars patenting any compound that was previously described in a publication, patent, or other disclosure. A metformin molecule that has been commercially available since 1957 cannot anchor a new composition-of-matter patent regardless of what new use is discovered for it.
That forecloses the most valuable IP position. But it does not foreclose all IP positions. Method-of-use patents, which claim the use of a known compound for a specific new therapeutic application, are available for genuinely novel indications if the indication itself was not previously described and is not obvious from the prior art. These are filed under Section 101 as process claims: ‘A method of treating [new disease] comprising administering [known compound] to a patient in need thereof.’
The durability of method-of-use patent protection for generic drugs is limited by a structural problem specific to the generic pharmaceutical market: skinny labeling. Under the Hatch-Waxman framework, when an originator’s drug has multiple approved indications and some indications are covered by Orange Book-listed patents while others are not, a generic filer can use a Section viii statement to carve out the patented indication from its proposed label. The generic then markets the drug for only the unpatented indications. The generic label literally omits the patented use. The patent remains listed in the Orange Book. The generic is approved with a narrower label.
The problem for a company that has obtained a method-of-use patent on a repurposed generic indication is that pharmacy-level generic substitution routinely places patients on the generic even when the prescriber intended the branded product for the patented indication. Most prescriptions are filled by generic substitution at the pharmacy unless the prescriber specifically writes ‘dispense as written.’ A patient prescribed fluvoxamine for a new indication covered by a method-of-use patent will often receive a generic fluvoxamine without the patented indication on the label, and the method-of-use patent provides limited practical protection against that substitution.
Method-of-Use Patents: Prosecution Strategy and Enforcement Reality
Filing method-of-use patents for repurposed generics is worth doing, but the prosecution and enforcement strategy must account for the specific vulnerabilities these patents face.
During prosecution, the primary challenges are obviousness under 35 U.S.C. Section 103 and inadequate written description under Section 112. Patent examiners will search prior art for any suggestion that the drug might be useful for the new indication. For drugs with broad pharmacological activity, prior art is often dense. Metformin, for instance, has been the subject of decades of speculative publications about its potential anticancer and anti-aging properties. Any publication that suggests metformin might be useful in a particular cancer type, even without supporting clinical data, can be cited as prior art for obviousness analysis. Overcoming that requires demonstrating unexpected results: clinical or preclinical data showing a magnitude or type of effect that was not predictable from the prior art.
The written description requirement for method-of-use claims is also stringent for new indications supported primarily by mechanistic rationale without robust clinical data. The Federal Circuit has required that method claims be supported by data demonstrating that the claimed method actually works, not merely that it might work based on pharmacological theory.
On enforcement, the structural weakness is the ‘induced infringement’ theory. Direct patent infringement requires unauthorized practice of the patented method. When a physician prescribes a generic drug for the patented indication, the physician is practicing the method. The generic manufacturer is not directly infringing because it does not practice the method of treatment. But the generic manufacturer may be liable for induced infringement under 35 U.S.C. Section 271(b) if it actively encourages the infringing use. Courts have found induced infringement where a generic’s label, even a skinny label, contained language that encouraged the patented use.
The key case is GlaxoSmithKline v. Teva Pharmaceuticals (Federal Circuit, 2021, on rehearing). GSK held method-of-use patents on carvedilol (Coreg) for congestive heart failure. Teva filed a Paragraph IV certification, and GSK sued. The dispute centered on whether Teva’s skinny label, which carved out the CHF indication, nonetheless induced infringement by including language about heart failure treatment that effectively instructed the patented use. The Federal Circuit found induced infringement. That decision, while fact-specific, supports the value of method-of-use patents for repurposed drugs when the generic’s label retains language touching on the patented indication. It also illustrates that skinny labeling does not automatically insulate generic manufacturers from infringement liability.
Secondary Patents: Formulations, Dosing Regimens, and Combination Products
When the repurposed use of a generic requires a novel formulation, the IP position improves significantly. If the new indication requires a modified-release profile, an alternative route of administration, a new salt form, or a fixed-dose combination with another compound, each of those elements can support patent claims that are stronger than method-of-use claims alone.
Amantadine illustrates this precisely. The drug was originally approved as an antiviral (Symmetrel, marketed by Shire). It was subsequently found to reduce dyskinesia in Parkinson’s disease patients receiving levodopa, a use that became widespread off-label. When Adamas Pharmaceuticals developed an extended-release amantadine formulation (Gocovri), specifically designed to achieve the plasma concentration profile needed for dyskinesia control with once-nightly dosing, they supported it with a 505(b)(2) NDA referencing the existing amantadine safety database and their own clinical trial data (the EASE LID-3 trial, published in JAMA Neurology in 2017). FDA approved Gocovri in 2017. Adamas obtained patents on the extended-release formulation and the dosing regimen. Those formulation patents provided a durable IP position that Adamas defended in Hatch-Waxman litigation.
The Adamas/Gocovri situation is a replicable model: take a generic molecule with demonstrated off-label efficacy, invest in clinical trials with a proprietary formulation that improves on standard generic dosing, file a 505(b)(2) NDA relying on the generic’s safety record, and obtain formulation and dosing patents that can be listed in the Orange Book. This approach requires capital, a commercial sponsor, and a formulation innovation that can withstand obviousness challenges. But it works, and it produces listed Orange Book patents that trigger the Hatch-Waxman litigation stay when generic filers challenge them.
Orphan Drug Exclusivity as a Substitute for Patent Protection
For repurposed generics targeting rare diseases (defined in the U.S. as conditions affecting fewer than 200,000 patients annually), orphan drug designation from the FDA provides seven years of market exclusivity from the date of approval. In the EU, orphan designation provides ten years of market exclusivity. These exclusivity periods are independent of patent protection and can apply even when no patents cover the drug.
That makes orphan drug designation the most accessible IP substitute for repurposed generics in rare disease settings. A company that identifies a credible new use for a generic drug in a rare disease, runs the clinical trials, and obtains FDA approval gets seven years in which the FDA cannot approve another application for the same drug in the same indication, regardless of whether any patent covers it.
The Sobi nitisinone-AKU case is the direct example. Nitisinone was already generic when the AKU indication was developed. Sobi’s commercial position rests on orphan exclusivity, not on any composition-of-matter patent. The manufacturing process for nitisinone is established and available to generic manufacturers, but the orphan exclusivity period blocks competing applications for the AKU indication until that exclusivity expires.
The risk with orphan exclusivity as an IP strategy is price sensitivity. Congress and HHS have increased scrutiny of drugs that receive orphan designation for conditions that are adjacent to larger market opportunities, concerned about companies using orphan exclusivity for commercial leverage in markets larger than the statute contemplated. The Orphan Drug Act’s consent provision allows a second applicant to obtain approval for the same drug in the same orphan indication by consent of the first approval holder, or by demonstrating clinical superiority. In practice, those provisions rarely disrupt the first approved product’s commercial position, but the regulatory and legislative environment around orphan drug pricing is under sustained pressure.
Data Exclusivity and Its Interaction with Generic Repurposing
New Clinical Investigation exclusivity (three years in the U.S.) applies when an applicant conducts new clinical studies essential to the approval of a change to an already-approved drug, including a new indication. A company that runs new clinical trials for a new indication of an existing approved drug, and files a 505(b)(2) NDA or an sNDA based on those trials, receives three years of exclusivity during which the FDA cannot grant approval to another applicant relying on those same studies.
Three years is a short exclusivity window. For a specialty drug with high pricing power, it may be sufficient. For a commodity generic with a low price, three years of exclusivity produces inadequate return on clinical trial investment. That is the core economic disincentive for repurposing generic drugs without access to orphan designation or a proprietary formulation.
The Hatch-Waxman five-year New Chemical Entity (NCE) exclusivity does not apply to repurposed generics because it requires the active moiety to not have been previously approved. An off-patent drug’s active moiety has already been approved.
Key Takeaways, Part III
Composition-of-matter patents are unavailable for off-patent molecules. Method-of-use patents are available but face prosecution obstacles (obviousness based on prior mechanistic speculation) and enforcement limitations (skinny labeling, pharmacy substitution). The GlaxoSmithKline v. Teva Federal Circuit decision supports induced infringement liability when a generic’s label language encourages the patented use, even after carve-out. Formulation and dosing regimen patents, as demonstrated by the Adamas/Gocovri amantadine model, provide stronger and more enforceable IP than method-of-use claims alone. Orphan drug exclusivity (7 years U.S., 10 years EU) is the most accessible commercial protection mechanism for rare disease applications of repurposed generics. Three-year new clinical investigation exclusivity is too short to justify investment without an accompanying orphan designation or formulation patent.
Investment Strategy Note
Portfolio managers evaluating repurposed generic programs should weight orphan drug designation and formulation patent status as primary valuation drivers, not method-of-use patents. A repurposed generic with orphan designation and a proprietary formulation patent carries a risk-adjusted NPV that can approach specialty pharmaceutical valuations. The same drug without those protections is worth only the clinical data value, which is modest without commercial exclusivity. Assess whether the indication qualifies for orphan designation before committing capital to clinical development.
Part IV: Evergreening, Patent Thickets, and How They Affect Generic Repurposing Access
The Mechanics of Pharmaceutical Evergreening
Evergreening is the practice of filing secondary patents on modifications, formulations, or new uses of an existing drug to extend effective market exclusivity beyond the expiration of the original composition-of-matter patent. The term is pejorative in health policy contexts but describes a legal and commercially rational strategy.
The secondary patent categories used for evergreening include patents on polymorphic crystal forms of the API (Form I vs. Form II crystal structures, for instance), enantiomers (single-enantiomer versions of racemates, such as the switch from omeprazole/Prilosec to esomeprazole/Nexium that AstraZeneca executed with commercial success), prodrug versions, sustained-release formulations, new salt forms, pediatric formulations, and new therapeutic indications. Each category supports a new Orange Book patent listing that triggers a 30-month stay on ANDA approval when challenged by a Paragraph IV certification.
AbbVie’s management of the adalimumab (Humira) patent estate is the most documented recent example of evergreening at scale. The compound patent on adalimumab expired in the U.S. in 2016. AbbVie assembled a portfolio of over 165 patents covering manufacturing processes, formulations, dosing regimens, citrate-free formulations (reducing injection site pain), and combination therapies. That thicket held biosimilar entry at bay until January 2023 in the U.S., producing an additional seven years of effective exclusivity beyond compound patent expiration. AbbVie generated over $200 billion in global Humira revenue during that extended exclusivity period.
The relevance to generic repurposing is two-directional. First, when a large pharmaceutical company has built a patent thicket around a branded drug still generating meaningful revenue, generic repurposing efforts for new indications of that drug must navigate those layered patents even after the compound patent expires. If the new indication requires a formulation or dosing approach covered by a thicket patent, the generic repurposing program faces IP complications even for an off-patent molecule.
Second, the evergreening model applied to repurposed generics is exactly what the Adamas/amantadine strategy exemplifies: use a novel formulation or dosing regimen to create new Orange Book-listable patents on top of a generic molecule, supporting a commercial product with a durable IP position. That strategy is a feature for commercial repurposing sponsors. It is a barrier for public health-motivated repurposing efforts that want the cheap generic version to reach patients, not a new proprietary formulation.
Section viii Carve-Outs and the Skinny Label Litigation Landscape
When a branded drug has multiple Orange Book-listed patents covering different indications, generic filers can use Section viii of the ANDA framework to carve out the patented indications and seek approval only for unpatented indications. The generic’s label will be narrower than the brand’s label, omitting the patented therapeutic uses.
The practical tension with this mechanism is that it does not reliably prevent the generic from being dispensed for the patented indication. Physicians prescribe by drug name for the approved indication, and pharmacists substitute generics based on therapeutic equivalence ratings, not label scope. A patient receiving carvedilol for CHF (the patented Coreg indication) may be dispensed generic carvedilol, which under its skinny label does not include the CHF indication, because the pharmacy’s dispensing system substitutes the generic for the branded product regardless of the specific clinical indication.
That substitution dynamic is both the commercial problem for branded manufacturers holding method-of-use repurposing patents and the patient access problem for generic repurposing advocates. When the generic is substituted into the patented use without the label support of the patented indication, patients receive the drug without the clinical guidance developed for that specific use.
Post GlaxoSmithKline v. Teva, brand manufacturers have more litigation ammunition against generic filers whose skinny labels retain language that encourages the patented use. But the core pharmacy substitution dynamic has not changed. The GSK v. Teva outcome makes generic label drafting more consequential, not the substitution mechanics themselves.
IP Valuation: What a Repurposed Generic Patent Estate Is Actually Worth
When an IP team is valuing a repurposed generic program for licensing, acquisition, or internal portfolio prioritization, the valuation framework must account for the specific exclusivity mechanisms available and their duration.
A program with orphan drug designation in both the U.S. and EU, a proprietary extended-release formulation supported by process and formulation patents, and three-year new clinical investigation exclusivity on the label update has a stacked exclusivity profile: roughly seven to ten years of orphan exclusivity, additional patent term on the formulation (typically 10-15 years from filing depending on prosecution history), and three years of data exclusivity. The overlapping exclusivity periods produce an effective commercial window that can support specialty drug economics.
Contrast that with a program where the only IP protection is a single method-of-use patent with no orphan designation. That program’s commercial value depends almost entirely on whether the method-of-use patent will be enforced against generic substitution, which based on pharmacy substitution mechanics is uncertain. Without orphan designation and formulation IP, the method-of-use patent may be worth little more than a modest licensing royalty stream from any commercial manufacturers who wish to explicitly label the new indication.
The key multiplier is orphan designation. A program that qualifies for orphan designation can command 10-30x the pricing of the generic commodity form, support a standalone commercial launch from a small specialty pharma company, and justify clinical trial investment of $30-100 million. A program that cannot qualify for orphan designation needs either a formulation patent or a very large commercial market to generate adequate return on development investment.
Part V: The Economic Structure of Generic Repurposing, and Why the Market Currently Fails
Why Pharmaceutical Companies Don’t Invest in Generic Repurposing
The market failure in generic drug repurposing is not irrational behavior by pharmaceutical companies. It is a predictable response to the incentive structure. Pharmaceutical R&D investment is driven by expected return on capital, and generic repurposing offers inadequate expected return for most large pharmaceutical companies under current conditions.
The R&D cost to run a Phase II/III clinical program establishing efficacy for a new indication of a generic drug runs from $20 million for a small trial in a rare disease to $200 million or more for a large trial in a common indication. The regulatory cost of an sNDA submission adds several million dollars. If the program succeeds, the available exclusivity is limited to three years of new clinical investigation exclusivity (unless orphan designation applies), a potentially unenforceable method-of-use patent, and whatever pricing premium the market will sustain for the newly labeled generic versus the commodity generic.
Large pharma companies earn their cost of capital on drug programs with peak revenue potential exceeding $500 million. A repurposed generic targeting a broad indication at generic pricing cannot realistically achieve that revenue threshold. A repurposed generic targeting a rare disease can, but only with orphan designation and aggressive pricing, which is increasingly under regulatory and political pressure. That leaves a narrow commercial window within which generic repurposing is financially attractive to pharmaceutical companies without special circumstances.
Mid-sized and small specialty pharma companies have a better fit with generic repurposing economics when orphan designation is available. Companies including Catalyst Biosciences, Strongbridge Biopharma (now Xeris), and Recordati Rare Diseases have built commercial models around repurposing off-patent compounds in rare disease settings. Their economics work because orphan pricing (often $50,000-$200,000 per patient per year) applied to a clinical development cost of $30-70 million and a small patient population produces adequate return on capital.
Outside orphan disease settings, the economic case for commercial investment in generic repurposing requires additional policy support. The specific mechanisms proposed and partially implemented include the NIH’s National Center for Advancing Translational Sciences (NCATS) repurposing program, the EU’s COST Action CA17104 (STRATAGEM) network supporting non-commercial repurposing research, advanced market commitments from government health systems that guarantee revenue for successfully repurposed generics meeting public health criteria, and prize mechanisms that delink repurposing development cost from drug pricing by awarding a cash prize for successful new indication development in exchange for the drug entering the commodity generic market.
The Academic Research Gap: From Clinical Discovery to Regulatory Authorization
Academic research groups identify many of the most promising generic repurposing opportunities. They conduct the investigator-initiated trials, publish the pivotal efficacy data, and generate the scientific foundation for regulatory submissions. What they cannot do is file those regulatory submissions, because they are not drug manufacturers and cannot satisfy the CMC requirements of an sNDA.
The Michigan Medicine model illustrates the gap and one attempt to address it. University of Michigan researchers conducted significant work identifying metformin’s potential in reducing cancer risk and improving outcomes in diabetic oncology patients. That work informed a substantial body of clinical evidence published in peer-reviewed journals. It has not translated into a formal FDA-approved new indication for metformin because no commercial manufacturer has filed an sNDA and no non-manufacturer pathway exists for the academic group to file independently.
A 2024 analysis in Frontiers in Pharmacology mapped the specific structural obstacles for non-manufacturer sponsors seeking to advance generic drug repurposing in the U.S. cancer context. The authors identified the CMC data requirement, the product sample requirement, and the legal responsibility for the drug product as the three specific elements of the sNDA process that exclude academic sponsors. Their proposed solution aligns with the FAS ‘labeling-only’ 505(b)(2) proposal: allow non-manufacturers to reference existing FDA CMC determinations and use commercially purchased drug products to satisfy sample requirements.
The EMA pilot program has moved further toward accommodating academic sponsors. The STAMP (Scientific Advice for companies and applicants) procedure allows academic sponsors to receive scientific advice from EMA on repurposing programs at reduced or waived fees. The PRIME (Priority Medicines) scheme, while primarily designed for novel drugs with unmet needs, has been extended to some repurposing programs. But neither scheme resolves the marketing authorization holder requirement, which requires a commercial entity to hold the MA and be responsible for pharmacovigilance.
Public-Private Partnership Models That Have Actually Worked
Several partnership models have produced licensed repurposed generics in practice, each with structural features worth understanding.
The NIH-NCI Experimental Therapeutics (NExT) program funds clinical development of repurposed compounds, including generic drugs, for cancer indications. NExT has supported trials of metformin in breast cancer, aspirin in colorectal cancer prevention, and hydroxychloroquine in pancreatic cancer. These trials generate clinical data that could support regulatory submissions, but the NExT program does not include a regulatory filing component. The data goes to the scientific literature. Commercial translation requires a separate sponsor.
The Drugs for Neglected Diseases initiative (DNDi), a non-profit product development partnership, has successfully moved repurposed drugs through regulatory approval for neglected tropical diseases. Fexinidazole, a drug originally developed in the 1970s and shelved, was repurposed by DNDi for sleeping sickness (human African trypanosomiasis). DNDi conducted phase II/III clinical trials, obtained EMA approval in 2018 (the first new oral treatment for sleeping sickness in 50 years), and worked with Sanofi to handle manufacturing and regulatory submission. The DNDi model explicitly separates development from commercial exploitation: DNDi holds the data, Sanofi provides manufacturing and regulatory infrastructure, and the drug is priced at cost for endemic-country patients.
The CureAccelerator platform, operated by CSDD at Tufts University, connects companies with repurposing opportunities in their existing compound libraries to academic and clinical investigators interested in testing those compounds. This exchange model addresses a specific inefficiency: pharmaceutical companies often have compounds in their libraries that they have abandoned for their original indication but that may have value for other conditions. CureAccelerator facilitates agreements under which the company provides drug supply, the academic investigator runs the trial, and IP arrangements are negotiated in advance. This does not resolve the regulatory filing problem for generics, but it has moved several compounds into clinical testing that would not otherwise have been studied.
Key Takeaways, Part V
Commercial pharmaceutical companies rationally underinvest in generic repurposing outside orphan disease settings because the available exclusivity is too short and the pricing power too limited to justify the clinical development cost. Orphan drug designation fundamentally changes the economic calculus, enabling specialty pricing that can justify $30-100 million in clinical investment. Academic institutions identify repurposing opportunities but cannot file sNDAs because they lack manufacturing capability, and no legislative solution to that gap exists yet. The DNDi fexinidazole model demonstrates that non-profit product development partnerships can move repurposed drugs through regulatory approval when the right manufacturing and regulatory filing infrastructure is provided. Prize and advanced market commitment mechanisms have been proposed but not implemented at scale for generic repurposing outside neglected disease contexts.
Investment Strategy Note
For institutional investors and biotech portfolio managers, the investment signal in generic repurposing programs is: does this program have orphan designation plus a proprietary formulation? If both are present, the economics can support specialty pharmaceutical valuations and 10-15x investment returns. If only orphan designation is present, the economics work but are thinner (5-8x potential). If neither is present, the program requires a policy change (the FAS labeling-only pathway) or a large enough commercial market to sustain commodity-price revenue over a multi-year exclusivity window. Absent a legislative catalyst, programs in the third category are philanthropic bets, not investment theses.
Part VI: Specific Drug and Company Case Studies
Sildenafil: Serendipity While On-Patent
Sildenafil is the canonical repurposing story, but its commercial mechanics are different from the generic repurposing problem. Pfizer developed sildenafil as a PDE5 inhibitor for pulmonary arterial hypertension and angina. The unexpected persistent erection side effect observed during angina trials prompted the decision to redirect the program. Pfizer filed a new NDA for erectile dysfunction. The FDA approved Viagra in 1998. Sildenafil was still on patent; Pfizer had full commercial control and priced Viagra at a level that generated roughly $1.9 billion in annual peak revenues.
The relevant lesson is timing: serendipitous repurposing with commercial exclusivity produces blockbuster economics. The same sildenafil later lost patent protection, and Revatio (sildenafil 20mg for pulmonary arterial hypertension) became generically available. The PAH indication, which Pfizer had developed and sought to protect with secondary patents, became accessible to generic manufacturers who filed ANDAs referencing Revatio. Pfizer’s IP strategy to separate the ED and PAH commercial positions through dose-level IP distinctions (Viagra at 25/50/100mg vs. Revatio at 20mg) illustrates exactly the kind of formulation-level IP differentiation that off-patent repurposing programs need to replicate.
Rituximab: IP Valuation of a Repurposed Biologic
Rituximab (Rituxan, co-marketed by Genentech and Roche) was approved by the FDA in 1997 for B cell non-Hodgkin’s lymphoma. Researchers subsequently identified its utility in autoimmune conditions including rheumatoid arthritis (approved 2006, in combination with methotrexate), and later multiple sclerosis, where its CD20-mediated B cell depletion mechanism addresses the pathological B cell role in MS disease activity. The MS use was off-label for rituximab but generated the scientific rationale for ocrelizumab (Ocrevus, Roche), a humanized anti-CD20 antibody specifically engineered and clinically validated for relapsing and primary progressive MS, approved by the FDA in 2017.
The rituximab-to-ocrelizumab transition illustrates a commercial pattern worth tracking: an off-label use of a generic-adjacent biologic (rituximab biosimilars entered the U.S. market in 2019) becomes the scientific validation for a novel patented biologic designed specifically for that indication. Roche and Genentech captured the MS indication through a new molecular entity rather than attempting to obtain regulatory approval for rituximab in MS, sidestepping the generic repurposing problem entirely. Ocrevus generated $6.4 billion in global revenues in 2023. The IP estate on ocrelizumab (composition-of-matter patents on the humanized antibody sequence) provides durable exclusivity through the mid-2030s.
The rituximab/ocrelizumab transition is not replicable for small-molecule generics where there is no obvious ‘next-generation’ patentable molecule. But it illustrates the commercial logic behind large pharma’s preference for developing novel IP over repurposing generics, even when the scientific path to a new indication runs directly through an off-patent drug.
Nitisinone and Alkaptonuria: The Orphan Playbook in Practice
Nitisinone is a 4-hydroxyphenylpyruvate dioxygenase (HPPD) inhibitor originally developed as an agricultural herbicide and subsequently approved for hereditary tyrosinemia type 1 (HT-1) under the brand name Orfadin, with Sobi holding the marketing authorization. HT-1 is a rare inborn error of metabolism affecting roughly 1 in 100,000 live births globally.
The alkaptonuria indication emerged from researchers at the Royal Liverpool University Hospital who observed that nitisinone’s mechanism, inhibiting HPPD and thereby reducing homogentisic acid accumulation, addressed the same enzymatic pathway dysregulated in AKU. The National Alkaptonuria Centre ran the DevelopAKUre clinical program, a consortium funded by the European Commission under Horizon 2020, conducting three clinical trials (SULTAN, SOFIA, SONIA-2) to establish nitisinone’s efficacy in AKU.
The EMA granted marketing authorization for nitisinone 10mg in AKU in 2020 under the brand name Orfadin (Sobi), with Sobi holding 10 years of orphan exclusivity in the EU. The U.S. FDA granted orphan drug designation and, following Sobi’s regulatory filing, approval for the AKU indication. Sobi prices Orfadin at approximately $200,000+ per patient per year for AKU, consistent with orphan drug pricing in the EU and U.S.
The DevelopAKUre academic consortium conducted all the clinical development work. Sobi held the manufacturing infrastructure, regulatory expertise, and marketing authorization holder status, and will collect orphan exclusivity revenues for the next decade. The arrangement is a functioning model of academic-commercial collaboration for generic repurposing, but the commercial value accrued to Sobi, not to the academic consortium that generated the clinical evidence.
Metformin in Oncology: The Case That Has Not Closed
Metformin is the most-prescribed diabetes drug in the world, generic since the early 1990s, and available at less than $10 for a month’s supply. It is also one of the most heavily studied potential repurposing candidates in oncology. Epidemiological data consistently shows that diabetic patients taking metformin have lower incidence of multiple cancer types and, in retrospective analyses, better outcomes in some malignancies including breast, colorectal, and prostate cancer.
The mechanistic rationale is credible: metformin activates AMPK, inhibits mTOR signaling, reduces circulating insulin and IGF-1, and has direct antiproliferative effects in cancer cell lines. The National Cancer Institute’s NExT program funded several prospective trials including MA.32 (metformin in early-stage breast cancer, run by NCIC CTG in patients without diabetes), NCIC CTG CO.26 (metformin plus best supportive care in chemotherapy-resistant colorectal cancer), and an ECOG-ACRIN prostate cancer trial.
The MA.32 trial enrolled 3,649 patients over five years. Primary results reported in 2022 showed no significant improvement in invasive disease-free survival overall. However, subgroup analyses suggested potential benefit in hormone receptor-negative breast cancer, an area that requires further validation. The CO.26 trial showed no survival benefit. The prostate cancer trial has not yet reported its primary endpoint.
As of 2025, metformin in oncology does not have a supported regulatory submission path in the U.S. because the prospective trial data does not clearly support a new indication, and no commercial sponsor has filed. The academic research groups that ran the trials cannot file. The generic manufacturers have no incentive to file. The FDA has no mechanism to update the label based solely on publicly available trial data without a sponsor.
Metformin’s oncology story is the clearest current example of the public health cost of the regulatory gap: credible biology, a large body of clinical evidence, a globally available cheap drug, and no path to formal indication approval.
Key Takeaways, Part VI
The sildenafil case shows that serendipitous repurposing while on-patent produces blockbuster economics; the generic repurposing problem emerges only after patent expiration. The rituximab-to-ocrelizumab transition illustrates large pharma’s preference for developing novel patented molecules over pursuing new indication approval for biosimilar-adjacent biologics. The nitisinone-AKU case is the working template for academic-commercial generic repurposing in rare diseases: academic consortium runs clinical trials, commercial entity holds marketing authorization and collects orphan exclusivity revenues. Metformin in oncology illustrates the public health cost of the non-manufacturer regulatory gap: credible clinical evidence exists but cannot reach a formal new indication without a commercial sponsor.
Part VII: Reformulation Strategy as an IP Architecture Tool
Why Reformulation and Repurposing Overlap Strategically
Reformulation, changing the physical or chemical form of an existing drug without necessarily changing its indication, is frequently the mechanism through which repurposing programs create patentable IP. The combination matters because reformulation can be the difference between a repurposed generic with thin IP and a repurposed drug with an enforceable commercial position.
The key regulatory tool supporting this combination is the 505(b)(2) NDA, which allows an applicant to file for approval of a drug that differs from an approved reference drug in formulation, route of administration, dosage form, or indication, while relying in part on previously approved data for the unchanged elements. A company pursuing repurposing through reformulation files a 505(b)(2) referencing the existing generic’s safety and toxicology record, adds its own clinical data for the new indication or formulation benefit, and obtains approval with new Orange Book-listed patents on the reformulation.
The Adamas/amantadine model (Gocovri for dyskinesia in Parkinson’s disease) is the direct example. Amantadine was off-patent. Its use for dyskinesia was off-label. Adamas developed an extended-release capsule formulation designed to achieve peak plasma concentrations during sleep and declining concentrations during waking hours, a profile that matched the pharmacokinetic rationale for dyskinesia reduction with once-nightly dosing. The EASE LID-3 Phase III trial validated the efficacy of the specific formulation for the specific indication. FDA approved Gocovri in 2017. Adamas patented the extended-release formulation and dosing regimen. Those Orange Book-listed patents triggered a 30-month stay when Sun Pharma filed a Paragraph IV ANDA. The Hatch-Waxman litigation ultimately resulted in a settlement allowing Sun Pharma generic entry at an agreed date.
Gocovri peak revenues were approximately $80 million annually. The total clinical development cost for the repurposing program was approximately $40-60 million. The IP estate enabled commercial pricing at over $3,000 per month versus cents per dose for generic amantadine. That pricing differential was sustained by the Orange Book patent protection and the 30-month litigation stay during which Adamas had market exclusivity.
The Reformulation Technology Roadmap for Generic Repurposing
Pharmaceutical formulation science offers multiple technical approaches that can transform a commodity generic into a proprietary reformulated product with patentable distinctions. Understanding which technologies are appropriate for a given molecule and indication is a critical early decision point for any repurposing program with commercial intent.
Extended-release systems include matrix tablets, where the API is embedded in a hydrophilic polymer matrix (typically hydroxypropyl methylcellulose, HPMC) that controls release through swelling and erosion; reservoir systems, where the API core is coated with a semipermeable membrane controlling diffusion; osmotic pump systems (OROS technology, pioneered by ALZA Corporation), where water influx through a semipermeable membrane drives API release through a laser-drilled orifice at a controlled rate; and multiparticulate systems, where the API is loaded into pellets or beads with individual release-controlling coatings. Each technology produces a distinct pharmacokinetic profile and can support formulation patent claims based on the specific release mechanism and the resulting plasma concentration-time curve.
Lipid-based drug delivery systems, including solid lipid nanoparticles, self-emulsifying drug delivery systems (SEDDS), and nanostructured lipid carriers, are appropriate for poorly water-soluble APIs where bioavailability enhancement is the formulation goal. Cocrystal and amorphous solid dispersion technologies (hot-melt extrusion, spray drying) improve solubility of crystalline APIs. Each technology creates patentable formulation innovations when combined with clinical data demonstrating improved efficacy or safety relative to the generic reference product.
Route-of-administration changes generate strong IP when the change has clinical rationale. Azelastine’s reformulation from a nasal spray to an ophthalmic solution (Lastacaft, Allergan) generated separate IP. Ketamine’s reformulation from IV administration to a nasal spray (Spravato, Janssen) enabled a 505(b)(2) NDA for treatment-resistant depression, with Orange Book patents on the esketamine nasal spray device and formulation providing commercial exclusivity that supported a $400+ million annual revenue product built on a generic molecule.
The esketamine/Spravato case is instructive. Ketamine was off-patent and had been used off-label for depression for years. Janssen invested in Phase III clinical trials specifically for the intranasal esketamine formulation, obtained FDA approval in 2019 for treatment-resistant depression, and listed formulation and device patents in the Orange Book. The drug is administered in certified healthcare settings under a Risk Evaluation and Mitigation Strategy (REMS) program, which further differentiates the commercial product from compounded ketamine alternatives. The REMS requirement, while operationally burdensome, also functions as a market access barrier that protects the commercial product against compounding pharmacy substitution.
IP Valuation of Reformulated Repurposed Generics
A reformulated repurposed generic’s IP estate has higher value than method-of-use patents alone because formulation patents are more readily enforced through the Hatch-Waxman framework. Orange Book listing is the mechanism: a listed formulation patent triggers a 30-month stay on ANDA approval when a Paragraph IV challenger disputes it. That stay creates a finite period of commercial exclusivity during which the innovator can recoup development investment before generic competition begins.
Valuing the IP estate of a reformulated repurposed generic requires modeling the expected duration of market exclusivity based on the listed Orange Book patents’ expiration dates, the probability of successful Paragraph IV challenge (which depends on claim scope, prior art exposure, and prosecution history), and the pricing premium sustainable relative to the commodity generic during the exclusivity period.
For a drug with a 10-year formulation patent, a 30-month Hatch-Waxman stay triggered by a Paragraph IV filing, and a successful settlement allowing generic entry at the patent’s expiration, the effective commercial exclusivity window is approximately 10 years from NDA approval. If the drug can sustain $100 million in annual peak revenue at a pricing premium of 10-50x the generic commodity price, the total commercial value over 10 years is $700 million-$1 billion discounted at pharmaceutical industry cost of capital rates.
That range supports acquisition prices in the $200-400 million range for programs with proven clinical efficacy and issued formulation patents, which is consistent with transaction multiples observed in specialty pharma M&A for orphan-adjacent or rare disease reformulation programs.
Key Takeaways, Part VII
Reformulation is the primary tool for creating enforceable Orange Book-listed IP on repurposed generic molecules. Extended-release systems, lipid-based delivery systems, route-of-administration changes, and fixed-dose combinations each support distinct formulation patent claims when backed by clinical data demonstrating formulation-specific benefit. The Adamas/Gocovri amantadine and Janssen/Spravato esketamine cases are replicable commercial templates: take off-patent molecules with established off-label efficacy, invest in proprietary formulation development and Phase III trials for the specific indication, file 505(b)(2) NDAs with listed Orange Book patents, and defend against Paragraph IV challengers through the Hatch-Waxman litigation stay. A reformulated repurposed generic with 10-year formulation patent coverage, orphan designation in a rare indication, and $100 million annual peak revenue potential can support M&A transaction values of $200-400 million.
Part VIII: Policy Landscape and What Needs to Change
The FAS Labeling-Only 505(b)(2) Proposal: Mechanics and Prospects
The Federation of American Scientists’ 2024 proposal for a ‘labeling-only’ 505(b)(2) pathway addresses the non-manufacturer regulatory gap with specificity that earlier policy discussions lacked. The proposal is worth understanding in detail because it represents the most actionable legislative option currently in circulation.
Under the proposed pathway, a non-manufacturing sponsor (academic institution, non-profit, or government agency) would file for a new indication of a drug for which multiple generic products are already marketed. The application would reference the FDA’s prior CMC determinations for the existing approved generic products, rather than requiring the applicant to submit its own CMC package. The applicant would use commercially available generic drug products to satisfy any required bioanalytical testing rather than providing drug product samples from its own manufacturing facility. The applicant would submit its own clinical data demonstrating efficacy and safety for the new indication, along with a proposed label update.
If approved, the label of the existing generic products would be updated to include the new indication. The non-manufacturing sponsor would receive whatever regulatory exclusivity the new indication qualifies for, specifically three-year new clinical investigation exclusivity if new clinical studies were essential to the approval. Generic manufacturers who already hold approved ANDAs would not be required to add the new indication to their labels but could do so voluntarily.
The mechanism for updating labels of existing generic products without requiring those manufacturers to refile raises administrative questions that the FDA would need to resolve through implementing regulations. The Hatch-Waxman framework’s Orange Book listing requirements for any new method-of-use patents on the new indication would also need to be addressed. But the core concept is sound and directly addresses the gap that is currently preventing academic clinical evidence from reaching formal regulatory approval.
The pathway requires legislative action because the current statutory framework for 505(b)(2) requires that the application reference a listed drug and that the applicant take responsibility for the product’s safety, which existing law has been interpreted to require manufacturing capacity. A statutory amendment would need to create an explicit carve-out for non-manufacturer applicants relying on existing CMC determinations.
International Harmonization: The Distance Between the U.S. and EU
The regulatory frameworks for generic repurposing in the U.S. and EU have evolved separately and produce different operational challenges. Harmonization would reduce the cost of running global repurposing programs and allow evidence generated in one jurisdiction to be used more efficiently in another.
The core structural difference is that the EU marketing authorization framework, unlike the Hatch-Waxman ANDA system, does not have a generic approval pathway that separates CMC review from clinical review as cleanly. EU generic approvals under Article 10 of Directive 2001/83/EC (the ‘abridged procedure’) allow approval based on demonstrated bioequivalence to a reference medicinal product, without new clinical trials. But new indication applications for existing authorized products must use a variation procedure, which requires the marketing authorization holder to file. There is no EU equivalent of the ANDA Paragraph IV mechanism or the proposed FAS labeling-only pathway.
The EMA’s repurposing pilot program has created de facto accommodations for academic sponsors, but these are procedural workarounds rather than formal regulatory pathways. The European Commission has signaled interest in creating a dedicated repurposing authorization procedure in its Pharmaceutical Strategy for Europe, published in 2020, which identified support for repurposing of off-patent medicines as a priority. Whether that signals an actual legislative proposal in the near term is uncertain.
For companies running global repurposing programs, the practical implication is that EU and U.S. regulatory strategies cannot be fully aligned without different sponsorship arrangements in each jurisdiction. A U.S. 505(b)(2) NDA filed by an academic sponsor under a new labeling-only pathway would not automatically translate into an EU marketing authorization application by the same sponsor.
Advanced Market Commitments and Pull Incentives for Generic Repurposing
The most durable solution to the commercial underinvestment problem may be pull incentives that reward successful generic repurposing with guaranteed revenue regardless of whether the drug can command premium pricing. Advanced market commitments (AMCs) guarantee a purchaser price for a successfully developed product in advance of development completion, giving developers revenue certainty that de-risks the investment.
AMC mechanisms have been used in vaccine markets, most notably the Gavi AMC for pneumococcal vaccines, which committed $1.5 billion in donor funds to guarantee purchase of a new pneumococcal conjugate vaccine at above-commodity prices. That commitment attracted commercial manufacturer participation in vaccine development that would not have occurred based on expected developing-country pricing alone.
An AMC structure for generic repurposing would work similarly: a government health system or multi-payer consortium commits in advance to purchasing a successfully licensed generic repurposed drug at a price sufficient to generate adequate return on the clinical development investment. The developer, whether commercial or academic, runs the trials knowing that success leads to a guaranteed market at a guaranteed price. The drug is then sold to the payer pool at the committed price, not at the commodity generic price.
The economics need not be extravagant. If the U.S. Medicare program committed to purchasing a successfully licensed generic repurposed drug at $500 million over 10 years for any program that passed Phase III and received an FDA-approved label update, that commitment would be sufficient to support clinical development investment of $50-100 million with adequate return. For a drug that reduces Medicare costs by reducing hospitalizations or disease progression, the $500 million commitment would likely generate net savings.
The political barrier is that AMCs require upfront government commitment before clinical results are available, which requires Congressional action and appropriations. The prize mechanism, which awards a lump sum payment to a successful developer in exchange for the drug entering the public domain at commodity pricing, is structurally simpler but produces less predictable developer incentives.
Key Takeaways, Part VIII
The FAS labeling-only 505(b)(2) proposal is the most specific and actionable legislative option for resolving the non-manufacturer regulatory gap in the U.S. It requires a statutory amendment. The EU’s repurposing authorization gap is acknowledged by the European Commission but has not yet produced a formal legislative proposal. International harmonization of repurposing regulatory pathways is a medium-term aspiration that requires parallel legislative action in multiple jurisdictions. Advanced market commitments for generic repurposing could de-risk commercial investment without requiring premium drug pricing, but require upfront government appropriations. The prize mechanism is simpler to implement but produces weaker developer incentives than an AMC.
Investment Strategy Note
Investors and portfolio managers evaluating generic repurposing program bets should track legislative activity around the FAS labeling-only 505(b)(2) proposal and the EU Pharmaceutical Strategy implementation timeline. If the labeling-only pathway passes into law, programs that have already completed Phase III trials and are waiting only for a filing mechanism become immediately actionable. A portfolio of such programs, funded through philanthropic or NIH support and waiting on a regulatory pathway, represents a significant option on legislative success. The programs with the clearest near-term value on that option are those in large-indication settings (oncology, CNS) where no orphan designation exists but compelling Phase III evidence has been generated, specifically metformin in breast cancer, aspirin in colorectal cancer prevention, and fluvoxamine in post-COVID neurological sequelae.
Conclusion
Off-patent drugs with new indications are not a niche opportunity. They represent a category of validated, globally available, low-cost molecules where the clinical evidence for new therapeutic uses often already exists, or can be generated at a fraction of the cost of novel drug development. The barriers to translating that evidence into formally licensed, reimbursed, widely accessible therapies are regulatory, economic, and structural, and they are solvable.
The regulatory gap is specific and addressable: the FDA needs a non-manufacturer filing pathway. Congress needs to authorize it. The EU needs a dedicated repurposing marketing authorization procedure. The IP gap is manageable when orphan designation is available or when proprietary reformulation creates enforceable Orange Book-listed patents. The economic gap requires either orphan-level pricing power, a reformulation IP strategy that supports specialty pricing, or a government pull incentive that guarantees commercial return.
The companies and academic institutions that navigate all three gaps, securing orphan designation where available, developing proprietary formulations that create patentable IP, and engaging government health systems around advanced market commitments, are the ones that will extract value from this underexploited category. The public health case for solving these problems does not need amplification. The commercial case is real and, for programs positioned correctly, compelling.
This analysis is for informational purposes for pharmaceutical industry professionals, IP teams, and institutional investors. Nothing herein constitutes legal or investment advice. All regulatory and litigation determinations are fact-specific.