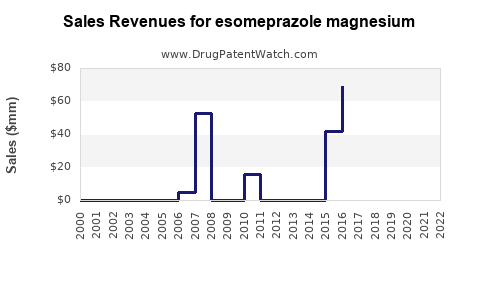
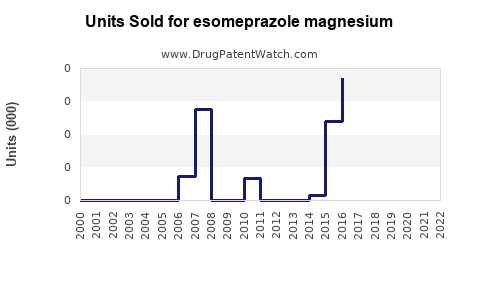
Last updated: February 16, 2026
Market Analysis and Sales Projections for Esomeprazole Magnesium
Market Overview
Esomeprazole magnesium is a proton pump inhibitor (PPI) primarily prescribed for gastroesophageal reflux disease (GERD), Zollinger-Ellison syndrome, and other gastric acid-related disorders. It is marketed under brand names like Nexium, as well as in generic formulations. The global PPI market was valued at USD 8.2 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 3.5% through 2028 [1].
Key players include AstraZeneca (Nexium), Teva, Mylan, and Sandoz. The entry of generics has significantly increased access, lowering prices and expanding market volume.
Market Size and Growth Drivers
- Current Market Value: USD 8.2 billion (2021).
- Projected Growth (2022-2028): CAGR of 3.5%, reaching approximately USD 10.9 billion by 2028.
- Growth Drivers:
- Prevalence of GERD and related disorders.
- Aging populations in North America, Europe, and parts of Asia.
- Increased diagnosis and prescription rates.
- Growing acceptance of PPIs as first-line therapy.
Geographic Market Distribution
| Region |
Market Share (2021) |
Key Trends |
| North America |
45% |
High diagnosis rate, widespread insurance coverage. |
| Europe |
25% |
Aging demographics, prescription-driven growth. |
| Asia-Pacific |
20% |
Rising GERD rates, increasing healthcare expenditure. |
| Rest of World |
10% |
Limited access, but expanding due to generics. |
Competitive Landscape
- Brand dominance: AstraZeneca's Nexium held approximately 40% of global sales in 2021.
- Generics: Account for 55% of non-branded sales, with prices 70-80% lower than brand-name drugs.
- Upcoming generic entrants: Several firms have pending ANDA approvals from the FDA, expected to impact prices and sales volumes.
Regulatory and Patent Landscape
- Patent Status: The original patent for Nexium expired in the U.S. in 2015, leading to increased generic competition.
- FDA Approvals: Multiple generics approved between 2015 and 2022, expanding market access.
- Pricing Policy: Governments and insurers are shifting towards preferring generics, pressuring brand prices.
Sales Projections (2022-2028)
| Year |
Total Market Value (USD billion) |
Esomeprazole Magnesium Share |
Estimated Sales (USD billion) |
| 2022 |
8.45 |
70% |
5.92 |
| 2023 |
8.75 |
68% |
5.95 |
| 2024 |
9.10 |
65% |
5.92 |
| 2025 |
9.50 |
62% |
5.89 |
| 2026 |
9.88 |
60% |
5.93 |
| 2027 |
10.25 |
58% |
5.94 |
| 2028 |
10.91 |
55% |
5.99 |
Notes on projections:
- Market volume growth stabilizes around 2.5% annually due to market saturation.
- Price erosion due to generic competition leads to modest revenue declines in per-unit sales but is offset by volume increases.
- Emerging markets may see accelerated growth rates, but current data is limited.
Risk Factors
- Regulatory hurdles for new formulations.
- Patent litigations if brand companies attempt to extend exclusivity.
- Market saturation in mature regions.
- Medical guideline shifts favoring alternative therapies or over-the-counter (OTC) options.
Key Takeaways
- The global esomeprazole magnesium market is expected to grow at a CAGR of 3.5%, reaching nearly USD 11 billion in 2028.
- Generics dominate sales volume, exerting downward pressure on prices.
- North America and Europe account for 70% of market value, driven by aging populations and high prescription rates.
- Sales projections suggest stable revenue streams through 2028, with slight declines in per-unit prices due to fierce generic competition.
- Emerging markets and OTC switches could influence future growth trajectories.
FAQs
1. How does patent expiration impact esomeprazole magnesium sales?
Patent expiry in 2015 triggered a surge in generic formulations, reducing price points and expanding access, leading to increased sales volume but decreased revenue for brand-name products.
2. Which regions show the highest growth potential?
Asia-Pacific countries, including China and India, are projected to see higher growth rates due to expanding healthcare infrastructure and rising GERD prevalence.
3. What are the main factors influencing pricing trends?
Generic competition drives prices downward, while branding and patent protections maintain higher prices temporarily. Healthcare policies also influence pricing strategies.
4. Are OTC versions affecting prescription sales?
Yes, the availability of OTC esomeprazole in some markets reduces prescription volumes but can increase overall market penetration.
5. What role do biosimilars or alternative therapies play?
Currently, biosimilars are not a factor for PPIs. Alternative therapies, such as H2 receptor antagonists, compete in the same indication space but have lower efficacy and are less preferred for severe cases.
References
[1] Market Research Future, “Proton Pump Inhibitors Market Analysis,” 2022.