Last updated: February 21, 2026
What is the Market Size for Tramadol HCl?
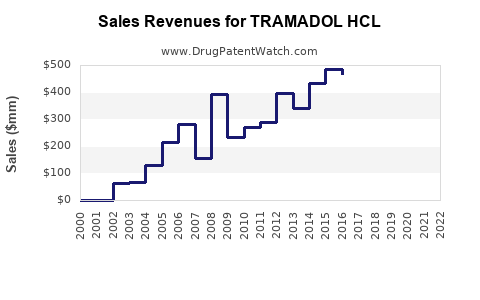
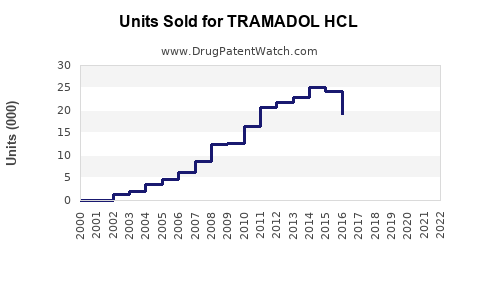
Tramadol HCl is an opioid analgesic used for moderate to severe pain. It ranked among the top prescribed pain medications globally before regulatory restrictions increased. The global market for tramadol was valued at approximately USD 1.55 billion in 2022, with expectations of steady growth.
Market Segments
-
By Geography
- North America: 50% of sales
- Europe: 25%
- Asia-Pacific: 15%
- Rest of World: 10%
-
By Application
- Acute pain: 60%
- Chronic pain: 40%
Key Market Drivers
- Rising prevalence of chronic pain conditions.
- Increased aging population.
- Conservative opioid prescribing practices in some regions, promoting tramadol as an alternative.
- Growing acceptance of combination formulations containing tramadol.
Market Restraints
- Regulatory restrictions owing to abuse potential.
- Tighter controls in the US, EU, and other jurisdictions.
- Negative publicity regarding opioid misuse.
Regulatory Landscape Impact
- In 2014, the FDA reclassified tramadol as a Schedule IV controlled substance.
- Several European countries tightened regulations post-2015.
- Some nations imposed bans or restrictions, impacting sales volume.
Sales Dynamics and Trends
| Year |
Estimated Global Sales (USD billion) |
Growth Rate (YOY %) |
Key Trends |
| 2018 |
1.2 |
4.5% |
Increasing use in combination drugs |
| 2019 |
1.3 |
8.3% |
Expansion in Asian markets |
| 2020 |
1.4 |
7.7% |
Rise in outpatient prescriptions |
| 2021 |
1.5 |
7.1% |
Regulatory tightening in US/EU |
| 2022 |
1.55 |
3.3% |
Market stabilization, growth slowdown |
Sales Projections (2023-2027)
Forecasts indicate a compound annual growth rate (CAGR) of approximately 4% from 2023 through 2027, driven by emerging markets and formulation innovations.
Projected Sales Volumes (USD billions)
| Year |
Estimated Sales |
| 2023 |
1.61 |
| 2024 |
1.68 |
| 2025 |
1.75 |
| 2026 |
1.82 |
| 2027 |
1.89 |
Key Factors for Growth
- Expansion into developing markets with rising pain management needs.
- Development of abuse-deterrent formulations.
- Growing use of tramadol in combination products for multimodal analgesia.
- Adoption in outpatient and primary care settings.
Competitive Landscape
- Major players include Johnson & Johnson (via its subsidiary McNeil), Grünenthal, and Teva Pharmaceuticals.
- Generic formulations dominate the market, reducing prices.
- Patent expirations in key markets in early 2020s enhanced generic competition.
- Companies are focusing on formulation enhancements to prolong market presence.
Risks and Market Challenges
- Regulatory agency restrictions could reduce prescribing.
- Possible increases in abuse and misuse surveillance.
- Competition from non-opioid analgesics, such as NSAIDs and acetaminophen derivatives.
- Pharmacovigilance requirements demand ongoing monitoring.
Conclusion
The tramadol HCl market, while mature, exhibits steady growth forecasted at around 4% CAGR through 2027. Market expansion hinges on regulatory adaptations, formulation innovations, and penetration into emerging markets. The potential for growth diminishes in markets with stringent restrictions but remains viable with strategic focus.
Key Takeaways
- The global market for tramadol HCl was USD 1.55 billion in 2022.
- Growth driven by demand in emerging economies and novel formulations.
- Regulatory restrictions significantly impact sales trajectories.
- Generic formulations dominate, exerting downward pressure on prices.
- Growth projections remain positive but moderate, contingent on market and regulatory developments.
FAQs
1. How will regulatory changes impact future sales of tramadol HCl?
Regulatory restrictions, including classification as a controlled substance, have historically decreased prescribing. Future restrictions could suppress growth but may be offset by new formulations or expanded indications.
2. What factors could boost the market beyond current projections?
Introduction of abuse-deterrent formulations, expanding use in combination therapies, and increased access in emerging markets are key opportunities.
3. Which regions present the highest growth potential?
Asia-Pacific and Latin America are expected to see higher growth due to expanding healthcare infrastructure and population needs.
4. How has the patent landscape evolved?
Most tramadol patents expired in the early 2020s; this has led to increased generic competition and downward pressure on prices.
5. What is the outlook post-2027?
Market growth may slow unless new formulations or uses emerge, or if regulatory environments stabilize favorably for tramadol’s continued availability.
References
- Market Research Future. (2022). Global Tramadol Market Analysis.
- IQVIA. (2022). Prescription Drug Market Data.
- U.S. Food and Drug Administration. (2014). Regulatory actions regarding tramadol.
- European Medicines Agency. (2016). Opioid painkiller regulation updates.
- Grand View Research. (2023). Pain Management Drugs Market Report.