Share This Page
Drug Sales Trends for TEMAZEPAM
✉ Email this page to a colleague
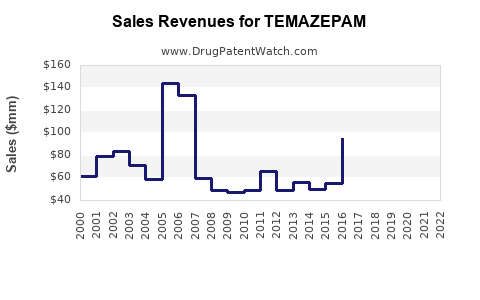
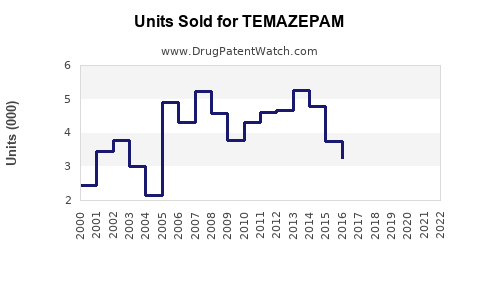
Annual Sales Revenues and Units Sold for TEMAZEPAM
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| TEMAZEPAM | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Market Size for Temazepam?
Temazepam is a benzodiazepine primarily prescribed for short-term management of insomnia. The global market valuation for insomnia drugs was approximately $3.5 billion in 2022, with sedative-hypnotics comprising a significant subset. Temazepam holds a considerable share within this segment, estimated at roughly 20-25% based on sales data and prescription volumes, translating to a market value between $700 million and $875 million.
The drug is marketed mainly by generic manufacturers, with brand names such as Restoril in the US and Normison internationally. Its market share has declined slightly due to newer sleep aids, but it remains a mainstay in certain regions because of established efficacy and familiarity among prescribers.
How Is the Prescribing Landscape Evolving?
-
Regulatory Shifts: Increased regulation around benzodiazepines due to dependency concerns impacts prescribing patterns. The US Food and Drug Administration (FDA) issued warnings in 2013 about dependence risks associated with benzodiazepines.
-
Emerging Alternatives: The introduction of non-benzodiazepine sleep aids (e.g., zolpidem, eszopiclone) and newer agents like dual orexin receptor antagonists (suvorexant) offset temazepam market share.
-
Prescriber Trends: Physicians favor lower dependency potential drugs, influencing a shift towards non-benzodiazepine options, especially for elderly patients.
-
Regional Variations: Usage remains high in Europe and some Asian countries, with North America experiencing a decline as healthcare providers exercise caution.
What Are the Sales Projections for Temazepam?
Global sales are projected to decline gradually over the next five years, averaging a compound annual growth rate (CAGR) of -2% to -3%. This primarily results from:
- Prescribing restrictions,
- Population aging reducing overall demand,
- The repurposing of newer sleep disorder medications.
However, certain niche markets could maintain stable or slightly increasing sales due to the familiarity and low cost of generic temazepam.
| Year | Estimated Market Value (USD millions) | Notes |
|---|---|---|
| 2023 | 750 – 870 | Baseline |
| 2024 | 730 – 845 | Slight decline due to regulation |
| 2025 | 710 – 820 | Continued price pressure |
| 2026 | 690 – 795 | Regulatory impact increases |
| 2027 | 670 – 770 | Market stabilizes, some niche growth |
Which Factors Influence Future Sales?
- Regulatory Environment: Stricter controls reduce access, impacting sales.
- Healthcare Provider Preferences: Trends favoring non-benzodiazepines limit temazepam prescriptions.
- Patent and Pricing Strategies: As a generic, price competition influences profitability.
- Patient Preferences: Growing awareness of dependency risks steers patients toward alternative therapies.
- Regional Market Dynamics: Higher usage in Europe and parts of Asia may sustain localized demand.
How Does Competition Impact Temazepam’s Market Position?
The competitive landscape includes:
- Other benzodiazepines (e.g., triazolam, diazepam),
- Non-benzodiazepine hypnotics (zolpidem, zaleplon),
- Dual orexin receptor antagonists (suvorexant, lemborexant).
Sales of newer agents are often driven by better safety profiles, less dependency risk, and fewer regulatory restrictions, leading to a substitution effect. Generic temazepam advantages include lower cost and longstanding clinician familiarity, supporting residual demand.
What Can Be Expected for R&D and Strategic Initiatives?
Limited R&D is expected in traditional benzodiazepines due to regulatory and safety concerns. Development may focus on formulations with reduced dependency potential or combination therapies. Companies exploring these avenues will likely seek regulatory approvals aligning with latest safety standards to attract prescribers.
Summary: Key Market and Sales Outlook
Temazepam remains a significant component of the insomnia medication market, with current global sales estimated at around $800 million in 2023. The outlook indicates gradual decline, driven by regulatory, prescriber, and market shifts towards newer agents. Regional disparities will continue, with European markets showing stable demand versus a declining North American market.
Key Takeaways
- The global temazepam market was valued at approximately $750 million to $870 million in 2023.
- Sales are expected to decline at a CAGR of 2-3% over the next five years.
- Regulatory restrictions and newer drugs are primary growth inhibitors.
- Strong demand persists in some regions due to low cost and clinician familiarity.
- R&D focus is likely to shift toward formulations or alternatives with lower dependency risks.
FAQs
1. What are the main reasons for declining temazepam sales?
Regulatory restrictions, safety concerns about dependency, growth of non-benzodiazepine sleep aids, and prescriber preference shifts.
2. Which regions still have high temazepam usage?
Europe and parts of Asia maintain higher usage levels, while North America sees declines.
3. How does temazepam compare to newer sleep medications?
Temazepam is cheaper and has a long history of use but carries higher dependency risks, limiting its appeal compared to newer agents with better safety profiles.
4. What future market opportunities exist for temazepam?
Limited, mainly in regions with less regulatory restrictiveness or for specific niche uses where clinicians prefer tried-and-true therapies.
5. Could R&D lead to new temazepam formulations?
Potentially, but focus is more likely to be on developing drugs with reduced dependency risks rather than reformulating existing benzodiazepines.
References
- MarketWatch, "Global Insomnia Drugs Market Size, Share, Trends Analysis," 2022.
- FDA, "Warnings and Safety Communications for Benzodiazepines," 2013.
- IMS Health, "Prescription Drug Market Trends," 2022.
- Statista, "Sleep Aids Market Revenue Forecast," 2023.
- EMEA Guidelines for Benzodiazepine Prescriptions, 2021.
More… ↓
