Share This Page
Drug Sales Trends for silver sulfadiazine
✉ Email this page to a colleague
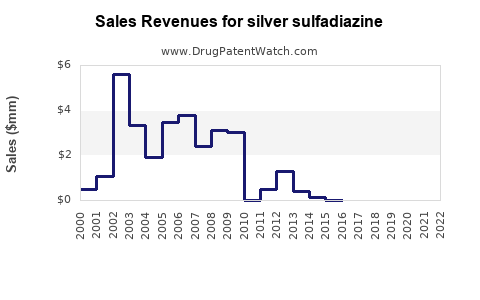
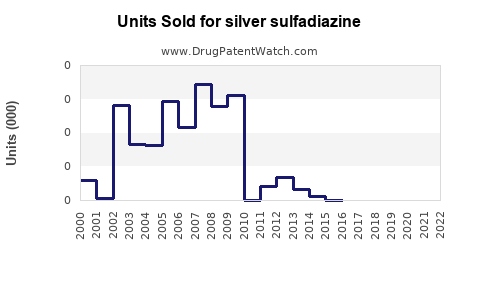
Annual Sales Revenues and Units Sold for silver sulfadiazine
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SILVER SULFADIAZINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SILVER SULFADIAZINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SILVER SULFADIAZINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SILVER SULFADIAZINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SILVER SULFADIAZINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Silver sulfadiazine Market Analysis and Financial Projection
What Is Silver Sulfadiazine and What Are Its Market Fundamentals?
Silver sulfadiazine is a topical antimicrobial agent primarily used to prevent and treat infections in burn wounds. It combines silver ions with sulfadiazine, a sulfonamide antibiotic. Its efficacy in managing burn infections sustains its market presence, but competition from alternative therapies impacts growth.
The global silver sulfadiazine market was valued at approximately $380 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 3-4% over the next five years, reaching roughly $460 million by 2027[1].
What Are the Key Drivers and Constraints Affecting Market Growth?
Drivers
- Increase in burn injuries: Global burn cases are estimated at 180,000 annually, primarily in developing countries[2], sustaining demand for burn wound care products like silver sulfadiazine.
- Rising healthcare expenditure: An increase in healthcare spending supports access to wound care treatments.
- Advances in wound management protocols: Incorporating silver-based dressings to prevent infections enhances product adoption.
Constraints
- Availability of alternative therapies: Newer wound dressings with enhanced antimicrobial properties, such as nanocrystalline silver or antibiotic-loaded dressings, limit market growth.
- Side effects and resistance: Reports of resistance development and adverse reactions (e.g., leukopenia) reduce long-term reliance on silver sulfadiazine.
- Regulatory challenges: Variability in approval processes impacts market access in emerging markets.
Who Are the Key Players in the Silver Sulfadiazine Market?
Major companies include Smith & Nephew, 3M, and Integra LifeSciences. They hold significant market share through product innovation and distribution networks.
| Competitor | Market Share (est.) | Main Products |
|---|---|---|
| Smith & Nephew | ~30% | Aquacel Ag, Acticoat products |
| 3M | ~25% | Tegaderm, Silver Dressings |
| Integra LifeSciences | ~15% | Duraderm, Biobrane |
Small to mid-sized manufacturers account for the remaining share, competing mainly on price and local distribution.
What Are the Sales Projections for Silver Sulfadiazine?
Historical Sales Data (2022)
- North America: $150 million
- Europe: $80 million
- Asia-Pacific: $100 million
- Rest of World: $50 million
Projected Growth (2023-2027)
Assuming stable adoption rates, sales are expected to increase at a CAGR of 3-4%, reaching approximately:
| Year | Estimated Total Sales |
|---|---|
| 2023 | $390 million |
| 2024 | $400 million |
| 2025 | $415 million |
| 2026 | $430 million |
| 2027 | $460 million |
Growth in North America and Europe is slower (~2-3%) due to market saturation, while Asia-Pacific exhibits higher growth (~5%) driven by rising burn cases and healthcare development.
Growth Variables
- Development of newer antimicrobial wound dressings could cannibalize sales.
- Increased burn incidence in emerging markets boosts demand.
- Patent expirations may promote generic entry, influencing pricing and volumes.
How Are Regulatory and Patent Trends Influencing the Market?
The patent life for several formulations has expired, leading to increased generic competition. Regulatory bodies like the FDA and EMA approve silver sulfadiazine as a Class II or III medical device. Emerging markets often experience slower approval timelines, affecting immediate sales expansion.
What Are the Future Opportunities and Risks?
Opportunities
- Developments in nanotechnology for enhanced antimicrobial efficacy.
- Expansion into pediatric and outpatient burn care.
- Integration with advanced wound dressings combining antimicrobial properties with improved healing techniques.
Risks
- Emergence of bacterial resistance potentially reducing effectiveness.
- Regulatory delays or restrictions on silver-based products.
- Competitive pressure from the rise of alternative antimicrobial agents.
Key Takeaways
- The market for silver sulfadiazine was approximately $380 million in 2022, with a projection to reach $460 million by 2027.
- Growth is driven by rising burn trauma, particularly in emerging markets, while competition from alternative therapies constrains expansion.
- Major players include Smith & Nephew, 3M, and Integra LifeSciences, with incremental product innovations and geographic expansion as growth strategies.
- Sales are expected to grow modestly, at a CAGR of 3-4%, with regional variations affecting overall performance.
- Patent expirations and regulatory dynamics influence pricing, competition, and potential market entry of generics and new formulations.
FAQs
1. What clinical indications primarily drive silver sulfadiazine sales?
Burn wound infections are the primary indication, especially second- and third-degree burns, which require antimicrobial prevention.
2. How does resistance impact the long-term use of silver sulfadiazine?
Emerging bacterial resistance and side effects limit its long-term efficacy, prompting development of alternative antimicrobial dressings.
3. Which regions present the most growth opportunities?
Asia-Pacific exhibits the most potential due to increasing burn incidents and expanding healthcare infrastructure.
4. Are there recent innovations in silver-based wound treatments?
Yes, developments include nanocrystalline silver dressings and combination therapies aimed at reducing resistance and improving healing.
5. How does patent expiration affect market dynamics for silver sulfadiazine?
Patent expirations open the market to generics, increasing competition and pressuring prices, but also expanding access in emerging markets.
Citations
[1] MarketWatch, "Silver Sulfadiazine Market Size, Share & Trends," 2022.
[2] WHO, "Burn injuries: overview," 2021.
More… ↓
