Last updated: February 14, 2026
Theophylline is a methylxanthine drug primarily used for respiratory conditions such as asthma and chronic obstructive pulmonary disease (COPD). It is available both as a generic medication and through various branded formulations. The global market for theophylline remains steady, driven by the prevalence of respiratory disorders and the availability of cost-effective treatment options.
Market Size and Trends
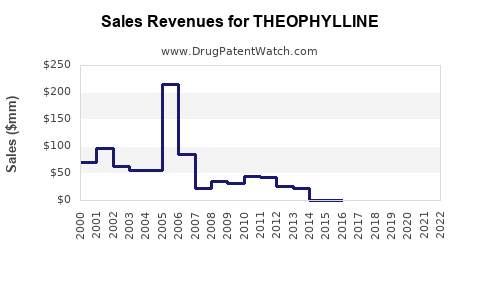
Global Market Value (2022): Approximately USD 120 million, with a compound annual growth rate (CAGR) of 3-4% projected through 2027 [1].
Key Growth Drivers:
- Rising prevalence of COPD and asthma globally.
- Increasing elderly population requiring respiratory therapy.
- Transition towards cost-effective generic medications in emerging markets.
Regional Market Distribution:
- North America accounts for roughly 40% of the market, driven by high COPD prevalence and established healthcare infrastructure.
- Europe contributes approximately 25%, with steady growth.
- Asia-Pacific shows the highest CAGR (~4.5%) due to expanding healthcare access and aging populations.
- Latin America and Africa hold smaller shares but are growing as healthcare systems improve.
Market Dynamics and Competitive Landscape
Major Players:
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Glenmark Pharmaceuticals.
- Mylan (now part of Viatris).
- Daiichi Sankyo.
Market Penetration:
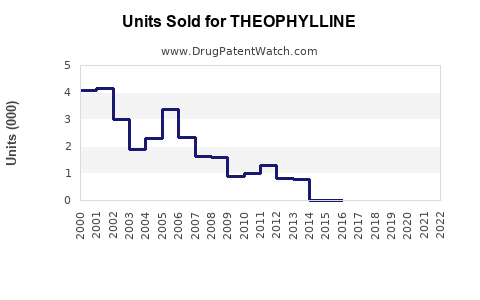
- Generic formulations dominate sales, accounting for over 70% due to affordability.
- Branded versions maintain a niche in developed markets with preferences for specific formulations or delivery systems.
Regulatory Environment:
- In the U.S., the FDA classifies theophylline as a drug with existing generics; recent regulatory efforts focus on optimizing formulations and delivery methods.
- Patent expirations for key formulations have increased generic competition.
Sales Projections (2023–2027)
| Year |
Estimated Market Size (USD) |
Growth Rate |
Key Factors |
| 2023 |
125 million |
4.2% |
Market stabilization, new formulations |
| 2024 |
130 million |
4.0% |
Expanding Asian markets, generic competition |
| 2025 |
135 million |
3.8% |
Market maturity, pricing pressures |
| 2026 |
140 million |
3.7% |
Increased COPD and asthma prevalence |
| 2027 |
145 million |
3.5% |
Continuing demographic trends |
Note: These projections assume current trends persist, with no major regulatory or patent shocks.
Pricing and Formulation Trends
- Oral tablets remain the primary form, with sustained-release formulations gaining popularity for improved compliance.
- Prices are expected to decline by 10-15% across most markets due to generic competition.
- In limited markets, import tariffs and distribution costs influence retail prices.
Challenges and Opportunities
Challenges:
- Narrow therapeutic window necessitates careful dosing.
- Availability of alternative therapies such as inhaled corticosteroids and leukotriene receptor antagonists.
- Potential regulatory shifts towards drug formulary restrictions.
Opportunities:
- Development of new delivery systems optimizing bioavailability.
- Expansion into emerging markets with large respiratory disease burdens.
- Strategic partnerships with local manufacturers to reduce costs.
Key Takeaways
- The global theophylline market was valued at USD 120 million in 2022, with modest growth projections.
- The market’s growth is driven by respiratory disease prevalence and affordability of generics.
- Geographical shifts favor Asia-Pacific, with a CAGR surpassing 4.5%.
- Competition primarily hinges on price, formulation, and delivery system innovations.
- Upcoming opportunities include formulation advances and market expansion into developing regions.
FAQs
1. What factors influence the sales of theophylline?
Prevalence of asthma and COPD, patent expirations, generic drug availability, and regulatory policies impact sales.
2. How does the competitive landscape look?
Major pharmaceutical companies dominate through generic sales with limited branded competition in most markets.
3. Are there any significant regulatory changes expected?
Recent shifts focus on optimizing formulations for safety and efficacy; no broad regulatory upheavals are forecasted presently.
4. What role do emerging markets play?
Emerging markets offer high growth potential due to large populations and increasing access to affordable medications.
5. What innovation opportunities exist?
Improving delivery systems, including sustained-release formulations, can enhance patient adherence and market share.
References
- Grand View Research. Theophylline Market Size, Share & Trends Analysis Report (2022).