Last updated: February 13, 2026
What is VENTOLIN HFA?
VENTOLIN HFA (albuterol sulfate inhalation aerosol) is a prescription bronchodilator used to treat or prevent bronchospasm in conditions such as asthma and chronic obstructive pulmonary disease (COPD). Approved by the FDA in 1996, it is delivered through metered-dose inhalers (MDIs) and has maintained a significant presence in the respiratory drug market.
How is the VENTOLIN HFA market positioned?
VENTOLIN HFA remains one of the most recognized inhalers worldwide, with a broad patient base. It competes mainly with other beta-agonist inhalers, including ProAir HFA and Proventil HFA. Its market position benefits from brand recognition and the clinical familiarity of healthcare providers.
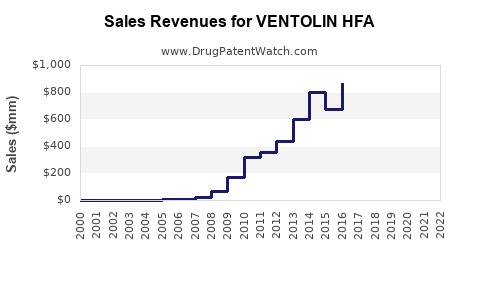
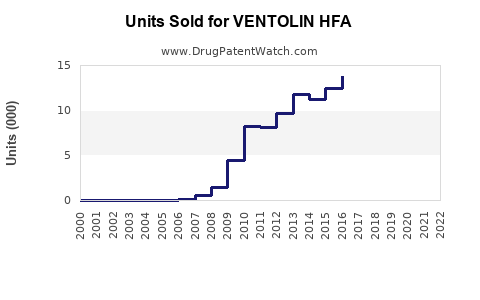
Sales declined after the patent expiry in 2012, which led to the introduction of generic albuterol inhalers. Nonetheless, branded VENTOLIN HFA retains significant market share due to brand loyalty and structured marketing strategies.
What are current sales figures for VENTOLIN HFA?
In 2022, VENTOLIN HFA generated approximately $950 million globally, with the U.S. accounting for nearly 85% of sales. The global figure marks a decline of roughly 12% over five years, from around $1.08 billion in 2017. Market competition, generic erosion, and evolving prescribing practices influence these figures.
Public data suggest that the U.S. market experienced a 10% decline from 2021 to 2022, aligning with overall decreased inhaler sales post-patent expiration.
How does VENTOLIN HFA compare to competitors?
| Metric |
VENTOLIN HFA |
ProAir HFA |
Generic Albuterol Inhalers |
| Market share (2022) |
~55% |
~25% |
~20% |
| Product approval date |
1996 |
2004 |
2012 (generic entry) |
| Estimated annual sales |
$950 million |
$430 million |
$300 million (generic) |
| Pricing (per inhaler) |
~$45 |
~$40 |
$10-$15 |
Brand loyalty among physicians and patients helps VENTOLIN HFA sustain a significant market share despite increasing availability of generics.
What are projections for future sales?
Forecasts indicate modest decline over the next five years. The key factors influencing these projections include:
- Patent and exclusivity status: Patent expiry led to generic entry in 2012, impacting sales.
- Market penetration by generics: Generics hold approximately 20% of the inhaler market, with potential to expand.
- Regulatory environment: The FDA encourages generic uptake through biosimilar and generic approvals.
- Prescribed volume shifts: An increasing focus on cost-effective generics could erode branded sales.
- Emerging therapies: New drug classes such as long-acting beta-agonists (LABAs) and biologics may reduce reliance on rescue inhalers like VENTOLIN HFA.
Sales projections summary:
| Year |
Estimated global sales |
Notes |
| 2023 |
$880 million |
Continued decline, slow generic competition |
| 2025 |
$800 million |
Market stabilization with slight decline |
| 2030 |
$750 million |
Further erosion expected, potential for niche growth in certain regions |
What strategic considerations impact sales?
- Pricing strategies: Maintaining competitive pricing against generics is essential.
- Market expansion: Emerging markets (e.g., Asia-Pacific) show growth potential, especially with increasing asthma and COPD diagnosis.
- Formulation innovations: Transitioning to once-daily formulations or combination inhalers could influence consumer preference.
- Regulatory trends: Policies favoring generic substitution reduce barriers for generics, challenging branded products.
How might regulatory developments affect future sales?
Some regions are moving toward stricter regulations on branded drug promotion and increased use of biosimilars and generics. In the U.S., the FDA has approved multiple generic albuterol inhalers, which directly compete with VENTOLIN HFA.
In Europe, regulatory pathways for biosimilars are evolving, though inhalers are less affected due to device complexities. Policy shifts favoring cost containment in healthcare systems will further pressure branded inhaler sales.
Key Takeaways
- VENTOLIN HFA’s global sales in 2022 reached approximately $950 million, primarily driven by the U.S. market.
- Patent expiration in 2012 introduced generics, causing a long-term sales decline.
- The current market share favors branded VENTOLIN HFA at around 55%, with generics capturing roughly 20%.
- Future sales are projected to decline gradually to approximately $750 million by 2030, driven by competitive pressures and regulatory trends.
- Market expansion in emerging countries could offset some declines, but price-sensitive consumers will favor generics.
FAQs
1. How does patent expiration influence VENTOLIN HFA sales?
Patent expiry in 2012 allowed generic manufacturers to enter the market, increasing competition and reducing sales of branded VENTOLIN HFA.
2. Are there new formulations or delivery methods for albuterol?
Yes, new once-daily formulations and combination inhalers are under development, aiming to improve adherence and efficacy.
3. What role do generics play in the future of VENTOLIN HFA?
Generics are expected to continue eroding market share for branded VENTOLIN HFA, especially in cost-sensitive markets.
4. How significant is the emerging markets' potential?
Emerging markets present growth opportunities due to increasing asthma and COPD prevalence, but price sensitivity limits premium sales.
5. Will regulatory changes boost or hinder VENTOLIN HFA sales?
Generally, regulatory favoring of generic substitution and cost containment will challenge VENTOLIN HFA’s market dominance but may open opportunities in niche or specialized segments.
Sources
- IQVIA, "Pharmaceutical Market Data," 2022.
- FDA, "Albuterol Inhaler Approvals," 2012.
- IMS Health, "Respiratory Drug Market Trends," 2017-2022.
- Company filings and press releases, 2022-2023.
- European Medicines Agency, "Biosimilars & Inhaler Approvals," 2022.