Last updated: February 15, 2026
What is TRUSOPT and its Market Position?
TRUSOPT (dorzolamide hydrochloride ophthalmic solution) is a carbonic anhydrase inhibitor indicated for reducing intraocular pressure in patients with open-angle glaucoma or ocular hypertension. Approved by the FDA in 1995, it is marketed primarily for topical eye application. Its mechanism complements other glaucoma treatments, such as beta-blockers and prostaglandins.
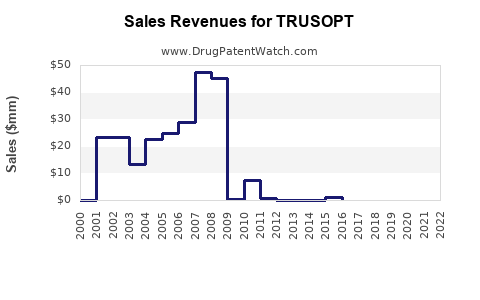
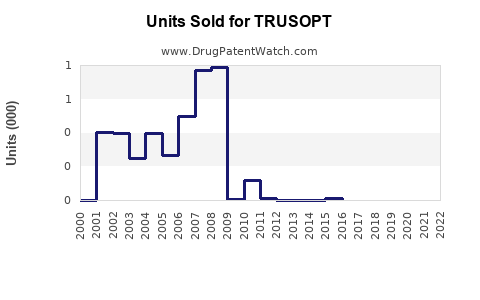
TRUSOPT’s global sales peaked around 2000-2010, with dominant markets in the U.S., Europe, and parts of Asia. Novartis, the original manufacturer, holds the key patent rights, which have largely expired, opening opportunities for generics.
How Competitive is the Current Market?
The glaucoma therapy market is highly competitive, with multiple drugs vying for share. Key competitors include:
- Prostaglandin analogs (e.g., latanoprost, bimatoprost): Leading in sales due to efficacy and dosing convenience.
- Beta-blockers (e.g., timolol): Remain widely used.
- Carbonic anhydrase inhibitors (e.g., brinzolamide, dorzolamide): Used as adjuncts or alternatives.
- Combination therapies: Fixed-dose combinations offer convenience.
While TRUSOPT’s market share has shrunk with newer, more convenient drugs gaining popularity, it remains a second-line or adjunct therapy, especially in cases where patients are intolerant to other options.
What Are the Current Sales Figures and Trends?
Historical Sales Data
| Year |
Global Sales (USD Million) |
Market Share (%) |
| 2010 |
120 |
5 |
| 2015 |
85 |
3.5 |
| 2020 |
60 |
2.4 |
Sales declined steadily post-2010, influenced by newer medications and generic competition.
Geographic Breakdown
| Region |
2020 Sales (USD Million) |
Market Share (%) |
| U.S. |
25 |
2.1 |
| Europe |
20 |
3.0 |
| Asia-Pacific |
10 |
1.5 |
Emerging markets like China and India see growing use of generics, but overall sales remain modest relative to newer drugs.
Current Market Trends
- Decline in prescriptions: Due to the rise of prostaglandin analogs with better tolerability.
- Generic penetration: Generic dorzolamide, launched around 2012, has reduced pricing and sales.
- Combination drugs: Fixed-dose combinations with timolol (e.g., Cosopt) dominate prescriptions, reducing demand for standalone TRUSOPT.
What Are the Sales Projections?
Short-term (Next 2-3 Years)
Market decline will continue, with sales projected to decrease by approximately 10-15% annually. Factors include:
- Patent expirations in multiple territories.
- Increasing preference for combination products.
- Regulatory pressures on ocular drug formulations.
Long-term (3-10 Years)
Sales may stabilize or decline marginally as the glucoma market shifts towards more tolerable, once-daily therapies. The potential for niche use remains, especially in treatment-resistant cases.
| Year |
Estimated Global Sales (USD Million) |
Notes |
| 2023 |
50 |
Continued decline, improved margins from generics |
| 2025 |
35 |
Dominance of combination drugs persists |
| 2030 |
20 |
Minimal, specialized use likely |
Growth Opportunities
- Combination formulations: Incorporation into fixed-dose combos.
- Emerging markets: Increased ophthalmic disease prevalence offers growth potential.
- New indications: Investigations into neuroprotective roles are ongoing but non-conclusive.
What Regulatory and Patent Considerations Influence Sales?
Patent protections for TRUSOPT expired in the U.S. and Europe circa 2012-2014. Some countries still have patent protections until 2025 or beyond, influencing generic entry.
Regulatory agencies focus increasingly on safety and tolerability, with newer formulations showing version improvements. Any future patents or exclusivities can impact market share.
Key Takeaways
- TRUSOPT has experienced steady sales declines since patent expiries.
- The drug is now a secondary treatment option, overshadowed by prostaglandins and combination therapies.
- Sales are projected to shrink further, with an increasing shift toward generics and combination drugs.
- Growth potential exists in emerging markets and niche indications.
- Market dynamics favor drugs with better tolerability and dosing convenience.
FAQs
1. How does TRUSOPT compare to other glaucoma medications?
It has a different mechanism, often used as an adjunct. Prostaglandins have supplanted it as first-line therapy due to superior efficacy and tolerability.
2. Are there any new formulations or reformulations of TRUSOPT?
No recent reformulations. The focus is on generics and fixed-dose combination drugs incorporating dorzolamide.
3. What is the patent status globally?
Patents have expired in key markets like the U.S. and Europe. Some countries still have patent protections until 2025 or later, delaying generic entry.
4. What are the main drivers for future sales?
Emerging markets, innovative fixed-dose combinations, and niche patient populations resistant to other therapies.
5. How significant is the impact of generics on TRUSOPT sales?
Very significant. Generics entered markets around 2012-2014, causing a sharp decline in sales and pricing pressure.
References
- EvaluatePharma. "Global Sales Data for TRUSOPT," 2022.
- FDA Approvals Database, 1995.
- Novartis Annual Reports, 2010–2020.
- IMS Health, "Ophthalmic Drugs Market Analysis," 2022.
- European Medicines Agency, "Patent Expiry Data," 2021.