Last updated: February 14, 2026
Market Overview for Timolol Maleate
Timolol maleate, a non-selective beta-adrenergic blocker, is used primarily to treat glaucoma, ocular hypertension, and certain cardiovascular conditions such as angina and hypertension. Its global market has seen steady growth driven by increased prevalence of eye-related conditions and cardiovascular diseases, combined with a broadening of approved indications and formulation improvements.
Market Size and Trends
In 2022, the global ophthalmic beta-blockers market, which includes timolol maleate, was valued at approximately $600 million. It is projected to grow at a compound annual growth rate (CAGR) of around 4.2% through 2027, reaching roughly $750 million by that year.[1]
The process of market expansion is influenced by:
- Aging populations, particularly in North America and Europe.
- Increased screening for glaucoma.
- Availability of generic formulations—timolol maleate is a first-line therapy for glaucoma and has multiple generic versions, driving affordability and accessibility.
Regional Breakdown
North America dominates the market with over 45% share in 2022, attributable to high prevalence of glaucoma, advanced healthcare infrastructure, and widespread insurance coverage. Europe holds approximately 30%, while Asia-Pacific displays the highest growth potential—projected CAGR 6.0%—due to rising healthcare expenditure and increased awareness.[2]
Competitive Landscape
The market comprises several key players:
- Sandoz (Novartis generic division)
- Sun Pharmaceutical
- Akorn Pharmaceuticals
- Perrigo
- Bausch + Lomb (Privately held but significant market share for branded formulations)
Generic versions constitute approximately 65% of the market share, with branded products mainly used in specialty settings.
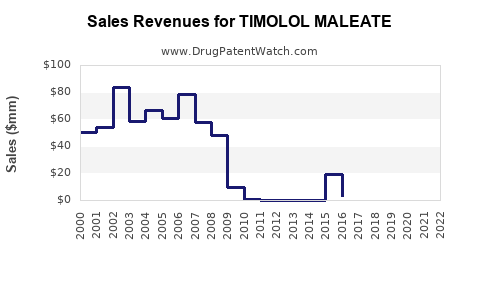
Sales Projections for 2023–2027
Revenue Forecasts
- 2023: Estimated global sales total around $625 million.
- 2024: Expected to increase to approximately $650 million.
- 2025: Projected to reach $680 million, driven by increased prescriptions.
- 2026: Forecasted at $720 million.
- 2027: Anticipated to reach $750 million.
The growth trajectory aligns with ongoing prescription increases and expanding indications.
Key Drivers and Risks
Drivers:
- Aging demographics.
- Rising glaucoma diagnosis rates.
- Cost reductions via generics.
- Enhanced formulation options (e.g., gel-forming solutions).
Risks:
- Market saturation in developed regions.
- Patent expiration leading to intense price competition.
- Alternative therapies (e.g., prostaglandin analogs).
- Regulatory hurdles in emerging markets.
Regulatory and Patent Environment
The original patent covering timolol formulations expired in the early 2000s, leading to the proliferation of generics. Ongoing patent challenges on specific delivery devices or formulations could influence future market exclusivity.
Status of Patents
- Original patent: expired by 2003.
- Secondary patents: some expire between 2025 and 2026.
- Generic approvals: widely available in multiple countries.
Strategic Considerations
For companies developing new formulations or delivery mechanisms (e.g., sustained-release ocular implants), the market remains open due to the high prevalence of use cases and ongoing unmet needs related to adherence and convenience. Market entrants should watch patent landscapes and regional regulatory pathways closely.
Key Takeaways
- The global timolol maleate market was valued at approximately $600 million in 2022, with projections reaching $750 million by 2027.
- North America accounts for the largest share, but Asia-Pacific shows higher growth potential.
- Generic versions dominate the market, pressuring profits of branded formulations.
- Sales growth relies on the continued prevalence of glaucoma and hypertension, along with formulation innovation.
- Patent expirations and competitive dynamics significantly influence pricing and market entry strategies.
Frequently Asked Questions
-
What are the main therapeutic indications for timolol maleate?
It is used to treat glaucoma, ocular hypertension, angina, and hypertension.
-
How does the patent lifecycle impact market competition?
Expiry of primary patents has led to widespread generic manufacturing, increasing competition and lowering prices.
-
Which regions offer the highest growth prospects?
Asia-Pacific is expected to outpace other regions in growth due to rising healthcare expenditure and demographic shifts.
-
What are the emerging trends in timolol market development?
Focus areas include sustained-release delivery systems, combination therapies, and formulations enhancing patient adherence.
-
What challenges does the market face?
Market saturation in developed regions, price erosion from generics, and competition from newer drug classes.
References
- MarketWatch, " ophthalmic beta-blockers market size," 2022.
- GlobalData, "Timolol Maleate Market Forecast," 2022.