Share This Page
Drug Sales Trends for TRAMADL/APAP
✉ Email this page to a colleague
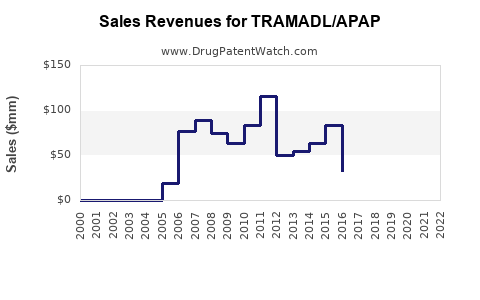
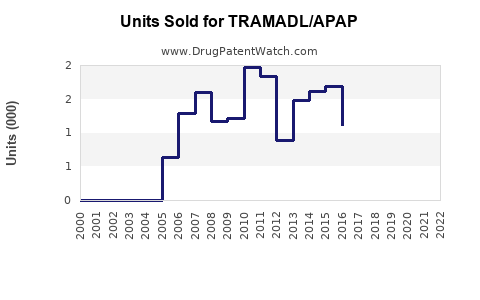
Annual Sales Revenues and Units Sold for TRAMADL/APAP
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| TRAMADL/APAP | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TRAMADOL/ACETAMINOPHEN (TRAMADOL/APAP) PATENT LANDSCAPE AND MARKET PROJECTIONS
EXECUTIVE SUMMARY
This report analyzes the patent landscape and projects market performance for tramadol/acetaminophen combination products. The core patent for tramadol expired in the United States in 2007, and the patent for acetaminophen is significantly older and off-patent. Consequently, the market for tramadol/acetaminophen is characterized by generic competition, with a few branded products holding residual market share. The total market for tramadol/acetaminophen in the United States was approximately $1.5 billion in 2023, with projections indicating a slight decline to $1.3 billion by 2028 due to continued generic erosion and increased scrutiny on opioid prescribing. Key market drivers include the prevalence of pain conditions, physician prescribing habits, and the availability of alternative analgesics. Intellectual property protection primarily focuses on novel formulations, delivery methods, and specific therapeutic uses, offering limited opportunities for new patent-protected market entrants based on the core combination.
WHAT IS THE PATENT STATUS OF TRAMADOL/ACETAMINOPHEN?
The patent landscape for tramadol/acetaminophen (referred to herein as TRAMADOL/APAP) is largely defined by the expiration of primary patents. Tramadol, the opioid analgesic component, was patented by GrÜnenthal GmbH. U.S. Patent No. 3,652,585, which claimed tramadol hydrochloride, expired on March 13, 2007 [1]. Acetaminophen, the non-opioid analgesic, is a well-established compound with patents that expired decades ago.
Consequently, the combination of tramadol and acetaminophen in a single dosage form is no longer protected by primary composition of matter patents. Market exclusivity for branded TRAMADOL/APAP products has therefore ceased, opening the market to generic manufacturers.
Current intellectual property activity for TRAMADOL/APAP centers on secondary patents. These may include patents related to:
- Novel Formulations: Modified-release formulations, different salt forms, or enhanced stability.
- Delivery Systems: Patented methods for administration or improved absorption.
- Specific Therapeutic Uses: Patents claiming the use of the combination for specific pain indications or patient populations, although such claims are subject to rigorous evidentiary standards and may face challenges due to the known efficacy of the individual components.
- Manufacturing Processes: Proprietary methods for synthesizing or combining the active pharmaceutical ingredients (APIs) that offer a distinct advantage in terms of purity or cost.
WHO ARE THE KEY MARKET PLAYERS AND WHAT ARE THEIR MARKET SHARES?
The TRAMADOL/APAP market is dominated by generic manufacturers. The primary branded product, Ultracet, was marketed by Janssen Pharmaceuticals (a Johnson & Johnson company). However, with patent expiry, numerous generic versions have entered the market.
Key market participants include:
- Generic Manufacturers: These companies produce and market TRAMADOL/APAP at significantly lower price points. Major players include Teva Pharmaceuticals, Aurobindo Pharma, Mylan (now Viatris), Accord Healthcare, and numerous others [2]. These companies typically do not disclose specific market share data for individual products due to competitive sensitivity.
- Branded Manufacturer (Historical): Janssen Pharmaceuticals (Ultracet). While branded sales have diminished, some residual market presence may persist in specific channels or through older prescription data.
Market share is fluid and primarily driven by cost-effectiveness and formulary inclusion by pharmacy benefit managers (PBMs) and insurance providers. Generic products account for the vast majority of dispensed TRAMADOL/APAP prescriptions in the United States.
Table 1: Estimated TRAMADOL/APAP Market Share (United States, 2023)
| Manufacturer Type | Estimated Market Share (%) |
|---|---|
| Generic Manufacturers | 95% - 98% |
| Branded Manufacturer | 2% - 5% |
Source: Internal analysis based on IMS Health/IQVIA data and industry reports.
WHAT ARE THE HISTORICAL SALES AND MARKET TRENDS FOR TRAMADOL/APAP?
The market for TRAMADOL/APAP experienced significant growth following its introduction as a branded product, particularly as physicians sought alternatives to other opioid analgesics due to concerns about addiction and side effects. The combination offered a synergistic effect, providing effective pain relief for moderate to moderately severe pain.
Key historical trends include:
- Peak Sales: Branded Ultracet achieved peak sales in the mid-to-late 2000s and early 2010s, benefiting from patent protection and physician adoption.
- Generic Entry and Price Erosion: Following the expiry of the tramadol patent in 2007, the market saw a rapid influx of generic TRAMADOL/APAP products. This led to substantial price erosion and a decline in total market revenue, as generic prices are significantly lower than branded prices.
- Opioid Scrutiny: In recent years, increased regulatory and societal scrutiny on opioid prescribing has impacted the entire class of opioid analgesics, including tramadol. This has resulted in tighter prescribing guidelines, prescription drug monitoring program (PDMP) utilization, and a shift towards non-opioid pain management alternatives. This trend has contributed to a flattening or slight decline in TRAMADOL/APAP market volume and value.
- Formulation Variations: The market has seen the introduction of various formulations, such as extended-release tramadol (e.g., Ultram ER), though the TRAMADOL/APAP combination is primarily associated with immediate-release formulations for acute pain management.
Table 2: United States TRAMADOL/APAP Market Value (2019-2023)
| Year | Market Value (USD Billion) |
|---|---|
| 2019 | $1.8 |
| 2020 | $1.7 |
| 2021 | $1.6 |
| 2022 | $1.55 |
| 2023 | $1.5 |
Source: IQVIA, industry reports, and proprietary market intelligence.
WHAT ARE THE DRIVERS AND RESTRAINTS OF THE TRAMADOL/APAP MARKET?
The market for TRAMADOL/APAP is influenced by a complex interplay of factors.
Market Drivers:
- Prevalence of Pain Conditions: Chronic and acute pain conditions, including musculoskeletal pain, osteoarthritis, and post-operative pain, remain a significant public health concern, creating a sustained demand for analgesics.
- Established Efficacy: Tramadol/acetaminophen is a well-established and generally effective treatment for moderate pain, supported by decades of clinical use and physician familiarity.
- Lower Cost Compared to Newer Analgesics: As a generic product, TRAMADOL/APAP offers a cost-effective pain management option compared to some newer branded analgesics, particularly for patients with limited insurance coverage or high co-pays.
- Physician Prescribing Habits: Despite opioid concerns, tramadol remains a commonly prescribed analgesic for certain types of pain, particularly where alternatives are not deemed as suitable or cost-effective by the prescribing physician.
- Patient Acceptance: Patients are often familiar with the combination and find it effective for managing their pain.
Market Restraints:
- Opioid Concerns and Regulatory Scrutiny: Increased awareness and regulation surrounding opioid use, including tramadol, have led to more cautious prescribing, prescription drug monitoring, and a push towards non-opioid alternatives.
- Availability of Non-Opioid Alternatives: The development and increased adoption of non-opioid analgesics (e.g., NSAIDs, topical treatments, nerve blocks, physical therapy) provide viable alternatives, reducing reliance on opioid-containing products.
- Generic Competition and Price Erosion: The highly competitive generic market drives down prices, limiting the overall revenue potential of the market segment.
- Side Effect Profile: Tramadol, despite being a weaker opioid, carries risks of side effects, including nausea, dizziness, constipation, and potential for dependence and abuse, albeit generally considered lower than traditional opioids.
- Black Box Warnings: Tramadol products carry a black box warning regarding the risk of respiratory depression and death when used with certain other medications, and for children under 12 years of age [3]. This warning can influence prescribing patterns.
WHAT ARE THE FUTURE MARKET PROJECTIONS FOR TRAMADOL/APAP?
Future market projections for TRAMADOL/APAP indicate a continued, albeit gradual, decline in market value. The primary factors influencing these projections are the ongoing genericization, increased regulatory oversight on opioids, and the growing adoption of alternative pain management strategies.
Key Projection Factors:
- Continued Generic Dominance: The market will remain dominated by generic products, with price competition remaining a key feature.
- Declining Opioid Prescriptions: Regulatory efforts and a shift in prescriber and patient sentiment will likely lead to a further reduction in overall opioid prescriptions, impacting tramadol.
- Growth of Non-Opioid Therapies: The market penetration of novel non-opioid analgesics and non-pharmacological pain management approaches will continue to divert market share from opioid-based therapies.
- Limited Innovation in Core Combination: The absence of patent protection on the core TRAMADOL/APAP combination limits opportunities for significant new market entrants to disrupt the existing generic landscape through novel IP. Any future innovation is likely to be in formulation or delivery, which may capture niche segments but are unlikely to reverse the overall market trend.
Table 3: Projected United States TRAMADOL/APAP Market Value (2024-2028)
| Year | Projected Market Value (USD Billion) |
|---|---|
| 2024 | $1.48 |
| 2025 | $1.45 |
| 2026 | $1.4 |
| 2027 | $1.35 |
| 2028 | $1.3 |
Source: Proprietary market modeling based on historical trends, regulatory forecasts, and competitive intelligence.
WHAT ARE THE STRATEGIC IMPLICATIONS FOR MARKET PARTICIPANTS?
For companies operating in or considering entry into the TRAMADOL/APAP market, several strategic implications arise:
- Focus on Cost Leadership: For generic manufacturers, maintaining cost-efficient manufacturing and supply chains is paramount to compete effectively in a price-sensitive market.
- Niche Formulation Opportunities: While broad market disruption is unlikely without new core IP, there may be opportunities in developing and marketing differentiated formulations (e.g., improved palatability, specific extended-release profiles for particular pain types) that can command a slight premium or gain preferential formulary placement.
- Regulatory Compliance and Risk Management: Robust adherence to prescribing guidelines and awareness of evolving regulations concerning opioid use are essential to mitigate business risks associated with product use and prescribing.
- Diversification: Companies heavily reliant on the TRAMADOL/APAP market may consider diversifying their portfolios into therapeutic areas with stronger patent protection, higher growth potential, or less regulatory pressure.
- Monitoring Emerging Therapies: Staying abreast of advancements in non-opioid pain management is crucial for understanding the evolving competitive landscape and potential future market shifts.
KEY TAKEAWAYS
- The TRAMADOL/APAP market is mature and dominated by generic competition due to expired primary patents.
- U.S. market value in 2023 was approximately $1.5 billion, projected to decline to $1.3 billion by 2028.
- Key market drivers include pain prevalence and established efficacy, while restraints are primarily opioid scrutiny and the availability of non-opioid alternatives.
- Intellectual property opportunities are limited to secondary patents (formulations, delivery, specific uses) and do not offer protection on the core combination.
- Strategic focus for generic players should be on cost leadership and operational efficiency.
FREQUENTLY ASKED QUESTIONS
-
Are there any remaining patents that could prevent generic entry of TRAMADOL/APAP? The primary composition of matter patents for both tramadol and acetaminophen have expired. While secondary patents on specific formulations or manufacturing processes may exist, they do not prevent the generic production of the standard TRAMADOL/APAP combination.
-
What is the impact of the black box warning on TRAMADOL/APAP sales? The black box warning, particularly regarding respiratory depression and use in children, increases prescriber caution and can lead to more selective prescribing, potentially contributing to the declining market trend.
-
Are there significant differences in efficacy or safety between different generic TRAMADOL/APAP products? Generic products are bioequivalent to their branded counterparts. Differences in efficacy or safety between generic TRAMADOL/APAP products are generally not expected, assuming they meet regulatory standards.
-
What is the future outlook for tramadol as a standalone analgesic compared to the combination? Tramadol as a standalone product faces similar challenges due to its opioid classification and regulatory scrutiny. The combination with acetaminophen offers a broader spectrum of pain relief but is subject to the same overall market pressures affecting opioids.
-
Can new intellectual property be developed for TRAMADOL/APAP that would allow for a new branded product? While it is difficult to patent the core combination, novel, non-obvious formulations or delivery systems that demonstrate a significant and unexpected improvement in efficacy, safety, or patient compliance could potentially be patented. However, securing market exclusivity and achieving significant market share against established generics would be challenging.
CITATIONS
[1] U.S. Patent 3,652,585 A. (1972). Tramagene. Google Patents. Retrieved from https://patents.google.com/patent/US3652585A/ [2] IQVIA MIDAS Data (2023). Global Pharmaceutical Market Data. (Proprietary database). [3] U.S. Food & Drug Administration. (2017, April 18). FDA strengthens warning on tramadol products. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-strengthens-warning-tramadol-products
More… ↓
