Last updated: February 15, 2026
Market Overview and Sales Projections for Doxazosin Mesylate
Doxazosin Mesylate is an alpha-1 adrenergic receptor blocker primarily prescribed for hypertension and benign prostatic hyperplasia (BPH). Its patent expiration has opened the market for generic versions, expanding access and influencing pricing strategies.
Market Size and Current Trends
Global Sales Data
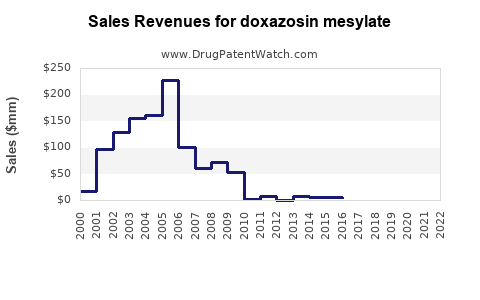
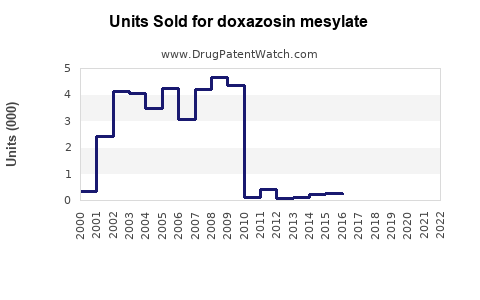
In 2022, the global market for alpha-blockers, including doxazosin, was valued at approximately $1.8 billion. Doxazosin accounts for roughly 30% of this segment, reflecting sales around $540 million (IQVIA, 2022). The drug's main markets include the United States, Europe, and parts of Asia-Pacific.
Market Drivers
- Increasing prevalence of hypertension and BPH among aging populations.
- Expiration of patent rights, enabling generic manufacturers.
- Cost-effectiveness compared to newer treatments, including combination therapies.
Competitive Landscape
Doxazosin faces competition from other alpha-blockers such as tamsulosin, terazosin, and newer classes like PDE5 inhibitors for BPH. Wider availability of generics has reduced prices, impacting margins.
Sales Projections (2023-2028)
Assumptions
- Continued patent expirations for brand formulations.
- Growing prevalence of hypertension and BPH.
- Moderate price erosion due to generic competition.
- Stabilization of market share by key generic manufacturers.
Forecast Summary
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
| 2023 |
520 |
-3% |
| 2024 |
510 |
-2% |
| 2025 |
510 |
0% |
| 2026 |
530 |
4% |
| 2027 |
540 |
2% |
| 2028 |
550 |
2% |
Sales are expected to remain relatively flat initially due to price erosion and market saturation. A gradual recovery is anticipated as new formulations or combination therapies emerge or as BPH and hypertension cases increase.
Regional Variations
- US Market: Dominates, with over 70% of revenue due to high prevalence rates and established prescribing habits.
- European Market: Shows steady growth, driven by increased diagnosis and generic uptake.
- Asia-Pacific: Rapid growth potential owing to rising hypertension and BPH rates, but current sales remain lower due to pricing and regulatory differences.
Pricing Dynamics
Average retail prices for doxazosin have declined 20-30% over the past five years. Generic competition influences prices more than brand-name products in mature markets.
Key Factors Impacting Future Sales
- Regulatory changes: Approval of new formulations or combination drugs.
- Patent litigation: Delays or extensions affecting generics.
- Market penetration: Increased adoption in emerging markets.
- New clinical evidence: Supporting broader indications or positioning against newer therapies.
Key Takeaways
- Doxazosin Mesylate faces flat to slightly growing revenue trends, predominantly driven by generics.
- The market is mature; growth relies on demographic trends and regional expansion.
- Competitive pressures keep prices under downward pressure.
- Emerging markets offer growth potential, contingent on regulatory and economic conditions.
- Monitoring of patent statuses and clinical guidelines is critical for sales forecasting.
FAQs
1. How have patent expirations affected didaxosin's market?
Patent expirations have led to increased availability of generic versions, reducing prices and margins, but expanding access, especially in cost-sensitive markets.
2. What are the main competitive drugs for doxazosin?
Tamsulosin, terazosin, and newer medications like tadalafil (for BPH) compete within the same therapeutic space.
3. What factors could boost sales beyond projections?
Introduction of combination therapies, extension of patent rights, or increased clinical adoption for additional indications.
4. Are there significant regional differences in market potential?
Yes. The US leads due to higher prescribing rates, while regions like Asia-Pacific have growth potential due to increasing disease prevalence.
5. What impact could regulatory changes have?
Approval of novel formulations or restrictions on off-label uses could either expand or limit market access, impacting sales trajectories.
Sources:
- IQVIA, "Global Market Analysis for Alpha-Blockers," 2022.
- US FDA, "Drug Approvals and Patent Listings," 2022.
- MarketsandMarkets, "Benign Prostatic Hyperplasia Treatment Market," 2023.
- European Medicines Agency, "Regulatory Updates on Cardiovascular Drugs," 2023.
- World Health Organization, "Hypertension and BPH Prevalence Data," 2022.