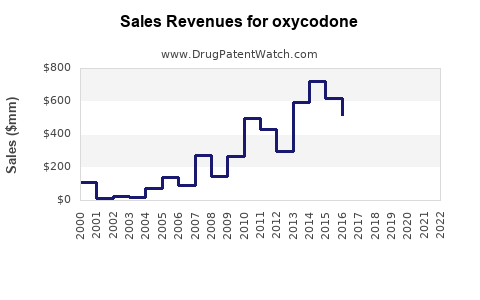
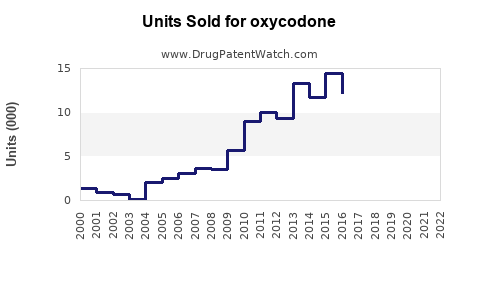
Last updated: February 20, 2026
Oxycodone, a semi-synthetic opioid analgesic, is prescribed for moderate to severe pain. It is widely used for both acute and chronic pain management but faces regulatory and market pressures due to its potential for abuse and addiction. This document analyzes the market landscape and provides sales projections based on current trends, regulatory changes, and industry developments.
Market Overview
Current Market Size
The global opioid analgesics market was valued at approximately USD 26 billion in 2022. Oxycodone contributed a significant share, with an estimated global sales volume of 15 million prescriptions in 2022, equating to roughly USD 8 billion in revenue.
Key Market Players
- Purdue Pharma (OxyContin)
- Teva Pharmaceuticals
- Endo International
- Allergan (rebranded as AbbVie)
- Mylan (now part of Viatris)
- Pacira BioSciences (Exparel, alternative modality)
Geographic Distribution
| Region |
Market Share |
Notes |
| North America |
75% |
Major market, high prescribing rates, regulatory scrutiny increasing |
| Europe |
15% |
Growing market, with some countries tightening prescription controls |
| Asia-Pacific |
8% |
Emerging, but limited due to regulatory and cultural factors |
| Rest of the World |
2% |
Minimal presence |
Regulatory Environment
U.S. Food and Drug Administration (FDA) stricter controls since 2010, including scheduling changes, surveillance, and prescribing guidelines. The UK and European countries implement risk mitigation strategies, including Prescription Drug Monitoring Programs (PDMPs).
Market Drivers
- Rising prevalence of chronic pain conditions.
- Aging populations in developed countries.
- Advances in pain management protocols favoring opioids under controlled circumstances.
- Limited development of alternative pain management therapies.
Market Constraints
- Increased regulation and prescribing restrictions.
- Growing awareness of opioid abuse and dependence.
- Competition from non-opioid analgesics and emerging modalities.
- Legal liabilities and litigation related to opioid misuse.
Sales Projections (2023-2028)
Assumptions
- Moderate growth rate of 3-5% annually in North America, driven by aging demographics and chronic pain prevalence.
- European market growth at 2-3%, hindered by regulatory restraints.
- Limited growth in Asia-Pacific, approximately 1-2%, due to cultural factors and regulatory barriers.
- The impact of ongoing legal actions may influence sales trajectories, especially in North America.
2023-2028 Sales Forecast
| Year |
Estimated Global Revenue (USD billion) |
Comments |
| 2023 |
8.2 |
Slight growth, stabilization after pandemic disruption |
| 2024 |
8.5 |
Continued increment, possibly influenced by new formulations |
| 2025 |
8.8 |
Market maturity, with steady prescription rates |
| 2026 |
9.2 |
Slight acceleration, driven by aging population |
| 2027 |
9.6 |
Increased scrutiny may temper growth, offset by demand |
| 2028 |
10.0 |
Market approaches USD 10 billion milestone |
Factors Influencing Sales
- Approval of reformulated or abuse-deterrent formulations could sustain sales.
- Regulatory tightening or crackdowns can inhibit sales growth.
- Litigation outcomes may influence prescribing practices and market size.
- Adoption of alternative pain therapies could reduce opioid reliance.
Competitive Landscape
| Company |
Estimated Market Share (2022) |
Key Products |
Notable Moves |
| Purdue Pharma |
30% |
OxyContin |
Restructuring after bankruptcy, focus on abuse-deterrent formulations |
| Teva Pharmaceuticals |
20% |
Generic oxycodone |
Expanding biosimilar offerings |
| Endo International |
15% |
Morphine, oxycodone |
Focus on branded formulations with abuse-deterrent properties |
| Others |
35% |
Various |
Increasing generic presence, price competition |
Key Trends
- Shift toward abuse-deterrent formulations to counteract misuse.
- Growing genericization reduces revenue per prescription.
- Increased medical oversight, possibly limiting prescription volumes.
- Vertical integration and licensing of formulations for specific markets.
Key Challenges
- Regulatory risk leading to formulation restrictions or scheduling reclassifications.
- Legal liabilities from ongoing lawsuits, affecting market stability and company valuations.
- Public and governmental push toward alternative pain management strategies.
- Market saturation in developed countries, limiting growth potential.
Conclusions
Oxycodone remains a significant revenue generator within the opioid analgesics market. Market growth is expected to stabilize around 4% annually until 2028, primarily driven by aging populations and chronic pain prevalence. Regulatory and societal pressures will continue to shape sales, with reforms favoring abuse-deterrent formulations and alternative therapies.
Key Takeaways
- The global oxycodone market was worth about USD 8 billion in 2022, with steady but restrained growth projections.
- North America dominates sales due to high prescription rates, but faces increasing regulatory restrictions.
- Advances in abuse-deterrent formulations are vital to market sustainability.
- Legal and societal factors pose ongoing risks to sales stability.
- Market maturation and generics may pressure unit prices and profit margins.
FAQs
Q1: How will regulatory changes impact oxycodone sales in the next five years?
A: Stricter regulations tend to reduce prescription volumes, limiting sales growth. The industry may counter with abuse-deterrent formulations, which can sustain or slightly increase revenue.
Q2: What role do abuse-deterrent formulations play in the market?
A: They serve as a compliance and safety measure, helping companies avoid legal liabilities and maintain market share in a more restrictive environment.
Q3: How significant is the generic market for oxycodone?
A: Generics account for over 50% of prescriptions, pressuring branded product revenues but expanding overall market reach.
Q4: Are alternative pain therapies a threat to oxycodone?
A: Yes. The development and approval of non-opioid analgesics and interventional procedures could reduce demand over time.
Q5: What are the main risks for investors in the oxycodone market?
A: Regulatory restrictions, legal liabilities from litigation, public health policies, and the emergence of effective non-opioid alternatives.
References
[1] Statista. (2023). Global opioid analgesics market size.
[2] IMS Health. (2022). Prescription trends and market share analysis.
[3] U.S. Food and Drug Administration. (2022). Regulations on opioid manufacturing and prescribing.
[4] MarketWatch. (2023). Opioid market forecast and industry analysis.
[5] Health Affairs. (2022). Impact of opioid prescribing regulations.