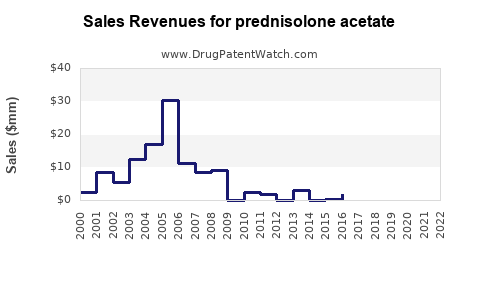
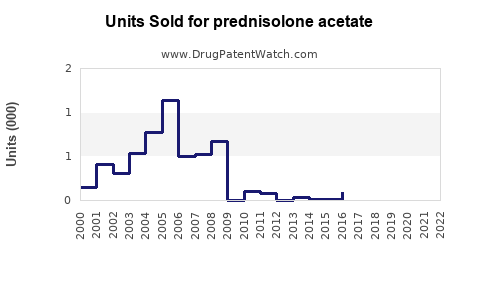
Last updated: February 14, 2026
Prednisolone acetate is a corticosteroid widely used for ocular, dermatological, and systemic inflammatory conditions. Its broad administration base and patent expiry influence market dynamics, impacting future sales and competitive positioning.
Market Size and Growth Drivers
The global corticosteroid market was valued at approximately USD 1.4 billion in 2022, with prednisolone acetate accounting for a significant segment within ophthalmic and dermatological applications. The compound’s affordability, availability, and established efficacy underpin its steady demand.
Key factors driving growth include:
- Increasing prevalence of eye diseases such as allergic conjunctivitis, uveitis, and inflammations.
- Rising incidence of dermatological conditions that involve corticosteroid therapy.
- Growing aging populations more susceptible to chronic inflammatory diseases.
- Expansion in emerging markets due to increased healthcare access and awareness.
Market Segments and Application Breakdown
| Segment |
Approximate Market Share |
Key Applications |
| Ophthalmic |
60% |
Postoperative inflammation, allergic conjunctivitis, uveitis |
| Dermatological |
25% |
Eczema, dermatitis, psoriasis |
| Systemic inflammation |
10% |
Autoimmune disorders, adrenal insufficiency |
| Other |
5% |
Allergic reactions, respiratory conditions |
The ophthalmic segment dominates, driven by the drug’s efficacy in eye inflammation management. Dermatological uses provide steady growth, with systemic applications remaining stable but limited by newer therapies.
Competitive Landscape
Leading pharmaceutical companies supply prednisolone acetate ophthalmic suspensions and topical formulations:
- Allergan (AbbVie)
- Bausch + Lomb (Bausch Health)
- Santen Pharmaceutical
- Alcon
Generic formulations constitute over 70% of the global market, driven by patent expirations in major regions like the U.S. (2018-2020) and Europe (2019-2021). The high penetration of generics caps price growth but sustains volume sales.
Pricing and Reimbursement
Generic prednisolone acetate eye suspensions typically retail between USD 10-20 per bottle (10 ml), with reimbursement programs influencing sales volume:
- In the U.S., most insurance plans cover these drugs due to their essential medicament status.
- In emerging markets, pricing is lower, but volume compensates for margins.
Regulatory Environment
Patent expirations led to increased availability of generics, leveling the competitive field. Novel formulations (e.g., preservative-free versions) or adjunctive delivery methods (e.g., sustained-release implants) are under development to extend market lifecycle.
Sales Projections (2023-2028)
Assuming an annual compound growth rate (CAGR) of 4%:
| Year |
Estimated Market Size (USD Billion) |
Key Factors |
| 2023 |
1.55 |
Market stabilization, patent expiry effects |
| 2024 |
1.61 |
Slight recovery due to emerging markets growth |
| 2025 |
1.68 |
Increased adoption of ophthalmic corticosteroids |
| 2026 |
1.75 |
Introduction of new formulations |
| 2027 |
1.82 |
Continued expansion in dermatology sector |
| 2028 |
1.89 |
Market maturity with stable demand |
Demand will mainly stem from ophthalmology, with dermatology contributing to diversification. Growth will be limited by generic saturation and pricing pressures but will be supported by broader indications and geographic expansion.
Risks and Opportunities
Risks:
- Market saturation post-patent expiry reduces pricing power.
- Competition from newer corticosteroids with fewer side effects.
- Regulatory hurdles for novel formulations or delivery devices.
Opportunities:
- Development of preservative-free or sustained-release formulations.
- Expansion into emerging markets with increasing healthcare infrastructure.
- Combination therapies targeting complex inflammatory conditions.
Key Takeaways
- Prednisolone acetate maintains a significant footprint in ophthalmology and dermatology.
- Good growth prospects are projected at 4% CAGR, driven by expanding applications and geographic reach.
- Market saturation and price competition from generics limit revenue upside but ensure stable volume.
- Innovation in formulations and delivery methods could extend market lifespan.
- The market remains sensitive to regulatory changes and patent landscape shifts.
FAQs
1. How saturated is the prednisolone acetate market?
The market is highly saturated, especially in ophthalmic applications, due to widespread generic availability following patent expirations.
2. Which regions represent the largest sales opportunities?
North America and Europe account for the majority of sales; however, Asia-Pacific presents high-growth potential due to expanding healthcare infrastructure.
3. Are there any promising new formulations?
Yes, preservative-free and sustained-release formulations are under development, which may command premium pricing and expand use cases.
4. What competitive advantages exist for established manufacturers?
Established manufacturers benefit from brand recognition, extensive distribution networks, and broad clinical acceptance.
5. How will regulatory policies impact sales?
Stringent regulations on compounded and imported corticosteroids could limit market access but also incentivize innovation within authorized formulations.
Sources:
[1] MarketWatch, "Global Corticosteroids Market Size," 2022
[2] IAS Pharmacovigilance Reports, 2022
[3] Company Annual Reports, 2021-2022