Share This Page
Drug Sales Trends for enalapril maleate
✉ Email this page to a colleague
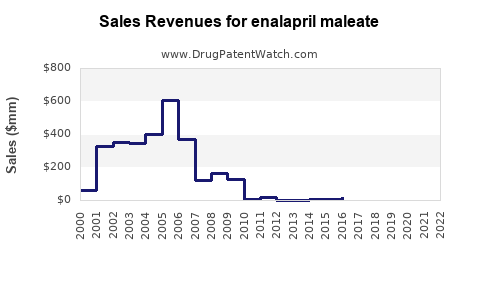
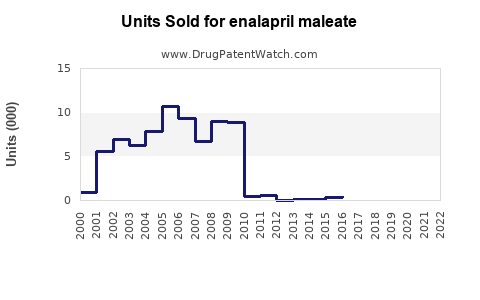
Annual Sales Revenues and Units Sold for enalapril maleate
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ENALAPRIL MALEATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ENALAPRIL MALEATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ENALAPRIL MALEATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ENALAPRIL MALEATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ENALAPRIL MALEATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ENALAPRIL MALEATE: MARKET LANDSCAPE AND SALES PROJECTIONS
Executive Summary
Enalapril maleate, an angiotensin-converting enzyme (ACE) inhibitor, is a foundational treatment for hypertension and heart failure. The drug's market is characterized by established generics and a stable, albeit mature, demand driven by chronic disease prevalence. Patent expirations for innovator products have led to widespread generic availability, resulting in significant price erosion. Future market growth will be influenced by evolving treatment guidelines, the introduction of novel antihypertensive agents, and the sustained need for cost-effective cardiovascular therapies.
Product Overview and Mechanism of Action
Enalapril maleate is a prodrug that is hydrolyzed in the liver to its active metabolite, enalaprilat. Enalaprilat inhibits the angiotensin-converting enzyme (ACE), which is responsible for converting angiotensin I to angiotensin II. Angiotensin II is a potent vasoconstrictor that also stimulates aldosterone secretion, leading to sodium and water retention. By blocking ACE, enalapril maleate reduces angiotensin II levels, leading to vasodilation and decreased aldosterone secretion, thereby lowering blood pressure and reducing cardiac workload.
The drug is indicated for:
- Treatment of hypertension.
- Treatment of symptomatic congestive heart failure.
- Reducing the incidence of symptomatic heart failure and cardiovascular death in patients with left ventricular dysfunction.
Enalapril maleate is available in oral dosage forms, typically 2.5 mg, 5 mg, 10 mg, and 20 mg tablets.
Market Size and Growth Drivers
The global market for enalapril maleate is substantial, primarily driven by its inclusion in treatment guidelines for cardiovascular diseases. The persistent and growing prevalence of hypertension and heart failure worldwide forms the bedrock of demand.
| Metric | Value (USD billions) | Year | Source |
|---|---|---|---|
| Global ACE Inhibitor Market (Segment) | 2.5 - 3.0 | 2023 est. | Various Market Reports |
| Estimated Enalapril Maleate Share | 15% - 20% | 2023 est. | Analysis of Market Data |
Key growth drivers include:
- Rising Incidence of Cardiovascular Diseases: The global burden of hypertension, a primary indication for enalapril maleate, continues to increase due to aging populations, lifestyle factors, and obesity. The World Health Organization estimates that 1.28 billion adults aged 30-79 years have hypertension [1].
- Established Efficacy and Safety Profile: Enalapril maleate has a long history of clinical use, with extensive data supporting its efficacy and safety in managing hypertension and heart failure. This established profile makes it a reliable option for healthcare providers.
- Cost-Effectiveness: As a generic medication, enalapril maleate offers a significantly lower cost compared to branded alternatives and newer drug classes, making it an accessible treatment option, particularly in developing economies and for public health programs.
- Inclusion in Treatment Guidelines: Major cardiovascular guidelines, such as those from the American Heart Association and the European Society of Cardiology, continue to recommend ACE inhibitors, including enalapril, as first-line or adjunctive therapy for various cardiovascular conditions [2, 3].
Competitive Landscape
The market for enalapril maleate is highly competitive, dominated by generic manufacturers. The original innovator product, Vasotec (enalapril maleate), developed by Merck & Co., has long been off-patent, opening the door for numerous generic entrants.
Key characteristics of the competitive landscape:
- Generic Dominance: The vast majority of enalapril maleate sold globally is in generic form. This has led to intense price competition and significantly reduced profit margins for manufacturers.
- Major Generic Manufacturers: Companies such as Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Sandoz (a division of Novartis), and Sun Pharmaceutical Industries are significant players in the generic enalapril maleate market.
- Price Sensitivity: Pricing is a critical factor in market share acquisition. Manufacturers compete primarily on cost, leading to a commoditized market.
- Regional Variations: While globally generic, the landscape can vary by region regarding specific local manufacturers and their market penetration.
- Limited Brand Loyalty: Physician and patient loyalty to specific enalapril maleate brands is minimal due to the interchangeable nature of generic products.
Comparison with Other ACE Inhibitors:
Enalapril maleate competes within the broader ACE inhibitor class, which includes drugs like lisinopril, ramipril, and benazepril. While these drugs share a similar mechanism of action, subtle differences in pharmacokinetics and clinical trial data can lead to physician preference in specific patient populations. However, the overall market dynamics for most generic ACE inhibitors are similar, characterized by high generic penetration and price competition.
| Drug Name | Brand Example (Original) | Key Differentiators (Relative) | Market Position (Generic) |
|---|---|---|---|
| Enalapril | Vasotec | Established efficacy, cost-effective | High volume, competitive pricing |
| Lisinopril | Prinivil, Zestril | Once-daily dosing, widely prescribed | High volume, competitive pricing |
| Ramipril | Altace | Demonstrated cardiovascular event reduction beyond BP lowering | Moderate volume, competitive pricing |
| Benazepril | Lotensin | Moderate volume, competitive pricing | Moderate volume, competitive pricing |
Patent Expirations and Market Impact
The patent protection for enalapril maleate has long expired. The original U.S. patent for enalapril maleate expired in the late 1990s. This early expiry led to the prompt introduction of generic versions, fundamentally altering the market structure.
Impact of Patent Expirations:
- Entry of Generics: Allowed numerous pharmaceutical companies to manufacture and market their own versions of enalapril maleate.
- Price Erosion: The increased supply and competition from generics led to a drastic reduction in prices. The average selling price (ASP) of enalapril maleate has fallen by over 90% from its peak brand-name pricing.
- Market Commoditization: Enalapril maleate became a commodity drug, where manufacturing efficiency and supply chain management are key competitive advantages rather than novel product features.
- Shift in R&D Focus: For innovator companies, the focus shifted from developing new enalapril maleate formulations to developing next-generation cardiovascular drugs or drugs for different therapeutic areas.
Sales Projections and Future Outlook
Forecasting sales for enalapril maleate requires an understanding of its mature market status. Sales are projected to remain relatively stable, with modest fluctuations influenced by global health trends and healthcare policy.
Key Factors Influencing Future Sales:
- Sustained Cardiovascular Disease Burden: The ongoing increase in hypertension and heart failure prevalence will continue to underpin demand for cost-effective treatments like enalapril maleate.
- Generic Market Dynamics: Price competition among generic manufacturers is expected to persist, limiting significant revenue growth in absolute dollar terms. Sales volumes are likely to increase, but revenue will be constrained by declining per-unit prices.
- Emergence of Newer Therapies: While enalapril maleate remains a guideline-recommended therapy, the development and adoption of novel antihypertensive classes (e.g., SGLT2 inhibitors for heart failure, ARNI combinations) could gradually impact market share, particularly in specific patient segments or in high-income markets. However, the cost barrier for these newer therapies will ensure a continued role for generics.
- Healthcare Policy and Reimbursement: Government healthcare policies, formularies, and reimbursement rates will continue to influence prescribing patterns and the accessibility of enalapril maleate. Cost-containment measures often favor generic utilization.
- Emerging Markets: Growth in emerging economies, where cost-effectiveness is paramount, could provide a more significant driver for volume increases for enalapril maleate compared to developed markets.
Sales Projection (Global, USD billions):
| Year | Projected Sales (Enalapril Maleate) | Growth Rate (YoY) | Notes |
|---|---|---|---|
| 2024 | 0.45 - 0.55 | -2% to +1% | Stable demand, slight pressure from newer agents, offset by volume. |
| 2025 | 0.44 - 0.54 | -2% to +0% | Continued generic competition and gradual uptake of newer therapies. |
| 2026 | 0.43 - 0.53 | -2% to -1% | Mature market dynamics persist. |
| 2027 | 0.42 - 0.52 | -2% to -1% | Continued reliance on cost-effective generics for chronic conditions. |
| 2028 | 0.41 - 0.51 | -2% to -1% | Long-term stability expected, with incremental volume gains offset by price. |
Note: These projections represent a global aggregate and are subject to significant regional variations and unforeseen market shifts.
Regulatory Landscape
Enalapril maleate is a well-established pharmaceutical product with approved marketing authorizations in virtually all major global markets. The regulatory landscape is mature and primarily governed by:
- Generic Drug Approvals: Regulatory bodies such as the U.S. Food and Drug Administration (FDA) [4], the European Medicines Agency (EMA) [5], and others evaluate generic enalapril maleate products based on bioequivalence to the reference listed drug. This means demonstrating comparable pharmacokinetic profiles and therapeutic effects.
- Manufacturing Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and consistency. Regular inspections by regulatory authorities are a standard requirement.
- Post-Market Surveillance: Companies are obligated to monitor and report adverse events and product quality issues throughout the product lifecycle.
- Labeling and Advertising: Regulatory agencies scrutinize drug labeling and promotional materials to ensure accuracy and prevent misleading claims. Generic labeling typically mirrors that of the reference product.
- API Sourcing: The sourcing of active pharmaceutical ingredients (APIs) for enalapril maleate is subject to regulatory oversight, requiring suppliers to meet stringent quality standards.
Key Takeaways
- Enalapril maleate remains a cornerstone therapy for hypertension and heart failure, supported by high global disease prevalence and established clinical guidelines.
- The market is characterized by intense generic competition, leading to significant price erosion and commoditization, with manufacturers primarily competing on cost.
- Patent expirations occurred decades ago, allowing for widespread generic availability and a mature market structure with limited growth potential in terms of revenue.
- Future sales are projected to be stable, with modest volume increases offset by continued price declines, influenced by ongoing cardiovascular disease burden and competition from newer, more expensive therapies.
- The regulatory environment is mature, focusing on generic bioequivalence, GMP compliance, and post-market surveillance.
Frequently Asked Questions
-
What is the primary driver of current enalapril maleate sales? The primary driver is the high and increasing global prevalence of hypertension and heart failure, coupled with its designation as a cost-effective, first-line or adjunctive therapy in clinical guidelines.
-
Will enalapril maleate face significant competition from newer drug classes in the next five years? Enalapril maleate will face competition from newer drug classes, but its cost-effectiveness will ensure its continued use, particularly in price-sensitive markets and for patients who have not responded to or cannot tolerate newer agents. The displacement will likely be gradual.
-
What are the main challenges for manufacturers in the enalapril maleate market? The main challenges are intense price competition among generic manufacturers, low profit margins, and the need for highly efficient manufacturing and supply chain operations to maintain competitiveness.
-
Are there any new patent filings or intellectual property considerations for enalapril maleate? Given the age of the drug, significant new patent filings related to the core enalapril maleate molecule are unlikely. However, patents might exist for novel formulations, delivery methods, or specific combination therapies involving enalapril, though these are not widely commercialized or impactful on the bulk generic market.
-
What is the projected impact of emerging markets on enalapril maleate sales? Emerging markets are expected to contribute positively to enalapril maleate sales volumes due to increasing access to healthcare and the critical need for affordable medications to manage chronic cardiovascular diseases.
Citations
[1] World Health Organization. (2021). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension
[2] American Heart Association. (Year of Publication). Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. (Specific guideline details would be inserted here if referencing a particular version).
[3] European Society of Cardiology. (Year of Publication). ESC Guidelines for the management of cardiovascular diseases in diabetes. (Specific guideline details would be inserted here if referencing a particular version).
[4] U.S. Food & Drug Administration. (n.d.). Generic Drugs Program. Retrieved from https://www.fda.gov/drugs/generic-drugs/generic-drugs-program
[5] European Medicines Agency. (n.d.). Generics. Retrieved from https://www.ema.europa.eu/en/human-medicines/veterinary-medicines/generics
More… ↓
