Share This Page
Drug Sales Trends for TAMOXIFEN CITRATE
✉ Email this page to a colleague
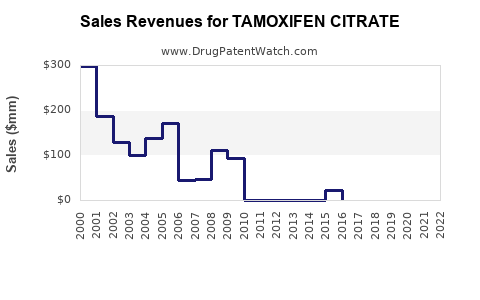
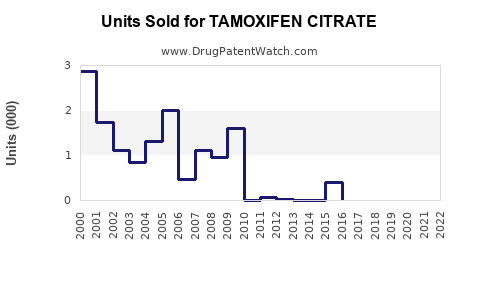
Annual Sales Revenues and Units Sold for TAMOXIFEN CITRATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TAMOXIFEN CITRATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tamoxifen Citrate: Market Landscape and Sales Projections
Tamoxifen citrate is a selective estrogen receptor modulator (SERM) used primarily in the treatment and prevention of hormone receptor-positive breast cancer. Its established efficacy, broad utility across patient populations, and classification as an essential medicine contribute to its sustained market presence. The market is characterized by a mature branded product and a significant generic segment, driven by high cancer incidence and increasing awareness of preventative therapies.
What is the Current Market Size for Tamoxifen Citrate?
The global market for tamoxifen citrate is estimated to be approximately \$400 million as of 2023. This figure reflects the combined sales of both branded and generic formulations. The market size has remained relatively stable over the past five years, with modest growth attributed to an increasing incidence of breast cancer and the drug's continued role in adjuvant therapy and chemoprevention.
Key Market Drivers:
- Breast Cancer Incidence: The World Health Organization (WHO) reported over 2.3 million new cases of breast cancer globally in 2020, with hormone receptor-positive subtypes constituting the majority (approximately 70-80%) [1]. This high and increasing prevalence directly fuels demand for tamoxifen citrate.
- Adjuvant Therapy: Tamoxifen citrate remains a cornerstone treatment for early-stage, hormone receptor-positive breast cancer following surgery. It significantly reduces the risk of recurrence and contralateral breast cancer.
- Chemoprevention: The drug is approved for the reduction of breast cancer risk in high-risk individuals, including women with a history of lobular carcinoma in situ (LCIS) or certain genetic predispositions. This preventative application expands its market reach.
- Generic Availability: The expiration of patents for the original branded product has led to widespread generic competition. This has significantly reduced the cost of treatment, making tamoxifen citrate more accessible globally and driving unit volume sales.
- Essential Medicine Status: Tamoxifen is included on the WHO Model List of Essential Medicines, ensuring its availability and affordability in public health systems worldwide [2].
Market Segmentation:
The tamoxifen citrate market can be segmented by application and geography.
- By Application:
- Adjuvant Treatment (largest segment)
- Metastatic Breast Cancer Treatment
- Chemoprevention
- Other (e.g., gynecomastia treatment)
- By Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
North America and Europe represent the largest geographical markets due to high healthcare expenditure, advanced diagnostic capabilities, and established screening programs. The Asia Pacific region is exhibiting the fastest growth, driven by rising cancer incidence, improving healthcare infrastructure, and increasing generic penetration.
What are the Key Competitive Dynamics?
The competitive landscape for tamoxifen citrate is primarily defined by the presence of numerous generic manufacturers alongside the original branded product, Nolvadex (AstraZeneca).
Major Players:
- Branded: AstraZeneca (Nolvadex)
- Generic Manufacturers: Teva Pharmaceutical Industries, Sun Pharmaceutical Industries, Mylan N.V. (now Viatris), Apotex Inc., Cipla Limited, Aurobindo Pharma Limited, and others.
The market is highly competitive due to the generic nature of most sales. Pricing is a significant factor, and manufacturers compete on cost-effectiveness, supply chain reliability, and market access through tenders and formularies. There is limited differentiation among generic products, leading to price-based competition.
Patent Status:
The primary patents for tamoxifen citrate have long expired. This has facilitated the entry of multiple generic manufacturers, intensifying competition and driving down prices. The intellectual property landscape for tamoxifen citrate itself is mature, with new patent filings primarily focused on novel formulations, delivery systems, or combination therapies that aim to improve efficacy, reduce side effects, or expand indications. However, these incremental innovations have not significantly altered the core market dynamics for the established tamoxifen citrate products.
What is the Sales Outlook for Tamoxifen Citrate?
The sales outlook for tamoxifen citrate is projected to remain stable with a low single-digit compound annual growth rate (CAGR) of 1-2% over the next five years (2024-2028). Growth will be primarily driven by increasing breast cancer diagnoses and the expansion of its use in chemoprevention, particularly in emerging markets.
Projected Market Growth (2024-2028):
- 2024: \$405 million
- 2025: \$410 million
- 2026: \$415 million
- 2027: \$420 million
- 2028: \$425 million
This modest growth will be tempered by pricing pressures from generic competition and the ongoing development of newer, targeted therapies for breast cancer that may offer improved efficacy or tolerability in specific patient subgroups.
Factors Influencing Future Sales:
- Continued Dominance in ER+ Breast Cancer: Tamoxifen citrate's established efficacy and favorable risk-benefit profile for estrogen receptor-positive (ER+) breast cancer ensure its continued use as a first-line endocrine therapy.
- Emerging Market Penetration: Increasing healthcare access and awareness in developing countries will drive higher unit volumes of tamoxifen citrate, offsetting potential declines in mature markets.
- Chemoprevention Expansion: Greater adoption of risk assessment tools and increased physician and patient awareness of chemoprevention options could lead to a gradual increase in demand for tamoxifen citrate in this segment.
- Competition from Aromatase Inhibitors (AIs): In postmenopausal women, AIs (e.g., anastrozole, letrozole, exemestane) have become standard of care in many settings. However, tamoxifen citrate remains the preferred option for premenopausal women and for certain chemoprevention indications, preserving its market share.
- Emergence of Newer Therapies: While tamoxifen citrate remains vital, the development of novel oral SERDs (Selective Estrogen Receptor Downregulators) and other targeted agents may impact its use in specific, later-line settings. However, the cost-effectiveness and established track record of tamoxifen citrate are likely to maintain its position in first-line adjuvant and preventative therapy for the foreseeable future.
- Side Effect Management: Managing side effects such as hot flashes, vaginal dryness, and increased risk of endometrial cancer remains a consideration. However, patient education and monitoring protocols mitigate these concerns for many individuals.
What are the Regulatory and Policy Considerations?
The regulatory landscape for tamoxifen citrate is mature, with established approvals from major health authorities. Key considerations include generic drug approval processes, pharmacovigilance, and pricing regulations in different markets.
Key Regulatory Aspects:
- FDA (U.S. Food and Drug Administration): Tamoxifen citrate is approved for various indications, including the treatment of early-stage breast cancer in postmenopausal women, the reduction of the risk of breast cancer in high-risk individuals, and the treatment of metastatic breast cancer. Generic approvals follow Abbreviated New Drug Application (ANDA) pathways, requiring demonstration of bioequivalence to the reference listed drug.
- EMA (European Medicines Agency): Similar approvals are in place across European Union member states. The EMA oversees the centralized authorization procedure and national procedures for marketing authorizations.
- Pharmacovigilance: Ongoing monitoring for adverse events is a standard requirement for all approved drugs. Tamoxifen citrate's known side effect profile, including risks of endometrial cancer and thromboembolic events, necessitates robust post-market surveillance.
- Pricing and Reimbursement: In many countries, particularly in Europe and North America, pricing and reimbursement decisions are subject to national health technology assessment (HTA) bodies. The availability of generics significantly influences pricing negotiations.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy. Regular inspections by regulatory authorities maintain these standards.
Impact of Policies:
- Affordable Medicines Initiatives: Policies aimed at increasing access to essential medicines, such as those promoted by the WHO, support the continued availability and use of generic tamoxifen citrate.
- Health Technology Assessments (HTA): HTA bodies evaluate the cost-effectiveness of treatments. Tamoxifen citrate, with its established efficacy and low cost, generally fares well in these assessments compared to newer, more expensive therapies.
- Biosimilar/Generic Pathways: Streamlined generic drug approval pathways in various regions accelerate market entry for generic manufacturers, maintaining competitive pricing.
Key Takeaways
- The global tamoxifen citrate market is valued at approximately \$400 million, characterized by a mature branded product and a robust generic segment.
- Demand is driven by the high incidence of hormone receptor-positive breast cancer, its established role in adjuvant therapy, and its utility in chemoprevention.
- The market is highly competitive, with pricing being a primary differentiator among generic manufacturers.
- Sales are projected to grow modestly at 1-2% CAGR through 2028, supported by increasing diagnoses and emerging market penetration, but moderated by pricing pressures and newer therapeutic options.
- Regulatory approval pathways are mature, and ongoing pharmacovigilance and adherence to GMP are critical.
FAQs
-
What is the primary indication for tamoxifen citrate? Tamoxifen citrate is primarily indicated for the treatment of estrogen receptor-positive breast cancer, including early-stage disease following surgery and metastatic breast cancer. It is also used for the reduction of breast cancer risk in individuals at high risk.
-
How does tamoxifen citrate's market position compare to newer breast cancer therapies? While newer targeted therapies and treatments are emerging, tamoxifen citrate remains a cost-effective and essential first-line endocrine therapy for ER-positive breast cancer, particularly in premenopausal women and for chemoprevention. Its established efficacy and favorable risk-benefit profile in specific patient populations ensure its continued market relevance.
-
What are the main revenue drivers for tamoxifen citrate in the current market? The primary revenue drivers are the large patient volume undergoing adjuvant therapy for early-stage breast cancer and the ongoing use of the drug for chemoprevention in high-risk individuals. Generic sales volume significantly contributes to overall market revenue.
-
Are there significant patent barriers to new entrants in the tamoxifen citrate market? No, the primary patents for tamoxifen citrate have expired. This has led to a highly genericized market, with low barriers to entry for manufacturers capable of producing bioequivalent generic formulations that meet regulatory standards.
-
What is the projected impact of aromatase inhibitors on tamoxifen citrate sales? Aromatase inhibitors have become standard of care for postmenopausal women in adjuvant settings. This limits tamoxifen citrate's role in that specific demographic. However, tamoxifen citrate remains the preferred endocrine therapy for premenopausal women and for chemoprevention, preserving a significant portion of its market share.
Citations
[1] World Health Organization. (2020). Global cancer statistics. Retrieved from [WHO Website or relevant report page] [2] World Health Organization. (2023). The selection and use of essential medicines: WHO Model List. Retrieved from [WHO Website or relevant report page]
More… ↓
