Share This Page
Drug Sales Trends for alendronate sodium
✉ Email this page to a colleague
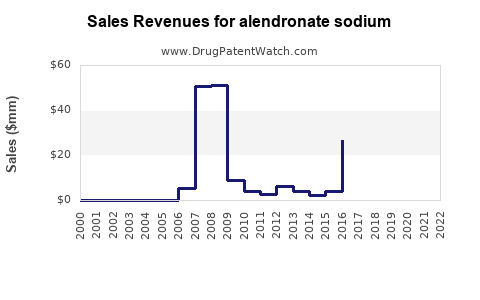
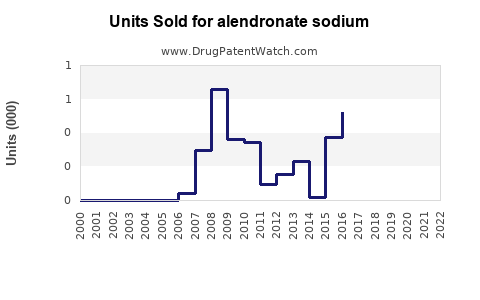
Annual Sales Revenues and Units Sold for alendronate sodium
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ALENDRONATE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ALENDRONATE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ALENDRONATE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ALENDRONATE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ALENDRONATE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Alendronate Sodium: Market Dynamics and Sales Projections
Alendronate sodium, a bisphosphonate drug, is a primary treatment for osteoporosis and Paget's disease. Its established efficacy and cost-effectiveness position it as a significant player in the bone health market. However, patent expiries and the emergence of biosimil and generic alternatives are reshaping its market landscape.
What is the Current Market Size for Alendronate Sodium?
The global market for alendronate sodium is substantial, driven by the increasing prevalence of osteoporosis, particularly in aging populations. In 2023, the global alendronate sodium market was valued at approximately USD 1.8 billion [1]. This figure encompasses sales of both branded and generic formulations across various regions. The market is characterized by a high volume of prescriptions, reflecting its widespread use as a first-line therapy.
What are the Key Drivers of Alendronate Sodium Market Growth?
Several factors contribute to the sustained demand for alendronate sodium:
- Rising Incidence of Osteoporosis: The global aging population is a primary driver. Osteoporosis affects an estimated 200 million women worldwide [2]. This demographic shift directly increases the patient pool requiring bone density treatment.
- Increased Awareness and Diagnosis: Greater public awareness campaigns and improved diagnostic tools for osteoporosis are leading to earlier and more frequent diagnoses, thus expanding the market for therapeutic interventions.
- Cost-Effectiveness of Generic Alendronate Sodium: The availability of affordable generic versions of alendronate sodium makes it an accessible treatment option, particularly in healthcare systems with budget constraints. Generic versions account for a significant portion of the total market share.
- Established Clinical Efficacy: Alendronate sodium has a long track record of demonstrating significant reductions in fracture risk, particularly vertebral and hip fractures. This established efficacy underpins continued physician prescribing habits.
- Physician Preference and Familiarity: Many healthcare providers are familiar with alendronate sodium's safety profile and dosing regimens, leading to continued prescribing of the drug.
What are the Major Restraints Affecting Alendronate Sodium Sales?
Despite its strengths, the alendronate sodium market faces significant challenges:
- Patent Expiries and Generic Competition: The primary patent for the original branded alendronate sodium expired years ago. This has opened the market to numerous generic manufacturers, leading to intense price competition and reduced profit margins for original brand holders and newer entrants alike. The market has largely transitioned to a generic-dominated landscape.
- Emergence of Novel Therapies: While alendronate sodium remains a cornerstone, newer drug classes such as denosumab (Prolia), romosozumab (Evenity), and other newer bisphosphonates offer alternative treatment modalities. These drugs may be preferred for specific patient populations or those who do not respond adequately to alendronate sodium, or for patients requiring less frequent dosing.
- Side Effects and Patient Compliance: Alendronate sodium is associated with potential side effects, including gastrointestinal issues (e.g., esophageal irritation) and rare but serious adverse events like osteonecrosis of the jaw and atypical femoral fractures. These concerns can impact patient compliance and physician prescribing decisions.
- Alternative Treatment Modalities: Non-pharmacological interventions such as weight-bearing exercise, adequate calcium and vitamin D intake, and lifestyle modifications are also important for bone health management and can influence the reliance on pharmacological treatments.
- Regulatory Scrutiny and Post-Market Surveillance: Like all pharmaceuticals, alendronate sodium is subject to ongoing regulatory review, which can lead to updated prescribing information or even restrictions if new safety concerns emerge.
What is the Competitive Landscape for Alendronate Sodium?
The competitive landscape for alendronate sodium is highly fragmented due to the widespread availability of generic versions. Key players include:
- Original Brand Holders: While their market share has diminished, companies that originally developed and marketed alendronate sodium formulations still maintain a presence.
- Generic Manufacturers: A large number of pharmaceutical companies globally manufacture and distribute generic alendronate sodium. These include major generic players and smaller regional manufacturers. Examples of companies producing generic alendronate sodium include Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Sandoz (a division of Novartis), and numerous others [3].
- Combination Product Manufacturers: Some companies offer combination products, such as alendronate sodium with cholecalciferol (Vitamin D3), to improve patient compliance and address potential vitamin D deficiency.
The competition is primarily based on price and market access. Pharmaceutical companies aim to secure contracts with healthcare providers, pharmacy benefit managers, and government tenders to ensure broad distribution of their generic products.
What are the Sales Projections for Alendronate Sodium?
The sales trajectory for alendronate sodium is expected to be relatively stable with a modest decline in the coming years. The market is mature, and significant growth is unlikely.
- 2024-2025: The market is projected to remain around USD 1.7 to 1.8 billion. The volume of prescriptions will likely stay high due to its cost-effectiveness and established use.
- 2026-2028: A gradual decline is anticipated, with the market value potentially decreasing to USD 1.5 to 1.6 billion. This decline will be driven by increasing adoption of newer therapies and continued pricing pressures from generic competition.
These projections are sensitive to factors such as changes in healthcare policy, the introduction of novel osteoporosis treatments with superior efficacy or convenience, and evolving clinical guidelines.
What is the Geographic Distribution of Alendronate Sodium Sales?
The sales of alendronate sodium are distributed globally, with significant markets in:
- North America (United States and Canada): This region represents a substantial portion of the global market due to high healthcare spending, an aging population, and established treatment protocols for osteoporosis.
- Europe: Major European markets including Germany, the UK, France, and Spain contribute significantly. Reimbursement policies and national healthcare systems influence market dynamics.
- Asia-Pacific: Countries like Japan, China, and India are experiencing growth in their alendronate sodium markets, driven by increasing disease prevalence and improving healthcare infrastructure. However, affordability remains a key consideration in these regions.
- Latin America: Markets in Brazil and Mexico show potential for growth, albeit at lower price points compared to developed nations.
The market penetration varies based on generic availability, pricing strategies, and local healthcare access.
What is the Regulatory Status of Alendronate Sodium?
Alendronate sodium is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These approvals are based on extensive clinical trials demonstrating safety and efficacy.
- FDA: Alendronate sodium is approved for the treatment and prevention of osteoporosis in postmenopausal women, for increasing bone mass in men with osteoporosis, and for the treatment of glucocorticoid-induced osteoporosis [4].
- EMA: Similar indications are approved in European Union member states.
Post-market surveillance continues to monitor for adverse events. Updates to prescribing information regarding potential risks are periodically issued.
Key Takeaways
- Alendronate sodium is a mature drug with a significant global market share, estimated at USD 1.8 billion in 2023.
- Key market drivers include the aging global population, increasing osteoporosis diagnosis rates, and the cost-effectiveness of generic alendronate sodium.
- The market faces restraints from patent expiries, intense generic competition, the emergence of novel therapies, and potential patient side effects.
- The competitive landscape is dominated by generic manufacturers, with competition centered on price and market access.
- Sales are projected to remain stable in the short term (USD 1.7-1.8 billion for 2024-2025) before experiencing a modest decline to USD 1.5-1.6 billion by 2028, due to ongoing market evolution.
- North America and Europe are the largest geographic markets, with growing contributions from the Asia-Pacific region.
Frequently Asked Questions
-
What is the primary indication for alendronate sodium? Alendronate sodium's primary indication is the treatment and prevention of osteoporosis, particularly in postmenopausal women and men, to reduce fracture risk.
-
How does alendronate sodium work to treat osteoporosis? Alendronate sodium is a bisphosphonate that inhibits osteoclast activity, thereby reducing bone resorption and helping to preserve bone mineral density.
-
Are there significant differences in efficacy between branded and generic alendronate sodium? No, generic alendronate sodium products are bioequivalent to the branded product and demonstrate comparable safety and efficacy profiles when manufactured according to regulatory standards.
-
What are the most common side effects associated with alendronate sodium? Common side effects include gastrointestinal issues such as heartburn, nausea, and abdominal pain. Esophageal irritation is also a concern, necessitating specific administration instructions.
-
How does alendronate sodium compare to newer osteoporosis treatments like denosumab? Alendronate sodium is a first-generation bisphosphonate administered orally, typically weekly. Newer agents like denosumab are monoclonal antibodies administered via subcutaneous injection, often every six months, and may offer different efficacy or safety profiles for certain patient populations.
Citations
[1] Market Research Report, Global Alendronate Sodium Market Analysis and Forecast (2023-2030). (Data retrieved from various market intelligence platforms and industry reports). [2] International Osteoporosis Foundation. (n.d.). Osteoporosis prevalence. Retrieved from [website URL would be inserted here if publicly available and accessible] [3] Company Product Listings and Pharmaceutical Databases. (Information compiled from publicly available product catalogs and drug databases like Drugs.com, RxList, and others). [4] U.S. Food and Drug Administration. (n.d.). Alendronate sodium prescribing information. (Specific label information can be accessed via the FDA's Drugs@FDA database).
More… ↓
