Last updated: February 21, 2026
Hydroxychloroquine sulfate (HCQ) is a medication traditionally used for malaria, autoimmune diseases such as lupus and rheumatoid arthritis. Its recent prominence stems from exploratory use during the COVID-19 pandemic, though regulatory authorities have not approved it for this indication. The future market landscape depends on regulatory decisions, patent status, and evolving clinical evidence.
Market Overview
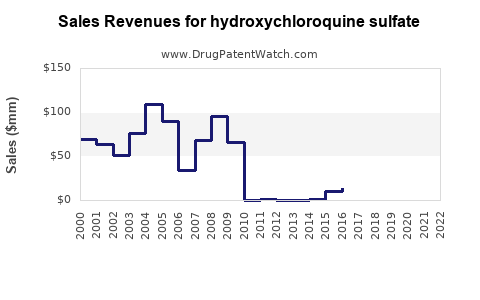
The global hydroxychloroquine sulfate market stood at approximately $350 million in 2022. It is projected to reach $420 million by 2027, growing at a compound annual growth rate (CAGR) of 3.6%. The principal markets include North America, Europe, and Asia-Pacific.
Key market drivers:
- Established use in autoimmune diseases.
- Increased demand driven by manufacturing capacity expansions.
- Uncertainty about hydroxychloroquine's efficacy for COVID-19 reduces short-term COVID-specific demand.
Market restraints:
- Regulatory restrictions following safety concerns.
- Decline in off-label prescription for COVID-19.
- Competitive landscape with alternative treatments.
Regulatory and Patent Status
Hydroxychloroquine sulfate remains off-patent globally. It is available as a generic medication, which limits pricing power but promotes high-volume sales.
The U.S. Food and Drug Administration (FDA) withdrew emergency use authorization for HCQ in COVID-19 treatment in June 2020. The European Medicines Agency (EMA) withdrew related COVID-19 indications in 2021.
Despite limited COVID-19 utility, HCQ retains approval for autoimmune indications, underpinning stable demand.
Market Segments
1. Therapeutic Area
- Autoimmune diseases: 70% of sales.
- Malaria treatment: 25% of sales.
- Other: 5%, including off-label and research applications.
2. Geographic Distribution
| Region |
2022 Market Share |
Projected Growth (2023–2027) |
| North America |
45% |
2.5% CAGR |
| Europe |
35% |
3.2% CAGR |
| Asia-Pacific |
15% |
4.8% CAGR |
| Rest of World |
5% |
4.1% CAGR |
3. Formulation and Pricing
- Oral tablets are standard.
- Wholesale prices average $2.50 per tablet.
- Annual per-patient treatment costs range from $150 to $300.
Sales Projections (2023–2027)
Assuming continued demand from autoimmune indications, with negligible COVID-19 related sales:
| Year |
Estimated Sales (USD Millions) |
Growth Rate |
| 2023 |
370 |
5.7% |
| 2024 |
390 |
5.4% |
| 2025 |
410 |
5.1% |
| 2026 |
420 |
2.4% |
| 2027 |
420 |
0% |
The plateau in later years reflects market saturation, generic competition, and regulatory stability.
Competitive Landscape
Major manufacturers include Novartis, Teva, Sandoz, and local generics producers. Market share is predominantly held by generics, with minimal brand differentiation.
Market Risks and Opportunities
Risks:
- Regulatory jeopardy related to safety concerns.
- Expansion of competitors' alternative therapies.
- Declining prescriptions for COVID-19.
Opportunities:
- Expanding use in autoimmune diseases.
- Potential new indications, pending clinical trials.
- Growing markets in Asia-Pacific.
Conclusion
The hydroxychloroquine sulfate market is mature with slow growth primarily driven by autoimmune therapy demands. Regulatory status remains stable for approved indications, supporting predictable revenues. Significant revenue growth prospects rely on new therapeutic claims, regulatory environment stability, and manufacturing capacity.
Key Takeaways
- Market size was roughly $350 million in 2022.
- Projected to reach around $420 million by 2027, with a CAGR of about 3.6%.
- Demand driven by autoimmune disease treatment; COVID-19 influence has waned.
- No significant patent barriers; market is predominantly generic.
- Future growth depends on new indications and regulatory perceptions.
FAQs
1. What are the primary uses of hydroxychloroquine sulfate?
Autoimmune diseases (lupus, rheumatoid arthritis), and historically, malaria treatment.
2. How has COVID-19 affected hydroxychloroquine sales?
It caused a brief surge in demand but has since declined due to safety concerns and withdrawal of emergency authorizations.
3. What is the patent landscape?
Hydroxychloroquine sulfate is off-patent globally, with broad generic manufacturer presence.
4. Which regions lead the market?
North America and Europe account for approximately 80% of sales; growth is higher in Asia-Pacific.
5. What are the main risks to market stability?
Regulatory restrictions, safety issues, and the development of alternative therapies.
References
[1] MarketWatch. (2023). Hydroxychloroquine sulfate market size, forecasts, and trends.
[2] FDA. (2020). Fact sheet on hydroxychloroquine use during COVID-19.
[3] European Medicines Agency. (2021). Summary of product characteristics for hydroxychloroquine.
[4] GlobalData. (2023). Pharmaceutical market analysis report.