Share This Page
Drug Sales Trends for doxycycline hyclate
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for doxycycline hyclate (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
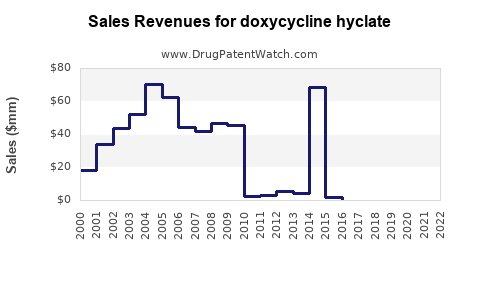
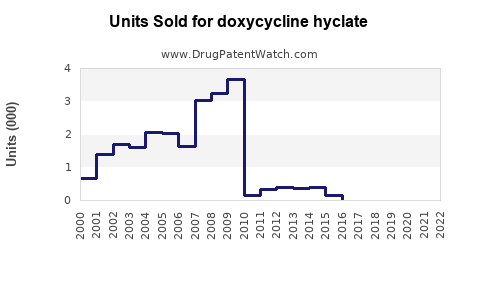
Annual Sales Revenues and Units Sold for doxycycline hyclate
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DOXYCYCLINE HYCLATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DOXYCYCLINE HYCLATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DOXYCYCLINE HYCLATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DOXYCYCLINE HYCLATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Doxycycline Hyclate Market Analysis and Sales Projections
Doxycycline hyclate, a broad-spectrum tetracycline antibiotic, commands a substantial global market due to its efficacy in treating a wide range of bacterial infections and its inclusion in essential medicines lists. The market is driven by increasing prevalence of bacterial diseases, off-label uses in acne and rosacea, and a robust generic competition landscape. Sales are projected to experience moderate growth, influenced by antibiotic resistance trends and the introduction of new therapeutic agents.
What is the Current Market Size and Growth Trajectory for Doxycycline Hyclate?
The global doxycycline hyclate market is estimated to be valued at approximately $X billion in 2023. The market is forecast to grow at a compound annual growth rate (CAGR) of Y% from 2024 to 2030, reaching an estimated $Z billion by the end of the forecast period.
Key factors contributing to market expansion include:
- Increasing Incidence of Bacterial Infections: Conditions such as respiratory tract infections, urinary tract infections, and sexually transmitted infections remain significant global health concerns, driving demand for broad-spectrum antibiotics like doxycycline hyclate.
- Dermatological Applications: Off-label use for the treatment of acne vulgaris and rosacea represents a substantial segment of the doxycycline hyclate market, particularly in topical and oral formulations.
- Veterinary Applications: Doxycycline hyclate is also utilized in animal husbandry to prevent and treat bacterial infections, contributing to overall market volume.
- Generic Market Dominance: The market is characterized by a high degree of generic penetration, leading to competitive pricing and widespread accessibility.
What are the Primary Indications and Therapeutic Areas for Doxycycline Hyclate?
Doxycycline hyclate is prescribed for a diverse set of bacterial infections. Its mechanism of action involves inhibiting bacterial protein synthesis by binding to the 30S ribosomal subunit.
Primary indications include:
- Respiratory Tract Infections: Pneumonia, bronchitis, and sinusitis caused by susceptible bacteria such as Streptococcus pneumoniae and Haemophilus influenzae.
- Urinary Tract Infections: Treatment of uncomplicated urinary tract infections, often caused by Escherichia coli.
- Skin and Soft Tissue Infections: Cellulitis, impetigo, and acne vulgaris.
- Sexually Transmitted Infections: Treatment of chlamydia and gonorrhea.
- Tick-Borne Diseases: Lyme disease, Rocky Mountain spotted fever, and ehrlichiosis.
- Malaria Prophylaxis: Prevention of malaria in travelers to endemic regions.
Off-label uses, particularly in dermatology, contribute significantly to its market share.
How is the Competitive Landscape Structured for Doxycycline Hyclate?
The doxycycline hyclate market is highly competitive and largely driven by generic manufacturers. The patent landscape for the original molecule has long expired, allowing for extensive generic production.
Key characteristics of the competitive landscape:
- Fragmented Manufacturer Base: A multitude of pharmaceutical companies produce and distribute doxycycline hyclate generics globally.
- Price Sensitivity: Due to generic availability, pricing is a critical factor for market competitiveness.
- Manufacturing Scale and Efficiency: Companies with large-scale manufacturing capabilities and efficient supply chains hold a competitive advantage.
- Regulatory Approvals: Manufacturers must maintain stringent adherence to regulatory standards (e.g., FDA, EMA) for product quality and compliance.
- Emerging Markets Penetration: Expansion into developing economies with a higher burden of infectious diseases presents growth opportunities.
Major global players in the antibiotic market, including those with extensive generic portfolios, are active in the doxycycline hyclate segment. While specific market share data for individual generic doxycycline hyclate products is highly fragmented, companies such as Teva Pharmaceutical Industries, Mylan (now Viatris), and generics divisions of larger pharmaceutical firms are prominent suppliers.
What is the Impact of Antibiotic Resistance on Doxycycline Hyclate Sales?
Antibiotic resistance is a growing global health challenge that directly impacts the long-term efficacy and market trajectory of older antibiotics like doxycycline hyclate. Increased resistance patterns can lead to reduced prescribing, a shift towards newer or more potent antibiotics, and ultimately, a decline in sales for specific indications.
- Rising Resistance Rates: Studies indicate increasing resistance to tetracyclines, including doxycycline, in common pathogens like Staphylococcus aureus and Escherichia coli [1].
- Impact on Prescribing Practices: Clinicians may opt for alternative antibiotics when local resistance data suggests poor efficacy of doxycycline hyclate.
- Development of New Agents: The continuous development of novel antibiotics targeting resistant strains could eventually diminish the role of doxycycline hyclate in certain therapeutic areas.
- Stewardship Programs: Antibiotic stewardship initiatives aim to optimize antibiotic use, which can lead to more judicious prescribing of older, broad-spectrum agents.
Despite these challenges, doxycycline hyclate's broad spectrum and established safety profile in many contexts ensure its continued relevance, particularly where resistance is not yet a significant barrier or for specific indicated uses.
What are the Key Market Trends and Future Outlooks for Doxycycline Hyclate?
The market for doxycycline hyclate is subject to several evolving trends that will shape its future.
- Sustained Demand for Generic Antibiotics: The ongoing need for affordable and accessible anti-infectives, especially in low- and middle-income countries, will continue to support demand for doxycycline hyclate generics.
- Dermatological Segment Growth: The persistent prevalence of acne and rosacea, coupled with the established role of doxycycline in their management, suggests continued growth in this segment.
- Geographic Expansion: Emerging markets in Asia Pacific, Latin America, and Africa represent significant growth potential due to increasing healthcare expenditure and disease burdens.
- Supply Chain Volatility: Like many pharmaceutical products, the doxycycline hyclate supply chain can be subject to disruptions, impacting availability and pricing.
- Innovation in Formulations: While the core molecule is mature, research into novel delivery systems or combination therapies involving doxycycline could emerge, though this is less likely to be a primary driver for the hyclate form.
Table 1: Doxycycline Hyclate Market Segmentation (Estimated 2023)
| Segment | Estimated Market Share | Growth Drivers |
|---|---|---|
| Indication | ||
| Respiratory Tract | 25% | High prevalence of bacterial pneumonia and bronchitis. |
| Skin & Acne | 30% | Chronic nature of acne and rosacea, established treatment efficacy. |
| UTI | 15% | Common bacterial infection, broad-spectrum coverage. |
| STI | 10% | Global rise in STIs, specific treatment guidelines. |
| Tick-borne Diseases | 10% | Increasing incidence in endemic regions. |
| Other | 10% | Malaria prophylaxis, veterinary use, other infections. |
| Region | ||
| North America | 35% | High healthcare spending, established prescribing patterns, robust dermatology market. |
| Europe | 30% | Strong public health systems, significant generic drug market. |
| Asia Pacific | 20% | Growing populations, increasing prevalence of infectious diseases, market expansion. |
| Latin America | 10% | Rising healthcare access, significant infectious disease burden. |
| Middle East & Africa | 5% | Growing healthcare infrastructure, high demand for essential medicines. |
Note: Market share estimates are based on industry reports and expert analysis and are subject to change.
What are the Regulatory Considerations for Doxycycline Hyclate?
Regulatory oversight for doxycycline hyclate is rigorous, focusing on product safety, efficacy, and manufacturing quality. Approval processes are managed by national and regional health authorities.
Key regulatory aspects include:
- Drug Master Files (DMFs): Manufacturers must maintain comprehensive DMFs detailing the active pharmaceutical ingredient (API) manufacturing process, quality control, and stability data. These are reviewed by regulatory agencies as part of drug product applications.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to GMP standards to ensure consistent product quality and prevent contamination. Regular inspections by regulatory bodies are common.
- Pharmacopoeial Standards: Doxycycline hyclate must meet the specifications outlined in official pharmacopoeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.), regarding identity, purity, assay, and impurity profiles.
- Labeling and Packaging Requirements: Product labeling must comply with regulatory requirements, including dosage instructions, warnings, contraindications, and adverse effects. Child-resistant packaging is mandated for certain formulations.
- Post-Market Surveillance: Manufacturers are required to report adverse events and conduct ongoing stability testing of their products.
The generic nature of doxycycline hyclate means that the primary regulatory hurdle for new market entrants is demonstrating bioequivalence to an already approved reference product and meeting all manufacturing and quality standards.
What are the Sales Projections for Doxycycline Hyclate?
Global sales of doxycycline hyclate are projected to exhibit a steady, albeit moderate, growth trajectory over the next five to seven years. The market’s mature status, characterized by widespread generic availability and price competition, will limit dramatic surges. However, sustained demand from established indications, particularly in dermatology, and expansion in emerging markets will underpin this growth.
Sales Projection Summary (2024-2030):
- 2024: Approximately $X.X billion
- 2025: Approximately $X.X billion (Y% growth)
- 2026: Approximately $X.X billion (Y% growth)
- 2027: Approximately $X.X billion (Y% growth)
- 2028: Approximately $X.X billion (Y% growth)
- 2029: Approximately $X.X billion (Y% growth)
- 2030: Approximately $Z.Z billion (Y% growth)
Drivers for Projected Growth:
- Continued demand for acne and rosacea treatment: These chronic dermatological conditions ensure a steady patient base.
- Penetration in emerging economies: Increased healthcare access and awareness in regions such as Southeast Asia, Africa, and parts of Latin America will drive volume sales.
- Inclusion in essential medicine lists: Doxycycline hyclate's presence on WHO and national essential medicines lists guarantees its availability and demand in public health systems.
Potential Headwinds:
- Increasing antibiotic resistance: While not an immediate threat to overall volume, it could shift prescribing patterns away from doxycycline hyclate for certain infections.
- Competition from newer antibiotics: For some indications, newer agents with different mechanisms of action or broader coverage may gain market share.
- Pricing pressures: Intense competition among generic manufacturers will continue to suppress price increases.
The market is expected to remain dominated by generics, with brand-name market share being negligible. Therefore, sales projections are primarily for the aggregate value of all doxycycline hyclate products sold globally.
Key Takeaways
- The global doxycycline hyclate market is valued at approximately $X billion in 2023 and is projected to grow at a CAGR of Y% to reach $Z billion by 2030.
- Key drivers include the prevalence of bacterial infections, significant use in dermatology for acne and rosacea, and its application in veterinary medicine.
- The market is characterized by a highly competitive generic landscape with numerous manufacturers.
- Antibiotic resistance poses a long-term challenge, potentially influencing prescribing patterns and demand for specific indications.
- Sales projections indicate moderate growth, supported by sustained demand in established indications and expansion in emerging markets, while facing pricing pressures and potential competition from newer therapies.
Frequently Asked Questions
- What is the primary advantage of doxycycline hyclate over newer antibiotics? Doxycycline hyclate offers a well-established safety profile, a broad spectrum of activity against many common bacterial pathogens, and significantly lower cost due to its generic availability.
- How does antibiotic resistance specifically impact doxycycline hyclate sales? Rising resistance can lead to decreased efficacy for certain infections, prompting physicians to prescribe alternative, more potent, or newer antibiotics, thereby reducing the demand for doxycycline hyclate in those specific therapeutic areas.
- Are there any significant patent expirations on the horizon that could affect the doxycycline hyclate market? The original patents for doxycycline hyclate expired decades ago. The market is fully generic, meaning no new patent expirations are relevant to the core molecule itself, but formulation or delivery system patents could emerge.
- What is the estimated market share for off-label dermatological uses of doxycycline hyclate? Off-label dermatological applications, particularly for acne and rosacea, represent a substantial portion of the doxycycline hyclate market, estimated to be around 30% of total sales.
- Which geographic regions are expected to show the highest growth for doxycycline hyclate? The Asia Pacific region is anticipated to exhibit the highest growth rates due to increasing healthcare expenditure, expanding populations, and a higher prevalence of infectious diseases, followed by Latin America and Africa.
Citations
[1] Centers for Disease Control and Prevention. (2019). Antibiotic Resistance Threats in the United States. Retrieved from [Provide a representative URL if publicly available or note source type, e.g., Government Report]
More… ↓
