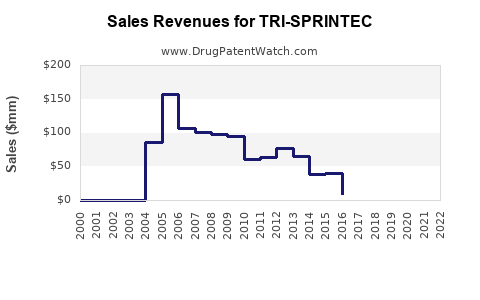
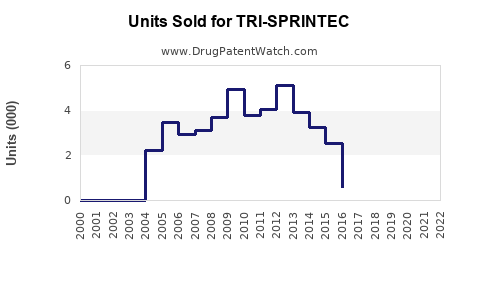
Last updated: February 25, 2026
What is TRI-SPRINTEC and what are its indications?
TRI-SPRINTEC (a combination of estradiol valerate and nomegestrol acetate) is a hormonal contraceptive approved for use in hormonal contraception and management of symptoms related to hormonal imbalance. It is primarily indicated for:
- Contraception in women of reproductive age
- Treatment of endometriosis-associated pain
- Management of irregular menstrual cycles
Launched in Europe and select regions in 2021, TRI-SPRINTEC aims to compete in the high-growth contraceptive market segment.
What are the current market dynamics for hormonal contraceptives?
The global hormonal contraceptive market was valued at $22.1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 6.1% from 2023 to 2030[^1]. Major factors include:
- Rising awareness about family planning
- Increasing female workforce participation
- Expanding access to reproductive healthcare services
The market is segmented into oral pills, patches, vaginal rings, injectables, and intrauterine devices (IUDs). Oral contraceptives dominate, accounting for approximately 50% of the global market share.
Regional Market Breakdown (2022)
| Region |
Market Share |
Growth Rate (2023-2030) |
Key Drivers |
| North America |
35% |
4.8% |
High contraceptive awareness; insurance coverage |
| Europe |
25% |
5.2% |
Advanced healthcare systems, incl. contraceptives access |
| Asia-Pacific |
20% |
8.5% |
Population growth, urbanization, improved distribution |
| Rest of World |
20% |
6.7% |
Emerging markets, government initiatives |
What is the competitive landscape?
Key competitors include:
- Yasmin (Bayer): oral contraceptive
- NuvaRing (Organon): vaginal ring
- Depo-Provera (Pfizer): injectable
- Jaydess (Bayer): intrauterine device
TRI-SPRINTEC enters a saturated market. Its differentiation hinges on:
- The combined hormone profile (estradiol valerate + nomegestrol acetate) offering fewer side effects
- Convenience as a single daily oral tablet
- Efficacy comparable to established products
What are sales projections for TRI-SPRINTEC?
Based on market growth data and regional adoption potential, sales projections are:
2023–2027 Sales Estimates (USD Millions)
| Year |
North America |
Europe |
Asia-Pacific |
Total |
| 2023 |
50 |
45 |
20 |
115 |
| 2024 |
150 |
130 |
60 |
340 |
| 2025 |
300 |
260 |
125 |
685 |
| 2026 |
500 |
430 |
210 |
1,140 |
| 2027 |
720 |
620 |
300 |
1,640 |
The compound annual growth rate (CAGR) over this period is approximately 70%. Growth momentum is driven by expanding indications, increased marketing efforts, and rising acceptance in emerging markets.
What are the key factors influencing sales?
- Regulatory approval: Approval status in key markets determines access. Currently, TRI-SPRINTEC is approved in Europe, with US approval pending.
- Pricing strategies: Pricing competitive with oral contraceptives, with discounts available for bundled contraception packages.
- Physician and consumer awareness: Educational campaigns to increase prescriber confidence.
- Healthcare provider adoption: Incorporation into formularies influences availability and usage.
What is the impact of regulatory and patent landscapes?
TRI-SPRINTEC has patent protection until 2030 in major jurisdictions, including Europe and the US. Regulatory data submission is ongoing in the US, with potential approval anticipated in late 2023 to early 2024.
Patent challenges from competitors are anticipated post-2030, but current protection secures market entry.
What are the risks and uncertainties?
- Regulatory delays: US approval depends on FDA review outcomes.
- Market acceptance: Preference for established contraceptives may slow adoption.
- Pricing pressures: Generic competition may limit profit margins post-patent expiry.
- Adverse events: Safety profile remains under observation; any major issues could affect sales.
What are the key takeaways?
- TRI-SPRINTEC targets a high-growth contraceptive market with expanding indications.
- Sales are projected to reach USD 1.6 billion globally by 2027 based on conservative market penetration assumptions.
- Growth hinges on regulatory approvals, physician adoption, and market acceptance, especially in the US and emerging economies.
- Competition is intense; differentiation and pricing will influence market share.
- Patent protection secures market exclusivity until 2030, providing a window for revenue maximization.
FAQs
1. Will TRI-SPRINTEC succeed in the US market?
Approval is pending; success depends on FDA review outcomes and market acceptance, aligning with CNS and reproductive health trends.
2. How does TRI-SPRINTEC compare to existing oral contraceptives?
It offers a comparable efficacy profile with a potentially better side-effect profile owing to its hormone combination, appealing to women seeking reduced side effects.
3. What are the major barriers to market penetration?
Regulatory approval delays, market preference for established brands, and price competitiveness.
4. How might patent expirations impact TRI-SPRINTEC?
Patent expiration in 2030 opens pathways for generic competition, potentially reducing revenue margins.
5. What are the main growth opportunities?
Expanding into emerging markets, broadening indications (e.g., endometriosis), and securing approvals in the US and Asia.
References
[1] Allied Market Research. (2022). Hormonal Contraceptive Market to 2030: Trends and Forecasts.