Last updated: February 15, 2026
What is Sulindac?
Sulindac is a nonsteroidal anti-inflammatory drug (NSAID) primarily used to treat osteoarthritis, rheumatoid arthritis, and other inflammatory conditions. It is a prodrug metabolized into its active form, sulindac sulfide, which inhibits cyclooxygenase enzymes (COX-1 and COX-2). It is marketed under brand names such as Clinoril.
What is the Current Market Size for Sulindac?
The global NSAID market reached approximately $18.2 billion in 2022, with a compound annual growth rate (CAGR) of around 4.5% projected through 2030 [1]. Sulindac accounts for an estimated 2% of this market, reflecting its niche positioning among older, generic NSAID drugs.
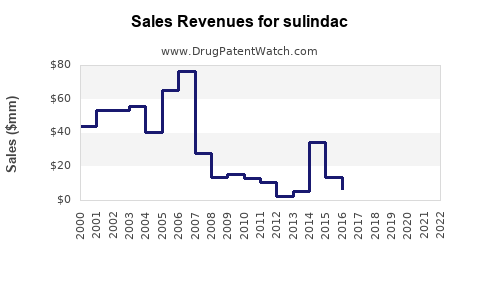
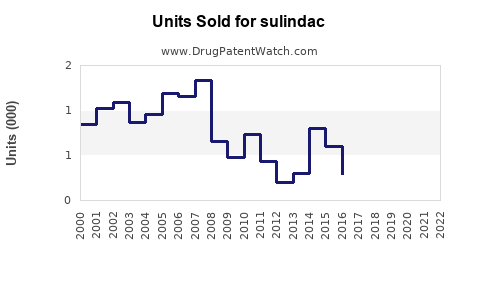
In the U.S., the prescription volume of Sulindac was approximately 1.2 million prescriptions in 2022. The drug is available off-patent, with generic versions dominating market share.
How Competitive is the Sulindac Market?
The NSAID class features high competition:
- Market leaders include ibuprofen, naproxen, and diclofenac.
- Sulindac competes on niche indications and safety profile, primarily in patients intolerant to other NSAIDs.
- Generic manufacturers hold the majority of sales volume, limiting pricing power.
No recent patent protections exist for Sulindac, constraining revenue growth potential without new formulations or indications.
What are the Trends Affecting Future Sales?
Prescribing Trends
Physicians favor NSAIDs with better cardiovascular and gastrointestinal safety profiles. Celecoxib (a selective COX-2 inhibitor) holds a dominant position in that arena, reducing Sulindac’s base market.
Use of older NSAIDs like Sulindac wanes in favor of medications with improved safety profiles or alternative therapies, such as acetaminophen or opioids, though the latter faces regulatory constraints.
Regulatory Environment
No recent FDA restrictions target Sulindac. However, safety concerns associated with GI complications and cardiovascular risks influence prescribing patterns.
Patent and Market Exclusivity
Absence of patent protection since the late 1990s means no exclusivity period, impacting the drug’s ability to sustain premium pricing.
Emerging Indications
Potential research into Sulindac’s role in cancer prevention, particularly colon cancer, exists but remains investigational. If proven, this could unlock new market segments.
What Are Sales Projections for Sulindac?
Based on current prescriber behavior and market trends, sales are expected to decline in the short term:
- Forecasted to decrease at an annual rate of 2-3% over the next five years due to competition and prescriber shifts.
- Estimated U.S. sales revenue in 2023: approximately $60 million.
- Global sales projected to reach roughly $300 million by 2028, driven mainly by emerging markets and increased off-label research.
In the absence of new indications or formulations, Sulindac’s revenue is unlikely to grow significantly beyond current levels.
What Opportunities and Challenges Exist?
Opportunities
- Developing a new formulation with improved safety profile may extend market life.
- Exploring alternate indications, including chemopreventive uses, could create new revenue streams.
Challenges
- High competition from established NSAIDs with better safety profiles.
- Price erosion due to generic proliferation.
- Shifting prescriber preferences toward selective COX-2 inhibitors or non-NSAID pain management options.
Key Takeaways
- Sulindac operates in a saturated, highly competitive NSAID market, with declining sales due to safety concerns and market preferences.
- No patent protections exist, limiting pricing power and revenue growth.
- Short-to-medium-term sales are projected to decline, with limited upside unless new indications or formulations are introduced.
- Market dynamics favor newer, safer NSAIDs and alternative therapies, constraining Sulindac’s position unless specific niche uses are identified.
FAQs
1. Can Sulindac’s market growth be expected to rebound? No. The drug faces market decline without new indications, formulations, or significant safety improvements.
2. Are there ongoing clinical trials for Sulindac? Yes. Investigations into cancer prevention, particularly colon cancer, are underway. Results could influence future sales.
3. How does Sulindac compare to other NSAIDs in safety? It has higher gastrointestinal toxicity risk but may be preferred in specific patient populations intolerant to other NSAIDs.
4. What regulatory risks exist for Sulindac? None currently; however, ongoing safety concerns mean future restrictions are possible if adverse events increase.
5. Is there potential for branded commercialization? Limited, due to patent expiration and intense generic competition. Niche applications offer the primary growth avenue.
References
[1] MarketWatch. "NSAID Market Size, Share & Trends." Published 2022.