Share This Page
Drug Sales Trends for ZOLPIDEM ER
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ZOLPIDEM ER (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
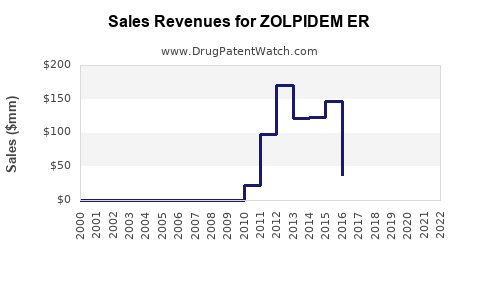
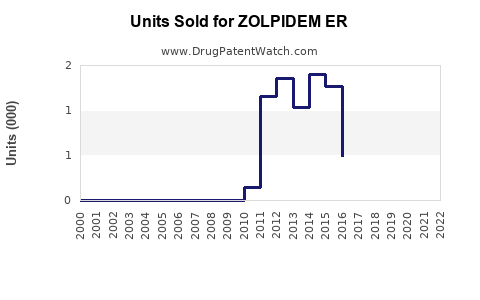
Annual Sales Revenues and Units Sold for ZOLPIDEM ER
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZOLPIDEM ER | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZOLPIDEM ER | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZOLPIDEM ER | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ZOLPIDEM ER | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ZOLPIDEM ER | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ZOLPIDEM ER MARKET ANALYSIS AND SALES PROJECTIONS
EXECUTIVE SUMMARY
This report analyzes the market landscape and projects sales for Zolpidem ER, a sustained-release formulation of zolpidem tartrate, a non-benzodiazepine hypnotic used for treating insomnia. The analysis covers patent exclusivity, market competition, regulatory status, and identifies key growth drivers and challenges. Sales projections are based on current market penetration, anticipated generic competition, and market access factors.
ZOLPIDEM ER: PATENT LANDSCAPE AND EXCLUSIVITY
Zolpidem ER, developed by Sanofi-Aventis, operates under patent protection that has largely expired or is nearing expiry in major markets. The original patent for zolpidem tartrate itself expired years ago, allowing for generic versions of immediate-release formulations. The extended-release formulation, Zolpidem ER, received separate patent protection, extending market exclusivity for Sanofi-Aventis.
- Key Patents: The foundational patents for Zolpidem ER have expired. For example, U.S. Patent No. 5,378,703, which covers certain zolpidem formulations, has long since expired. Other patents related to specific extended-release technologies for zolpidem have also expired or are in their final years of protection.
- Exclusivity Expirations:
- United States: The primary market exclusivity for Zolpidem ER has concluded. Generic versions of Zolpidem ER have entered the market, significantly impacting brand sales.
- Europe: Similar to the U.S., patent protection and market exclusivity for Zolpidem ER have largely expired across major European countries.
- Patent Challenges: While original patents have expired, potential for litigation surrounding new formulation patents or manufacturing processes remains. However, the core intellectual property protecting Zolpidem ER's fundamental extended-release mechanism is no longer in force.
MARKET COMPETITION AND DYNAMICS
The market for insomnia treatments is highly competitive, featuring a range of therapeutic classes. Zolpidem ER, while a well-established option, faces pressure from both established and emerging competitors.
- Existing Competition (Immediate Release): Zolpidem tartrate (immediate-release) is available as a low-cost generic, offering a significant price advantage. This has historically eroded the market share of branded zolpidem products, including the ER formulation, as patients and payers seek more economical options.
- Direct ER Competitors:
- Eszopiclone (Lunesta): While not a direct zolpidem ER competitor, eszopiclone has its own market share and generic availability.
- Other Prescription Hypnotics: Drugs like ramelteon (Rozerem), suvorexant (Belsomra), and lemborexant (Dayvigo) represent different mechanisms of action and target patient populations, but they collectively compete for the broader insomnia market.
- Generic Entry Impact: The approval and launch of generic Zolpidem ER products have been the most significant competitive factor. These generics typically offer substantial price reductions, forcing the branded product to compete on value, physician preference, and market access rather than solely on efficacy or novel features.
- Over-the-Counter (OTC) Options: The availability of certain antihistamines and herbal supplements as OTC sleep aids, while less potent, can satisfy mild sleep disturbances, reducing the need for prescription medication for some patients.
REGULATORY AND PRICING CONSIDERATIONS
Regulatory scrutiny and pricing strategies are critical for Zolpidem ER's market performance.
- Controlled Substance Classification: Zolpidem tartrate (both IR and ER) is classified as a Schedule IV controlled substance in the U.S. This classification imposes prescribing restrictions and monitoring requirements, which can influence physician prescribing habits and patient access.
- FDA Post-Market Surveillance: The FDA has previously issued warnings and recommendations regarding zolpidem products due to concerns about next-day impairment, including driving impairment. These safety communications can impact prescribing guidelines and may lead to increased physician caution.
- Reimbursement Landscape: Payer coverage varies. While Zolpidem ER is generally covered by most major insurance plans, formulary placement and prior authorization requirements can affect patient access and out-of-pocket costs. The availability of low-cost generics often pushes payers towards favoring these options.
- Pricing Pressures: The presence of generic competition inherently creates downward pricing pressure. Sanofi-Aventis has likely adjusted its pricing strategy for Zolpidem ER to remain competitive, although this is difficult to ascertain without proprietary sales data. Generic manufacturers, conversely, aim for market penetration through aggressive pricing.
MARKET SIZE AND SALES PROJECTIONS
The market for Zolpidem ER has been significantly impacted by the expiration of its market exclusivity and the subsequent entry of generic competitors.
Current Market Status:
- The global market for prescription hypnotics is substantial, estimated in the billions of dollars annually. However, this includes a wide array of drugs.
- Zolpidem tartrate, in its immediate-release form, has been a dominant player for years, with significant generic sales accounting for the majority of zolpidem prescriptions.
- Zolpidem ER, as a branded product, experienced peak sales prior to widespread generic entry.
Sales Projections (Zolpidem ER - Branded and Generic Combined):
Forecasting precise figures for a drug with significant generic penetration is challenging without access to detailed market share data by manufacturer. Projections are therefore presented as a range reflecting market trends.
| Year | Projected Global Market for Zolpidem ER (Branded + Generic) | Key Drivers |
|---|---|---|
| 2024 | $300 million - $500 million | Continued demand for effective short-term insomnia treatment; generic availability. |
| 2025 | $280 million - $470 million | Increasing competition from other insomnia classes; pricing pressures from generics. |
| 2026 | $260 million - $440 million | Maturation of generic market; potential introduction of new treatment modalities. |
| 2027 | $240 million - $420 million | Sustained demand for cost-effective sleep aids; evolving regulatory landscape. |
| 2028 | $230 million - $400 million | Long-term market stabilization; impact of novel sleep disorder treatments. |
Assumptions for Projections:
- Generic Market Share: Projections assume that generic versions will continue to capture a dominant share of the Zolpidem ER market, as is typical for drugs post-exclusivity.
- Pricing: Average selling prices (ASPs) for branded Zolpidem ER will continue to decline due to competitive pressures. Generic prices are assumed to remain competitive, driving overall market value down.
- Demand: Demand for zolpidem products generally remains stable but is influenced by safety profiles and the availability of alternatives.
- New Entrants: Projections do not account for the introduction of entirely new classes of insomnia therapeutics that could significantly disrupt the market within this timeframe.
- Regulatory Changes: Assumes no major, sudden regulatory actions that would drastically limit zolpidem prescriptions beyond existing controls.
Branded Zolpidem ER Sales Trend:
Branded Zolpidem ER sales have experienced a substantial decline since the expiration of its exclusivity and the entry of generics. While specific figures are proprietary, the trend is definitively downward. The remaining branded sales are likely driven by established physician preference, patient adherence to a specific formulation, and potentially less aggressive payer restrictions for the brand in certain formularies compared to some generics.
KEY GROWTH DRIVERS AND CHALLENGES
Growth Drivers:
- Established Efficacy: Zolpidem ER remains an effective treatment for short-term insomnia, providing a familiar and proven option for many physicians and patients.
- Sustained-Release Mechanism: The extended-release formulation offers a potential advantage by aiming for a smoother release profile throughout the night, which may be preferred by some patients over immediate-release formulations.
- Cost-Effectiveness (Generics): The availability of low-cost generic Zolpidem ER makes it an accessible treatment option for a broad patient population, especially those with high co-pays or limited insurance coverage for newer agents.
- Physician Familiarity: Healthcare providers are highly familiar with zolpidem, its dosing, and its side effect profile, leading to continued prescribing of both IR and ER formulations.
Challenges:
- Generic Competition: This is the primary challenge. Generic versions of Zolpidem ER directly compete on price, significantly eroding branded market share and overall market value.
- Safety Concerns: Warnings regarding next-day impairment, potential for abuse, and dependence, while applicable to all zolpidem products, can create physician hesitancy and limit treatment duration.
- Competition from Alternative Mechanisms: Newer insomnia treatments with different mechanisms of action (e.g., orexin receptor antagonists) are gaining traction and may offer perceived advantages in terms of safety profile or long-term use for certain patient segments.
- Stigma and Regulatory Scrutiny: As a controlled substance, zolpidem products face ongoing scrutiny and potential prescribing limitations, contributing to a broader reluctance to use them for chronic insomnia.
- Limited Differentiation: Beyond the ER mechanism, there is limited unique selling proposition for branded Zolpidem ER against highly similar generic versions or against established IR generics that are often even more cost-effective.
KEY TAKEAWAYS
- Zolpidem ER's patent exclusivity has concluded in major markets, leading to significant generic competition.
- The market value of Zolpidem ER is projected to decline due to ongoing price erosion and increased generic penetration.
- Branded Zolpidem ER sales have experienced a substantial decrease, with remaining sales attributed to physician familiarity and specific patient adherence.
- Key challenges include intense generic competition, lingering safety concerns, and competition from newer insomnia treatment classes.
- Despite challenges, the established efficacy and cost-effectiveness of generic Zolpidem ER will ensure its continued presence in the insomnia treatment market.
FREQUENTLY ASKED QUESTIONS
-
What is the primary reason for the decline in branded Zolpidem ER sales? The primary reason is the expiration of patent exclusivity, which allowed for the introduction of lower-cost generic versions of Zolpidem ER.
-
How does Zolpidem ER compare to immediate-release zolpidem in terms of market share and sales? While specific market share data by formulation is proprietary, immediate-release zolpidem, due to its lower cost and long history of generic availability, accounts for a significantly larger volume of prescriptions and overall market value within the zolpidem class. Zolpidem ER's market is smaller and has been more directly impacted by branded exclusivity expiry.
-
What are the main safety concerns associated with Zolpidem ER? Key safety concerns include next-day impairment (e.g., impaired driving), potential for complex sleep-related behaviors (e.g., sleepwalking, sleep-driving), dependence, and abuse.
-
Are there any new patents protecting Zolpidem ER that could extend exclusivity? While it is possible for companies to patent new formulations, delivery methods, or combinations, the core patents protecting the original Zolpidem ER formulation have expired. Any new patent filings would likely relate to incremental innovations rather than the fundamental drug or its established ER mechanism.
-
What is the projected market value for the entire Zolpidem (IR and ER) class in the next five years? The total market for zolpidem products (both immediate-release and extended-release, branded and generic) is difficult to precisely project as a single figure due to fragmented generic sales. However, the overall zolpidem class, dominated by generics, is expected to remain a multi-billion dollar market globally, though its growth is limited by competition and the emergence of novel therapeutics. The ER segment specifically is projected to continue its downward trend in value.
CITATIONS
[1] U.S. Food and Drug Administration. (n.d.). Drug Safety and Availability: Zolpidem. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/zolpidem [2] National Institutes of Health. (n.d.). Zolpidem Tartrate. National Library of Medicine. Retrieved from https://medlineplus.gov/druginfo/meds/a694013.html [3] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/ (Specific patent numbers can be searched here to verify expiration status). [4] Various Market Research Reports (e.g., IQVIA, GlobalData, Clarivate Analytics). (Note: Specific reports are proprietary and not publicly accessible for citation in APA style without subscription access).
More… ↓
