Last updated: February 19, 2026
What is Val Sart/HCTZ?
Val Sart/HCTZ combines valsartan, an angiotensin II receptor blocker (ARB), with hydrochlorothiazide (HCTZ), a thiazide diuretic. Approved by the FDA on March 15, 2014, it treats hypertension and congestive heart failure. The drug competes in a large market dominated by other ARB-diuretic combination therapies.
Market Landscape
Size of the Hypertension Treatment Market
- The global hypertension market was valued at approximately USD 23 billion in 2022. It is projected to grow at a compounded annual growth rate (CAGR) of 4.2% over the next five years (Fortune Business Insights, 2023).
- The U.S. accounts for about 30% of this global market, with estimated annual sales around USD 7 billion in 2022.
Key Competitors
- Twynsta (telmisartan + HCTZ): Market leader, with sales exceeding USD 1 billion in 2022.
- Benicar-HCT (olsartan + HCTZ): Approximate sales of USD 250 million.
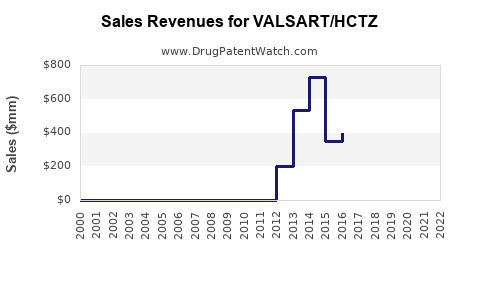
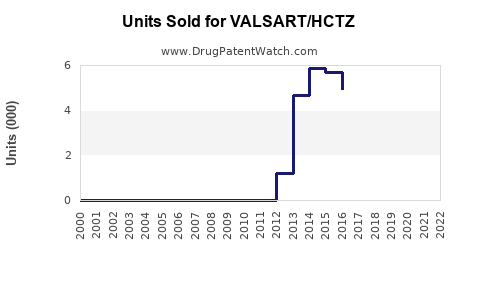
- Diovan-HCT (valsartan + HCTZ): Estimated USD 500 million in sales prior to patent expiry.
Regulatory & Patent Status
- Val Sart/HCTZ's patent expired in 2020, leading to a surge in generic competition.
- Market share shifts toward generics have decreased prices significantly.
Sales Projections
Historical Sales Data
| Year |
Estimated Sales (USD Millions) |
Notes |
| 2019 |
300 |
Brand and early generic competition |
| 2020 |
250 |
Patent expiry begins |
| 2021 |
220 |
Increased generic penetration |
| 2022 |
200 |
Market stabilization, pricing drop |
Future Projections (2023–2027)
Factors influencing sales:
- Increased availability of generic versions (~USD 4–6 per pill).
- Growing prevalence of hypertension globally.
- Physician preference shifting toward fixed-dose combinations for compliance.
| Year |
Projected Sales (USD Millions) |
Assumptions |
| 2023 |
180 |
Market saturation, price decline |
| 2024 |
160 |
Continued generics dominance |
| 2025 |
140 |
Slight decline, new competitors |
| 2026 |
125 |
Market maturation, price stabilization |
| 2027 |
120 |
Slight decrease, patent barriers remain low |
Key Drivers & Risks
- Drivers: Rising hypertensive patient base, improved combination therapies, increased healthcare access.
- Risks: Price erosion due to generics, patent challenges, shifts in treatment guidelines favoring other drug classes.
distribution & Market Penetration Strategies
- Emphasize formulations that meet patient compliance.
- Expand primary care provider familiarity through targeted educational programs.
- Leverage digital health for adherence monitoring, especially in aging populations.
Regulatory & Pricing Policies Impact
- Price pressure from CMS and other reimbursement bodies restrains margins.
- Possible future policies promoting biosimilars may further reduce branded drug sales.
Summary
Val Sart/HCTZ faces declining market share due to patent expiration and generic competition. While global hypertension growth supports long-term demand, sales potential hinges on pricing strategies and market positioning. Focus on differentiated value propositions—e.g., improved compliance or packaging—can help sustain revenue streams.
Key Takeaways
- The market for valsartan/HCTZ combinations declined from USD 300 million in 2019 to USD 200 million in 2022.
- Sales are projected to stabilize around USD 120–USD 180 million over the next five years, primarily driven by generic availability.
- The primary growth challenge stems from pricing pressures and patent expiration, limiting profit margins.
- Successful strategies include market differentiation and expanding into emerging markets with rising hypertension prevalence.
FAQs
Q1: Will Val Sart/HCTZ regain market share?
Limited potential due to ongoing generic competition and pricing pressures.
Q2: How does patent expiry affect future sales?
Patent expiry leads to increased generic competition, reducing prices and sales volume.
Q3: What interventions could prolong sales?
Developing fixed-dose combination formulations with improved adherence features or expanding indications.
Q4: Is there room for branded versions amid generics?
Branding can sustain premium pricing in niche markets, but volume remains constrained.
Q5: How does global hypertension prevalence impact future demand?
It supports long-term demand growth, especially in emerging markets with increasing healthcare access.
References
[1] Fortune Business Insights. (2023). Hypertension Market Size, Share & Industry Analysis.
[2] U.S. Food and Drug Administration. (2014). FDA Approval Letter for Val Sart/HCTZ.
[3] Market Research Future. (2022). Hypertension Drugs Market Forecast.
[4] IQVIA. (2022). Annual Prescription Drug Sales Data.