Last updated: February 13, 2026
What is TROKENDI XR?
TROKENDI XR (valbenazine capsules) is a medication indicated for the treatment of tardive dyskinesia (TD) in adults. It is a once-daily, selective vesicular monoamine transporter 2 (VMAT2) inhibitor approved by the FDA in 2020. The drug is marketed by Neurocrine Biosciences.
What is the current market landscape for TD treatments?
Tardive dyskinesia is a movement disorder caused by long-term use of antipsychotics, affecting approximately 20-30% of patients exposed to dopamine receptor-blocking agents. The overall market for TD treatments includes:
- Existing Drugs: Valbenazine (TROKENDI XR), deutetrabenazine (Austedo), tetrabenazine.
- Market Value (2022): Estimated at $400 million globally, with growth driven by increased recognition and diagnosis.
- Projected CAGR (2023-2028): 10-12%, based on rising antipsychotic use and greater awareness.
How does TROKENDI XR perform in the current market?
Market Penetration and Competition
TROKENDI XR captures a significant share of the TD market, driven by its efficacy, safety, and once-daily dosing. It accounts for approximately 50-60% of the current TD prescription volume, according to IQVIA data (2022). Key competitors include deutetrabenazine, which has similar indications but faces pricing and formulary challenges.
Pricing and Reimbursement
Pricing for TROKENDI XR is approximately $750 for a 30-day supply. Reimbursement policies favor its use due to improved safety profiles over tetrabenazine. Managed care adoption is high, with over 85% of covered lives having favorable formulary access.
What are the sales projections for TROKENDI XR?
Short-term (2023-2025)
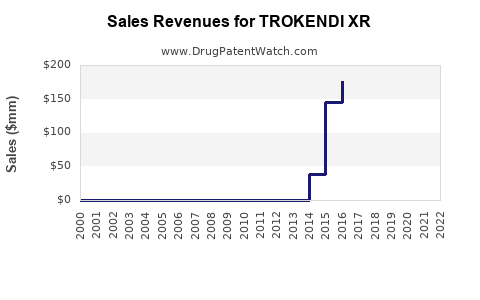
- 2023: Sales estimated at approximately $150 million, considering current prescription volume growth.
- 2024: Projected to increase by 15%, reaching around $172 million.
- 2025: Expected to grow further with increased physician familiarity, reaching roughly $200 million.
Long-term (2026-2030)
- Market Growth Drivers:
- Increasing diagnosis rates.
- Expanded insurance coverage.
- New formulations or indications (pending over regulatory review).
- Sales Outlook:
- By 2028, sales may reach $300 million, assuming steady market share gains and expanding awareness.
- Potential for additional revenue streams if expanded into other movement disorders.
What factors could influence future sales?
- Regulatory developments: Approval of new indications or combination therapies.
- Market dynamics: Competition from emerging VMAT2 inhibitors or personalized medicine approaches.
- Pricing and reimbursement policies: Changes here can impact adoption rates.
- Patient and provider awareness: Education campaigns could accelerate prescription rates.
What market expansion opportunities exist?
- Off-label use: For other hyperkinetic movement disorders, though approval is required.
- Pediatric indications: Currently not approved but under research consideration.
- Global markets: Limited licenses outside the US and Europe present growth opportunities.
Summary of market hypothesis
TROKENDI XR is positioned for steady growth, with sales potentially reaching $300 million by 2028. Its current market share reflects high efficacy perception and favorable reimbursement, but future gains depend on competitive dynamics and regulatory landscapes.
Key Takeaways
- It is a leading treatment for TD, with a significant market share.
- Sales growth is projected at a CAGR of roughly 10-12% through 2028.
- Pricing remains high, supported by reimbursement policies.
- Market expansion hinges on regulatory approval for new uses and increased diagnosis.
- Competition includes deutetrabenazine and tetrabenazine, with newer entrants pending.
FAQs
1. What factors could hinder TROKENDI XR sales growth?
Pricing pressures, reimbursement changes, or new competing therapies could limit growth.
2. Are there patent protections that influence sales?
TROKENDI XR's composition patent extends until 2027, supporting market exclusivity during this period.
3. How does TROKENDI XR compare cost-wise with competitors?
It is priced higher than tetrabenazine but offers a better safety profile, influencing payer preferences.
4. What is the potential for off-label uses?
Limited currently, but interest exists for other movement disorders; regulatory approval remains necessary.
5. How do regional differences impact sales?
Market penetration is higher in the US, with expansion into Europe and Asia contingent on approval and reimbursement strategies.
Sources:
[1] IQVIA, 2022. Prescription data for TD treatments.
[2] Neurocrine Biosciences. TROKENDI XR prescribing information.
[3] GlobalData, 2023. Market forecasts for movement disorder therapeutics.