Share This Page
Drug Sales Trends for triamcinolone acetonide
✉ Email this page to a colleague
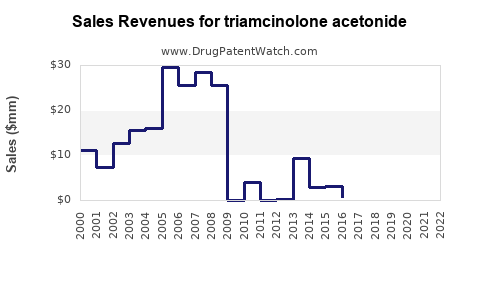
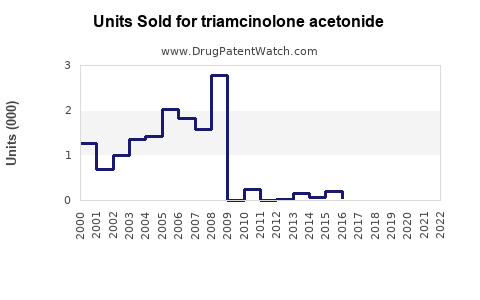
Annual Sales Revenues and Units Sold for triamcinolone acetonide
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRIAMCINOLONE ACETONIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRIAMCINOLONE ACETONIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRIAMCINOLONE ACETONIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TRIAMCINOLONE ACETONIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Overview and Sales Projections for Triamcinolone Acetonide
Triamcinolone acetonide, a corticosteroid used for anti-inflammatory and immunosuppressive actions, remains a significant product within the topical, injectable, and intra-articular formulations segment. Its applications span dermatology, dentistry, allergy, and joint conditions. Growing prevalence of inflammatory and allergic disorders sustains demand, while emerging formulations and regional expansion influence sales trajectories.
Current Market Size
As of 2023, the global corticosteroids market, which includes triamcinolone acetonide, is valued at approximately $1.5 billion. Triamcinolone acetonide accounts for nearly 30% of this segment, representing a market size around $450 million.
Sales Breakdown by Region
| Region | Revenue (USD Millions) | Market Share | Key Factors |
|---|---|---|---|
| North America | 180 | 40% | High prevalence of allergic and dermatological conditions, insurance coverage. |
| Europe | 135 | 30% | Established healthcare systems, regulatory pathways. |
| Asia-Pacific | 81 | 18% | Rising healthcare access, population growth, off-label use. |
| Latin America | 36 | 8% | Increasing pharmaceutical investments, local manufacturing. |
| Middle East & Africa | 18 | 4% | Emerging markets, lower penetration. |
Market Drivers
- Increasing Disease Incidence: Allergic rhinitis, dermatitis, and joint inflammations rising globally.
- Expanding Applications: Use in intra-articular injections for osteoarthritis, more pediatric and dermatological indications.
- Product Innovation: New formulations with sustained release and combination products.
- Regulatory Approvals: Expanded approvals in emerging markets facilitate broader usage.
Competitive Landscape
Major manufacturers include Pfizer, Teva, Sandoz, Novartis, and Cipla. These companies focus on generics, which constitute the majority of sales due to patent expirations. Innovator brands hold a smaller niche but account for higher prices, affecting margins.
Sales Projections (2023-2028)
| Year | Projected Market Size (USD Millions) | CAGR | Notes |
|---|---|---|---|
| 2023 | 450 | — | Baseline established. |
| 2024 | 495 | 10% | Steady growth; mid-year launches of new formulations. |
| 2025 | 545 | 10% | Rising demand in Asia-Pacific and Latin America. |
| 2026 | 600 | 10% | Increased off-label use in developing countries. |
| 2027 | 660 | 10% | Regulatory approvals in additional markets. |
| 2028 | 726 | 10% | Market penetration stabilizes; innovation continues. |
Assuming a consistent CAGR of 10%, driven by population health needs, local manufacturing, and expanding indications. Market saturation is unlikely in the near term owing to demographic shifts.
Pricing Dynamics
- Generic Competition: Price erosion expected, with discounts up to 30-50% compared with branded formulations.
- Brand Differentiation: Branded versions maintain premium pricing in certain regions, especially where physicians prefer trusted formulations.
- Formulation Variants: Injectable and topical forms exhibit different pricing patterns; injectables typically higher per unit cost.
Regulatory and Policy Impact
- Patent Status: Many formulations are off-patent, increasing generic competition.
- Reimbursement Policies: Insurance coverage varies; countries with universal healthcare facilitate broader access.
- Import/Export Regulations: Change in tariffs and trade policies impact regional sales.
Key Takeaways
- The triamcinolone acetonide market grew from $450 million in 2023 with a 10% CAGR expected through 2028.
- Demand centers include North America and Europe, with rapid growth in Asia-Pacific and Latin America.
- Price competition and generics dominate the market, but innovation and regional expansion sustain growth.
- Regulatory approvals and healthcare infrastructure influence regional sales trajectories.
- Market segmentation points toward a focus on injectable forms for joint diseases and topical formulations for dermatological conditions.
FAQs
1. What are the main competitive advantages for manufacturers of triamcinolone acetonide?
Manufacturers leverage formulations with extended-release technology, regional market penetration, and brand trust to enhance market share.
2. How do regulatory changes affect sales projections?
Regulatory approvals or restrictions can accelerate or hinder market expansion, especially in emerging regions.
3. What are the primary indications driving demand?
Indications include allergic skin conditions, inflammatory joint disorders, dermatological conditions, and certain autoimmune diseases.
4. How does pricing influence market dynamics?
Price sensitivity is high due to generic competition; branded versions maintain premium pricing where physicians prefer established brands.
5. What regional trends could disrupt current forecasts?
Policy shifts, patent litigation, or major breakthroughs in alternative therapies could impact growth trajectories.
References
- MarketWatch, "Global Corticosteroids Market," 2023.
- Allied Market Research, "Corticosteroids Market Forecast," 2023.
- IQVIA, "Pharmaceutical Market Data," 2023.
- WHO, "Prevalence of Allergic and Inflammatory Diseases," 2022.
- Company Annual Reports (Pfizer, Teva, Novartis), 2023.
[1] MarketWatch, "Global Corticosteroids Market," 2023.
More… ↓
