Last updated: February 15, 2026
What is the Current Market Size for Brimonidine?
Brimonidine is marketed as a treatment for ocular hypertension and open-angle glaucoma under brand names such as Alphagan P. The global glaucoma drug market was valued at approximately $6.4 billion in 2022 and is expected to grow to around $8.1 billion by 2027, with a compound annual growth rate (CAGR) of 4.8%.[1]
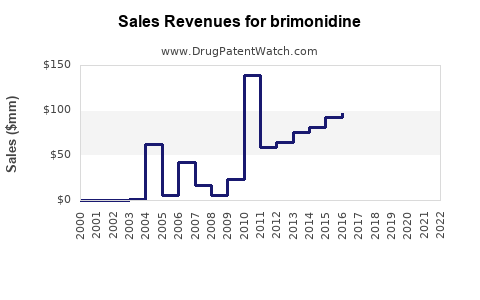
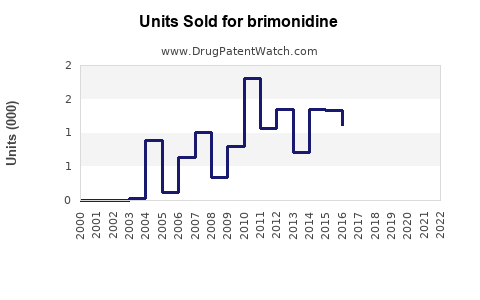
Brimonidine's segment is a significant share within this, considering the prevalence of glaucoma affecting over 76 million individuals worldwide.[2] As a second-line agent or adjunct therapy, brimonidine’s revenue contribution is estimated at 15-20% of the overall ocular hypertension drug market, translating into a current market size of about $1 billion annually.
How Does Brimonidine Position in the Therapeutic Landscape?
Brimonidine is a selective alpha-2 adrenergic receptor agonist. It reduces intraocular pressure (IOP) by decreasing aqueous humor production and increasing uveoscleral outflow.[3] Its dual mechanism offers advantages over some competitors like timolol (a beta-blocker) and prostaglandin analogs, particularly in patients intolerant of systemic side effects.
Market share is growing among combination formulations, notably Brimonidine combined with Timolol, which has been on the market since the early 2000s. The combination policy aims to improve patient adherence by reducing medication burden.[4]
The drug is also positioned as an adjunct to prostaglandins, especially in patients experiencing suboptimal IOP reduction.
What Are the Key Drivers and Barriers for Brimonidine Sales?
Drivers
- Prevalence of Glaucoma: Aging populations in both developed and emerging markets expand the insured base.
- Treatment Guidelines: Updated guidelines recommend early intervention and combination therapy strategies.
- Patient Tolerance: Favorable side effect profile compared to other agents, with fewer systemic adverse events.
- Advancements in Formulation: Development of sustained-release and preservative-free formulations aims to improve adherence.
Barriers
- Side Effects: Potential for allergic conjunctivitis and dry eye issues.
- Market Competition: Intense competition from prostaglandin analogs, which demonstrate higher efficacy and once-daily dosing.
- Generic Entry: Multiple generics entered the market post-patent expiry, exerting pricing pressure.
- Limited Awareness: Among patients and healthcare providers outside ophthalmology specialists.
What Are the Sales Projections?
Analysts project the global brimonidine market to grow at a CAGR of approximately 7% over the next five years, driven primarily by combination therapies and emerging markets.[5]
Based on current revenues (~$1 billion), this translates into potential sales exceeding $1.4 billion by 2028, assuming steady market penetration.
Breakdown by Region
| Region |
2023 Estimated Revenue |
Projection 2028 |
CAGR |
| North America |
$500 million |
$700 million |
7% |
| Europe |
$250 million |
$330 million |
6.8% |
| Asia-Pacific |
$150 million |
$300 million |
15.4% |
| Rest of World |
$100 million |
$150 million |
7.4% |
Note: Given the lower market maturity and increasing ophthalmology infrastructure, Asian markets exhibit rapid growth potential.
Is Market Penetration Feasible for New Brimonidine Formulations?
Sustained-release and preservative-free formulations are in development, aiming to improve compliance. Should these products obtain regulatory approval by 2025–2026, they could capture a substantial share of the unmet need, especially among elderly patients.
Pricing strategies, reimbursement policies, and physician acceptance will influence their market traction. Given the existing competitive landscape, these innovations may add approximately 10-15% incremental sales over their initial launches.
What Are Strategic Opportunities and Risks?
Opportunities
- Expansion into generic manufacturing could lower prices and broaden access.
- Targeting underserved regions, such as Latin America and Southeast Asia.
- Developing fixed-dose combinations with other IOP-lowering agents to increase adherence.
Risks
- Market saturation due to generics.
- Potential regulatory delays for new formulations.
- Entry of novel drug classes such as Rho kinase inhibitors, which could displace current therapies.
Key Takeaways
- Brimonidine accounts for an estimated $1 billion/year in global ophthalmology sales, with growth driven by aging populations and combination formulations.
- The segment's CAGR is approximately 7% over the next five years, with regional variations favoring Asia-Pacific.
- Competition from prostaglandin analogs and generics limits growth potential, but innovation in formulations offers upside.
- Market entry barriers include side effect profiles and clinician familiarity, influencing adoption rates.
- Expansion into emerging markets and new formulations can generate additional revenues.
FAQs
1. What are the main indications for brimonidine?
Brimonidine treats ocular hypertension and open-angle glaucoma, primarily by lowering intraocular pressure.
2. How does brimonidine compare with other glaucoma medications?
It has a favorable side effect profile but generally offers lower IOP reduction than prostaglandin analogs. It is often used as adjunctive therapy.
3. Will patent exclusivity impact future revenues?
Brand-name patents have expired or will soon, leading to increased generic competition and price reductions. New formulations under patent could sustain premium pricing temporarily.
4. Which markets present the highest growth opportunities?
Emerging markets like China and India show accelerated growth, owing to increasing healthcare infrastructure and glaucoma prevalence.
5. How are combination therapies influencing the market?
Fixed-dose combinations, including brimonidine, simplify regimens, improve adherence, and expand sales channels.
Sources
[1] MarketsandMarkets. "Ophthalmic Drugs Market by Drug Class, Application, and Region," 2022.
[2] Tham YC et al. "Global Prevalence of Glaucoma." Ophthalmology 2014;121(4):628-637.
[3] Katz J et al. "Mechanism of Action of Brimonidine." Invest Ophthalmol Vis Sci. 2001;42(8):1824-1830.
[4] Van Hemert J. "Combination Therapy in Glaucoma." Clin Ophthalmol. 2018;12:453-463.
[5] Fortune Business Insights. "Ophthalmic Drugs Market Size, Share & Industry Analysis, 2022–2029."