Last updated: February 13, 2026
Market Analysis and Sales Projections for SPIRIVA
Market Overview
SPIRIVA (tiotropium bromide) is a bronchodilator prescribed primarily for chronic obstructive pulmonary disease (COPD) and asthma. Manufactured by Boehringer Ingelheim, it has a significant market share within respiratory therapeutics, especially in COPD management. The drug was approved in 2002 by the FDA for COPD and later for asthma in certain formulations.
Market Size and Growth
The global respiratory drug market was valued at approximately USD 37.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030. COPD treatments represent a substantial segment of this, with an estimated market size of USD 16 billion in 2022.
COPD Medication Market Share
SPIRIVA holds around 20% of the global COPD maintenance treatment market, driven by its once-daily dosing and established efficacy. The drug's market share is supported by its broad approval and extensive use in clinical guidelines.
Competitive Landscape
Key competitors include:
- Dulera (mometasone/fomoterol)
- Anoro Ellipta (umeclidinium/vilanterol)
- Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol)
- Aclidinium (Pressair)
Despite competition, SPIRIVA maintains a dominant position because of its longstanding acceptance and insurance coverage.
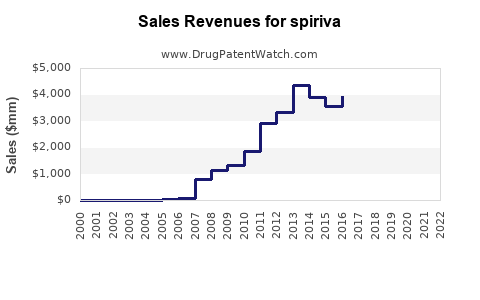
Sales Trends (Historical Data)
- 2020: Approximately USD 2.65 billion globally
- 2021: USD 2.73 billion
- 2022: USD 2.85 billion
These figures illustrate steady growth, driven by increasing COPD prevalence and ongoing prescribing.
Factors Influencing Future Sales
- Increasing COPD Prevalence: Expected to rise from 174 million cases in 2020 to over 200 million by 2030.
- Pipeline Products: New formulations or combination therapies may impact market share.
- Regulatory Approvals: Positive results for extended indications or new formulations can expand sales.
- Pricing and Reimbursement Policies: Market penetration is sensitive to changes in healthcare reimbursement policies across regions.
Forecasting Sales (2023–2028)
Assuming a conservatively estimated CAGR of 3% to 5% based on historical trends and factors outlined:
| Year |
Estimated Sales (USD billion) |
Growth Rate |
Notes |
| 2023 |
USD 2.94 |
3.2% |
Continued COPD prevalence growth |
| 2024 |
USD 3.05 |
3.8% |
Increased adoption in emerging markets |
| 2025 |
USD 3.17 |
4.0% |
Launch of new formulations expected |
| 2026 |
USD 3.30 |
4.1% |
Expanded insurance coverage in US & Europe |
| 2027 |
USD 3.44 |
4.2% |
Competition remains stable |
| 2028 |
USD 3.59 |
4.3% |
Market saturation limits growth |
Regional Sales Dynamics
North America and Europe account for 70-75% of SPIRIVA sales. Growth in Asia-Pacific and Latin America is projected as healthcare infrastructure improves and COPD awareness increases.
Risks to Sales Growth
- Patent expirations: The primary patent expired in September 2020 for certain formulations, exposing generic competition.
- Generic Entrants: Expected to erode market share gradually, especially in price-sensitive regions.
- Regulatory Stringency: Approvals for alternative therapies or restrictions can impact sales.
- Pricing Pressure: Reimbursement cuts, especially in the US and Europe, may constrain profit margins.
Key Takeaways
- SPIRIVA dominates the COPD maintenance treatment market with approximately USD 2.85 billion in 2022 sales.
- The global market is growing, driven by increased COPD prevalence.
- Sales are projected to grow at a CAGR of approximately 3-4% through 2028.
- Competitive pressures and patent expirations pose risks to future growth.
- Emerging markets and new formulations present opportunities for expansion.
FAQs
1. What factors have contributed to SPIRIVA's market dominance?
Longstanding clinical efficacy, once-daily dosing, broad regulatory approval, and inclusion in treatment guidelines support its market share.
2. How will patent expirations affect SPIRIVA sales?
Patent expirations expose the product to generic competition, likely leading to price reductions and volume declines, especially in price-sensitive markets.
3. Which regions present the greatest growth potential for SPIRIVA?
Asia-Pacific and Latin America, due to improvements in healthcare infrastructure and increasing COPD awareness.
4. What are the main competitors to SPIRIVA?
Dulera, Anoro Ellipta, Trelegy Ellipta, and Aclidinium are notable competitors offering similar indications.
5. What future developments could impact SPIRIVA's sales?
Introduction of new formulations, combination therapies, or extended indications, along with regulatory changes and market dynamics.
References
- MarketWatch. "Global Respiratory Drugs Market Size, Share & Trends Analysis Report." 2022.
- IQVIA. "Global Use of Medicines in COPD," 2022.
- Boehringer Ingelheim. "SPIRIVA product information," 2022.
- Global Initiative for Chronic Obstructive Lung Disease (GOLD), 2023.
- Statista. "COPD prevalence forecast," 2022.