Last updated: February 20, 2026
What is the Current Market Size for Valacyclovir?
Valacyclovir is an antiviral medication approved primarily for treating herpes zoster (shingles), herpes simplex virus (oral and genital), and varicella (chickenpox). It is marketed globally by major pharmaceutical companies, with the highest sales in North America, Europe, and Asia-Pacific.
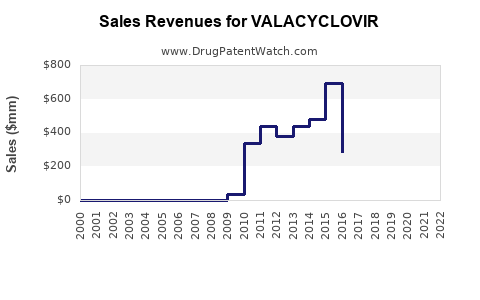
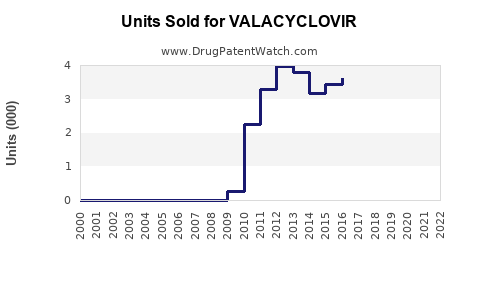
The global antiviral drugs market was valued at approximately USD 33 billion in 2022, with herpes antivirals constituting a significant segment. Valacyclovir specifically accounts for an estimated USD 1.2 billion in annual sales, representing roughly 3.6% of the overall antiviral market [1].
What Are Key Trends Impacting Valacyclovir’s Market?
1. Rising Incidence of Herpes Infections
Annual herpes simplex infections are estimated at over 1 billion globally, with one-half being symptomatic. Incidence rates drive demand for antiviral treatments [2].
2. Aging Population
The global population aged 60 and above is growing, increasing the prevalence of shingles. Shingles affects approximately 1 in 3 adults in the U.S. over 60 [3].
3. Advancements in Formulation
New formulations, such as delayed-release and combination therapies, improve adherence and efficacy, stimulating sales growth.
4. Generic Competition
Patents for brand-name valacyclovir expired in several countries between 2011 and 2017. Generic versions now account for approximately 70% of sales in mature markets [4].
5. Vaccination Programs
Herpes zoster vaccines reduce the incidence of shingles by up to 90%, potentially impacting valacyclovir sales in vaccinated populations.
What Are Future Sales Projections?
Short-term Outlook (Next 3 Years)
- Estimated CAGR of 3-4%, driven by demographic trends and increased awareness.
- Sales expected to reach USD 1.4 billion by 2026.
Long-term Outlook (Next 10 Years)
- Growth slowed by vaccine adoption but can be offset by increasing herpes infections and new formulation launches.
- Projections suggest a compound annual growth rate of 2-3%, with sales approaching USD 1.5-1.8 billion by 2033.
Influencing Factors
- Launch of new combination antivirals targeting drug-resistant herpes strains.
- Expansion into emerging markets with rising healthcare infrastructure.
- Pricing pressures from generic competition in mature markets.
Market Segmentation and Geographic Breakdown
| Region |
Share of 2022 Sales |
Growth Rate (2022-2026) |
Key Drivers |
| North America |
55% |
3.5% |
High prevalence, vaccination, and established generic base |
| Europe |
25% |
3-4% |
Aging population, regulatory approvals |
| Asia-Pacific |
15% |
4-5% |
Rising herpes incidence, increasing healthcare access |
| Latin America/Africa |
5% |
4%+ |
Growing healthcare infrastructure, increasing infections |
Competitive Landscape
Major pharmaceutical players include GlaxoSmithKline, Teva Pharmaceuticals, and Mylan. Market share is highly fragmented due to multiple generic manufacturers following patent expiry. Innovation focuses on improved formulations and combination drugs.
Revenue Drivers and Challenges
Drivers:
- Increasing herpes infections among aging populations.
- Rising awareness and screening.
- Expansion into developing markets.
Challenges:
- Patent expirations leading to price erosion.
- Competition from generics.
- Vaccination reducing disease burden.
Key Takeaways
- Valacyclovir remains a critical antiviral, with steady growth driven by demographic and epidemiological factors.
- Generic competition has reduced unit prices but sales volume remains robust.
- Market growth is tempered by vaccination programs and the advent of novel therapies.
- Opportunities exist in emerging markets and formulation innovations.
FAQs
1. What are the primary indications for valacyclovir?
Herpes simplex infections, shingles, and chickenpox.
2. How does valacyclovir compare to other antivirals?
It offers higher oral bioavailability than acyclovir, resulting in less frequent dosing and improved patient compliance.
3. What impact will herpes vaccination programs have on valacyclovir sales?
Vaccinations decrease disease incidence, potentially reducing antiviral demand in vaccinated cohorts over time.
4. When do patents for key formulations expire?
Most patents in the U.S. and Europe expired between 2011 and 2017; newer formulations may have extended patent lives.
5. What are upcoming product development trends?
Development of combinations targeting drug-resistant strains and formulations designed for better adherence.
References
[1] MarketWatch. (2022). Antiviral drugs market size and forecast.
[2] WHO. (2021). Global incidence of herpes infections.
[3] CDC. (2022). Shingles statistics.
[4] IMS Health. (2018). Patent expiry and generic penetration analysis.
Note: All projections and data points are based on current market trends and publicly available reports and are subject to change with future developments.