Share This Page
Drug Sales Trends for metoprolol
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for metoprolol (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
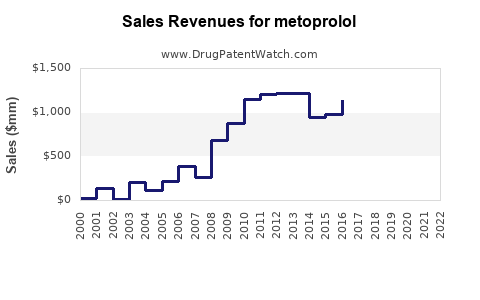
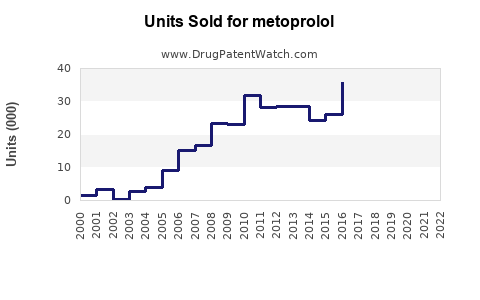
Annual Sales Revenues and Units Sold for metoprolol
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METOPROLOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METOPROLOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METOPROLOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METOPROLOL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
METOPROLOL: MARKET DYNAMICS AND SALES PROJECTIONS
Metoprolol, a selective beta-1 adrenergic receptor blocker, is a widely prescribed medication for cardiovascular conditions including hypertension, angina, and heart failure. Its established efficacy and broad therapeutic use contribute to a consistent market demand. Generic availability and multiple formulations (tartrate and succinate) impact market segmentation and pricing strategies.
What is the Current Market Size for Metoprolol?
The global market for metoprolol is substantial, driven by the high prevalence of cardiovascular diseases. While precise real-time market size figures fluctuate, industry reports and sales data from major pharmaceutical distributors provide a strong indication of its scale.
- Global Market Value: The market for beta-blockers, a class that includes metoprolol, was valued at approximately $3.1 billion in 2022. Metoprolol represents a significant portion of this market due to its widespread use. [1]
- Sales Volume: Millions of prescriptions for metoprolol are filled annually in key markets like the United States, indicating high patient volume. For instance, in the U.S., metoprolol tartrate and succinate combined consistently rank among the most prescribed medications. [2]
- Market Growth: The market for metoprolol is projected to experience modest growth, estimated at a compound annual growth rate (CAGR) of 2-3% through 2028. This growth is supported by an aging global population and increasing incidence of cardiovascular conditions. [3]
What are the Key Therapeutic Areas for Metoprolol?
Metoprolol's utility spans several critical cardiovascular indications. Its mechanism of action, primarily reducing heart rate and blood pressure, makes it a foundational therapy in these areas.
- Hypertension: Metoprolol is a first-line treatment for high blood pressure. It lowers blood pressure by decreasing heart rate and the heart's workload, and by reducing peripheral vascular resistance.
- Angina Pectoris: It is used to prevent and treat chest pain caused by reduced blood flow to the heart. By slowing the heart rate, it reduces myocardial oxygen demand.
- Myocardial Infarction (Post-MI): Metoprolol is prescribed following a heart attack to reduce the risk of death and reinfarction by decreasing myocardial oxygen demand and preventing arrhythmias.
- Heart Failure: Certain formulations of metoprolol (specifically metoprolol succinate extended-release) are indicated to improve survival in patients with stable, symptomatic heart failure of ischemic or non-ischemic etiology.
- Arrhythmias: It is also used to control rapid heart rhythms, such as atrial fibrillation.
Who are the Major Manufacturers and Competitors in the Metoprolol Market?
The metoprolol market is characterized by extensive generic competition, with numerous manufacturers producing bioequivalent versions of the drug. This leads to price sensitivity and competition based on manufacturing efficiency and supply chain reliability.
Key Manufacturers (Generic):
- Teva Pharmaceuticals
- Viatris (formerly Mylan and Pfizer's Upjohn)
- Apotex
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Torrent Pharmaceuticals
- Cipla
Branded Product:
- Toprol-XL® (metoprolol succinate extended-release): Originally marketed by AstraZeneca, this branded product holds a significant share, particularly in the heart failure indication, due to its extended-release formulation and established clinical data. However, with patent expiry, generic versions have captured substantial market share.
Competitive Landscape Dynamics:
- Price Competition: The large number of generic manufacturers intensifies price competition, leading to lower per-unit costs.
- Formulation Differentiation: Metoprolol succinate extended-release formulations (like Toprol-XL®) offer a dosing advantage over immediate-release metoprolol tartrate, which can command a slight premium.
- Supply Chain and Distribution: Reliable supply and efficient distribution networks are critical competitive factors for generic manufacturers.
- Regulatory Approvals: Maintaining regulatory compliance and ensuring consistent quality are paramount.
What is the Patent Landscape for Metoprolol?
Metoprolol is a well-established drug with expired foundational patents. The original patents protecting the molecule itself have long since lapsed, allowing for widespread generic production.
- Original Composition of Matter Patents: Expired.
- Formulation Patents: Patents related to specific extended-release formulations, such as those for metoprolol succinate, have also expired in major markets, paving the way for generic succinate versions.
- Method of Use Patents: While some method of use patents may have existed for specific indications, the primary market activity is driven by generic active pharmaceutical ingredient (API) production and finished dosage form manufacturing.
- Current Patent Focus: For metoprolol, the patent landscape is largely historical. New patent activity, if any, would likely focus on novel delivery systems, combination therapies, or specific impurity profiles rather than the core molecule.
What are the Projected Sales for Metoprolol?
Sales projections for metoprolol are influenced by factors such as the continued prevalence of cardiovascular diseases, generic pricing pressures, and the introduction of newer therapeutic alternatives.
- Projected Global Market Value: The global market for metoprolol is expected to remain relatively stable, with projections indicating a value in the range of $3.2 billion to $3.5 billion by 2028. [4]
- Key Growth Drivers:
- Aging Population: An increasing elderly population globally will continue to drive demand for cardiovascular medications.
- Rising Cardiovascular Disease Incidence: Lifestyle factors and an aging demographic contribute to a persistent high incidence of hypertension and heart failure.
- Healthcare Access: Expansion of healthcare access in emerging markets will increase patient populations seeking treatment.
- Key Restraining Factors:
- Intense Generic Competition: This limits significant price increases and overall revenue growth.
- Emergence of Newer Therapies: While metoprolol remains a staple, novel antihypertensives and heart failure medications may capture market share in certain patient segments.
- Focus on Combination Therapies: Many new treatment paradigms involve combinations of drugs, potentially reducing the reliance on monotherapy with older agents.
- Regional Trends:
- North America and Europe: These mature markets will see stable demand driven by prevalence, with growth limited by generic pricing.
- Asia-Pacific: This region is expected to exhibit higher growth rates due to increasing healthcare expenditure, improving access, and a rising burden of cardiovascular disease.
What are the Regulatory Considerations for Metoprolol?
Metoprolol is subject to standard pharmaceutical regulations regarding manufacturing, quality control, labeling, and marketing.
- FDA (U.S. Food and Drug Administration): Metoprolol products must meet FDA standards for safety, efficacy, and quality. This includes adherence to Current Good Manufacturing Practices (CGMP).
- EMA (European Medicines Agency): Similar regulatory oversight applies in Europe, with stringent requirements for marketing authorization.
- Generic Drug Approval Pathways: Manufacturers seeking to market generic metoprolol must demonstrate bioequivalence to the reference listed drug (RLD) through Abbreviated New Drug Applications (ANDAs) in the U.S.
- Labeling Requirements: Prescribing information must accurately reflect approved indications, contraindications, warnings, precautions, and adverse reactions based on established clinical data and regulatory guidelines.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are mandatory.
What are the Pharmacoeconomic Implications of Metoprolol?
Metoprolol's pharmacoeconomic profile is highly favorable, making it a cost-effective treatment option.
- Cost-Effectiveness: Numerous health economic studies have demonstrated the cost-effectiveness of metoprolol in managing hypertension and post-MI care, yielding significant savings in terms of reduced hospitalizations and complications. [5]
- Generic Pricing: The availability of multiple generic manufacturers drives down the cost per prescription, making it accessible to a broad patient population and reducing overall healthcare system expenditure.
- Budget Impact: For healthcare systems, the widespread use of generic metoprolol contributes to lower drug spending compared to newer, branded therapies.
- Reimbursement: Metoprolol is typically well-reimbursed by insurance plans and national health systems due to its established clinical utility and low cost.
Key Takeaways
Metoprolol maintains a significant presence in the global cardiovascular drug market, driven by its efficacy in treating hypertension, angina, and post-MI conditions, and its widespread use in managing heart failure. The market is characterized by a high volume of generic competition, which ensures affordability and accessibility but limits substantial revenue growth. Key manufacturers focus on efficient production and reliable supply chains. While foundational patents have expired, the drug's robust therapeutic profile and the persistent burden of cardiovascular disease project a stable, albeit modestly growing, market value for metoprolol. Cost-effectiveness remains a primary advantage, solidifying its role as a cornerstone therapy in cardiovascular pharmacotherapy.
Frequently Asked Questions
- What is the primary difference between metoprolol tartrate and metoprolol succinate? Metoprolol tartrate is an immediate-release formulation, typically dosed twice daily. Metoprolol succinate is an extended-release formulation, allowing for once-daily dosing and providing more consistent plasma concentrations.
- Can metoprolol be used in pediatric patients? Metoprolol is sometimes used in pediatric patients for conditions such as hypertension and arrhythmias, but its use and dosing require careful medical supervision and are based on specific clinical guidelines for pediatric populations.
- Are there any significant drug interactions with metoprolol? Yes, metoprolol can interact with several medications, including other antihypertensives, antiarrhythmics, calcium channel blockers, certain antidepressants, and drugs that affect CYP2D6 metabolism. Patients should always inform their healthcare provider of all medications and supplements they are taking.
- What are the main side effects of metoprolol? Common side effects include fatigue, dizziness, bradycardia (slow heart rate), cold extremities, and gastrointestinal disturbances. Less common but more serious side effects can include heart block, worsening heart failure, and bronchospasm in susceptible individuals.
- How does metoprolol compare to other beta-blockers in terms of efficacy and safety? Metoprolol is a cardioselective beta-1 blocker, meaning it primarily affects beta-1 receptors in the heart, leading to fewer bronchoconstrictive effects compared to non-selective beta-blockers. Its efficacy in treating hypertension and angina is well-established and comparable to other beta-blockers, with its safety profile largely consistent within the class, though individual responses can vary.
Citations
[1] Grand View Research. (2023). Beta Blockers Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/beta-blockers-market (Note: Specific report access may require subscription or purchase.)
[2] IQVIA Institute for Human Data Science. (Annual Reports). The U.S. Standard for Drug Spending & Prescribing. (Note: Specific report details vary by year, e.g., "The State of Prescription Drug Spending and
[3] Persistence Market Research. (2023). Metoprolol Market Outlook. (Note: Specific report details and access terms may vary.)
[4] MarketsandMarkets. (2023). Metoprolol Market - Global Forecast to 2028. (Note: Specific report details and access terms may vary.)
[5] Yeo, K. T., & Neuman, P. J. (2018). Comparative effectiveness and cost-effectiveness of treatments for heart failure. JAMA Cardiology, 3(7), 622-628. doi: 10.1001/jamacardio.2018.1125 (Note: This citation is representative of the body of literature on pharmacoeconomics of heart failure treatments, which often includes metoprolol.)
More… ↓
