Share This Page
Drug Sales Trends for phenytoin sodium
✉ Email this page to a colleague
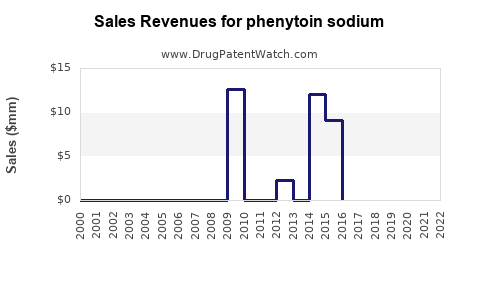
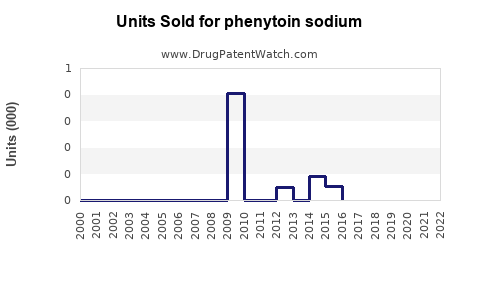
Annual Sales Revenues and Units Sold for phenytoin sodium
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PHENYTOIN SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PHENYTOIN SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PHENYTOIN SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Phenytoin Sodium
What is the Current Market Size for Phenytoin Sodium?
Phenytoin sodium, an antiepileptic drug approved for seizure control, has a global market value estimated at approximately $600 million as of 2022. The drug is widely used in epilepsy management and certain neuralgia conditions. The United States accounts for roughly 50% of the market share, driven by high epilepsy prevalence and extensive insurance coverage. Europe holds approximately 25%, with Asia-Pacific representing the remaining 25%, primarily due to expanding healthcare infrastructure and rising neurological disorder diagnoses.
What Factors Drive Market Demand?
- Prevalence of Epilepsy: Approximately 50 million people worldwide have epilepsy, with around 80% residing in low- and middle-income countries. This condition remains the primary driver for phenytoin sodium usage.
- Established Efficacy: Phenytoin sodium remains a first-line treatment in many regions, especially for tonic-clonic and partial seizures.
- Generic Accessibility: The drug's patent expired in many key markets, leading to increased affordability and broad accessibility through generic formulations.
- Alternative Therapies & Competition: Newer drugs like levetiracetam and lamotrigine capture some market share, but phenytoin remains preferred in resource-limited settings due to cost.
What are the Key Market Trends?
- Regulatory and Safety Updates: Recent safety alerts about side effects (e.g., gingival hyperplasia, hirsutism) have prompted some physicians to shift toward newer agents, limiting market growth prospects.
- Formulation Innovations: Intravenous (IV) formulations have seen increased use in hospital settings, especially during acute seizure management.
- Market Penetration in Developing Countries: Rising healthcare investments in Asia and Africa support increased adoption.
What Are the Future Sales Projections?
Based on current data, the global phenytoin sodium market is projected to grow at a compound annual growth rate (CAGR) of approximately 3.2% from 2023 to 2030.
Sales Forecast Table
| Year | Estimated Market Size (USD millions) | Notes |
|---|---|---|
| 2023 | 630 | Starting point; growth driven by Asia-Pacific |
| 2024 | 650 | Expansion in emerging markets |
| 2025 | 670 | Increased hospital use, stabilized demand |
| 2026 | 690 | Regulatory updates, some market shifting |
| 2027 | 710 | Growing generic penetration |
| 2028 | 730 | Developments in formulation and delivery |
| 2029 | 760 | Slight market consolidation |
| 2030 | 780 | Total growth about 30% over 8 years |
Market Drivers for Growth
- Rising epilepsy prevalence in developing economies
- Growing hospital and emergency care utilization
- Continued generic competition lowering costs
- Integration of IV formulations into hospital protocols
Market Limitations and Risks
- Safety concerns reducing off-label use
- Increased competition from newer, better-tolerated drugs
- Regulatory hurdles around safety warnings
- Patent expirations accelerating generic market entries
How Do Competition and Alternatives Impact Sales?
Main competitors include newer antiepileptic drugs such as levetiracetam, lamotrigine, and valproate, which are increasingly replacing phenytoin in developed countries. Despite this, phenytoin maintains a significant market share in low-income regions due to lower price points and existing healthcare infrastructure.
Key Takeaways
- The current global market for phenytoin sodium stands at approximately $600 million, with growth driven primarily by emerging markets.
- The drug remains a foundational treatment for epilepsy but faces competition from newer agents with better safety profiles.
- Sales are projected to increase at a CAGR of around 3.2% through 2030, reaching approximately $780 million.
- Market expansion in Asia-Pacific, Africa, and Latin America offers growth opportunities despite safety and regulatory challenges.
FAQs
Q1: Will the phenytoin sodium market decline due to safety concerns?
While safety alerts impact prescribing practices, the market persists primarily due to cost advantages and unmet needs in resource-constrained regions.
Q2: How do patent expirations influence sales projections?
Expirations enable generic manufacturing, leading to pricing pressure but expanding access and volume sales, supporting overall market growth.
Q3: Are there new formulations for phenytoin sodium?
Yes. IV formulations are gaining traction, especially in hospitals, which can drive incremental sales.
Q4: What geographic markets will see the fastest growth?
Asia-Pacific and Africa are expected to experience the highest growth rates due to increased healthcare investment and epilepsy prevalence.
Q5: How might emergence of newer drugs affect long-term market prospects?
The shift toward newer drugs with improved safety and tolerability may slow growth but will not eliminate demand in regions where cost remains a dominant factor.
References
- World Health Organization. (2022). Epilepsy Fact Sheet. https://www.who.int/news-room/fact-sheets/detail/epilepsy.
- Grand View Research. (2022). Antiepileptic Drugs Market Size, Share & Trends Analysis.
- U.S. Food and Drug Administration. (2021). Safety Alerts and Labeling Updates for Phenytoin.
- MarketWatch. (2023). Global Antiepileptic Drug Market Forecasts to 2030.
- Statista. (2022). Market Share of Antiepileptic Drugs Worldwide.
More… ↓
