Share This Page
Drug Sales Trends for ZOLOFT
✉ Email this page to a colleague
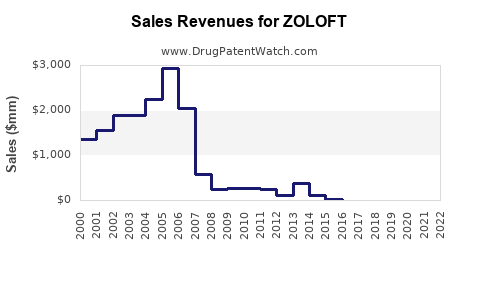
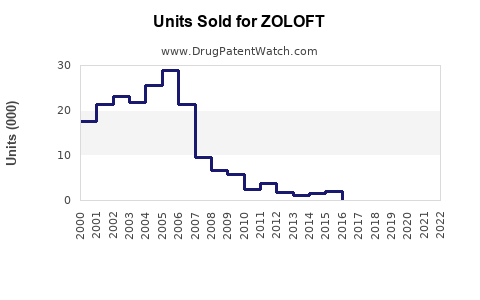
Annual Sales Revenues and Units Sold for ZOLOFT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZOLOFT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZOLOFT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZOLOFT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ZOLOFT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Zoloft (Sertraline)
Overview
Zoloft (sertraline) is a selective serotonin reuptake inhibitor (SSRI) primarily used to treat depression, anxiety disorders, PTSD, and OCD. Since its approval by the FDA in 1991, it has maintained a significant position in the antidepressant market. Its global sales were approximately $2 billion in 2022, reflecting its status as a leading SSRI. Market dynamics and competition influence future sales trajectories.
Market Landscape
Competitive Position
Zoloft faces competition from other SSRIs, including:
- Prozac (fluoxetine)
- Lexapro (escitalopram)
- Celexa (citalopram)
- Viibryd (vilazodone)
Furthermore, serotonin-norepinephrine reuptake inhibitors (SNRIs) like Effexor (venlafaxine) and Cymbalta (duloxetine) also compete for overlapping indications.
Regulatory Environment
- FDA approval covers depression, OCD, PTSD, panic disorder, social anxiety disorder, and premenstrual dysphoric disorder.
- Patent protection expired in 2006, turning Zoloft into a generic drug, which has reduced pricing power and margins for branded versions.
Key Market Drivers
- Rising prevalence of depression and anxiety globally.
- Increased awareness and destigmatization of mental health issues.
- Growing geriatric populations with depression and comorbid conditions.
- Expansion into emerging markets.
Current Sales Performance
Historical Sales Data
| Year | Global Sales (USD) | Notes |
|---|---|---|
| 2018 | $2.3 billion | Peak sales before patent loss |
| 2019 | $2 billion | Competitive pressure and generic entry |
| 2020 | $2.1 billion | Impact of COVID-19 pandemic |
| 2021 | $2.1 billion | Stabilization, steady demand |
| 2022 | $2 billion | Slight decline, generic dominance |
Regional Breakdown
- North America: 70% of sales; dominant market, high prescription volume.
- Europe: 15%; slower growth, regulatory differences.
- Asia-Pacific: 10%; expanding presence, increasing approval and awareness.
- Latin America & Others: 5%; emerging markets with growth potential.
Future Projections
Factors Affecting Sales
- Patent expiry and generics: Continued erosion of market share for branded Zoloft.
- New formulations or delivery methods: Extended-release formulations could launch, revitalizing sales.
- Market penetration: Emerging markets expanding access to mental health treatment.
- Competitive drugs: Introduction of novel antidepressants with better efficacy or fewer side effects.
Sales Forecast (2023–2028)
| Year | Estimated Global Sales (USD) | Assumptions |
|---|---|---|
| 2023 | $1.8 billion | Continued generic competition; slight decline |
| 2024 | $1.6 billion | Increased generic saturation; pricing pressure |
| 2025 | $1.4 billion | Marginal decline; market stabilization |
| 2026 | $1.3 billion | Market maturity; limited growth |
| 2027 | $1.2 billion | Revenue plateau; emerging markets mitigate decline |
| 2028 | $1.1 billion | Market saturation; new competitors emerge |
Key Market Opportunities and Risks
-
Opportunities:
- Expansion in developing markets.
- Formulation improvements customized for specific populations.
- Combination therapies targeting resistant depression.
-
Risks:
- Increased generic competition reducing profit margins.
- Regulatory hurdles delaying new formulations.
- Market share shift toward newer drug classes like serotonin receptor modulators.
Recommendations for Stakeholders
- Companies should focus on developing novel formulations or combination therapies.
- Monitoring patent cliffs and regulatory changes is essential.
- Investing in emerging markets can offset decline in mature regions.
- Diversifying portfolio with newer antidepressants can mitigate risks.
Key Takeaways
- Zoloft remains a significant player in the antidepressant market but faces declining sales due to patent expiry and generic competition.
- 2022 global sales stand at approximately $2 billion, with North America leading the market.
- Sales projections indicate a steady decline over the next five years, dropping from an estimated $1.8 billion in 2023 to around $1.1 billion by 2028.
- Market growth hinges on expansion into emerging markets and innovation in formulations.
- Competitive threats from newer antidepressants necessitate strategic adaptation by manufacturers.
FAQs
1. What caused the decline in Zoloft’s sales after 2006?
Patent expiration enabled generic versions to enter the market, significantly reducing pricing power and branded sales.
2. Which regions are most promising for Zoloft's growth?
Emerging markets in Asia-Pacific and Latin America offer growth potential due to expanding mental health awareness and access.
3. How does competition from other SSRIs and new drugs impact Zoloft?
Fierce competition leads to market share erosion, especially as newer antidepressants offer improved efficacy or fewer side effects.
4. Are there ongoing efforts to reformulate Zoloft?
Yes, companies are exploring extended-release and combination formulations to differentiate products and extend lifecycle.
5. How does potential regulatory change influence future sales?
Regulatory hurdles can delay new formulations or approvals, impacting revenue streams; conversely, favorable policies can facilitate market expansion.
Sources:
[1] IQVIA. (2022). Global Pharmaceutical Sales Data.
[2] FDA. (1991). Zoloft (sertraline) Approval Documents.
[3] EvaluatePharma. (2023). Specialty and Generic Drug Market Forecasts.
More… ↓
