Ranitidine - Generic Drug Details
✉ Email this page to a colleague
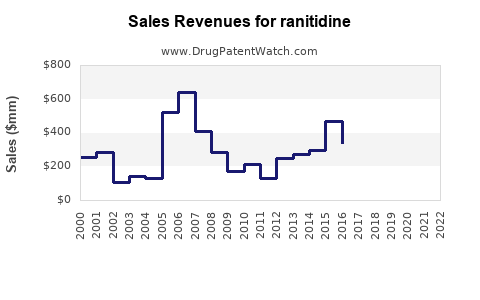
Summary for ranitidine
| US Patents: | 0 |
| Tradenames: | 9 |
| Applicants: | 48 |
| NDAs: | 85 |
| Drug Master File Entries: | 43 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 51 |
| Clinical Trials: | 111 |
| Patent Applications: | 5 |
| Drug Prices: | Drug price trends for ranitidine |
| What excipients (inactive ingredients) are in ranitidine? | ranitidine excipients list |
| DailyMed Link: | ranitidine at DailyMed |
Recent Clinical Trials for ranitidine
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Cairo University | NA |
| Beijing Wehand-Bio Pharmaceutical Co., Ltd | PHASE1 |
| Cancer Institute and Hospital, Chinese Academy of Medical Sciences | PHASE1 |
Generic filers with tentative approvals for RANITIDINE
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Get Started Free | ⤷ Get Started Free | 15MG/ML | Solution; Oral |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.