Share This Page
Drug Sales Trends for SUMAVEL DOSE
✉ Email this page to a colleague
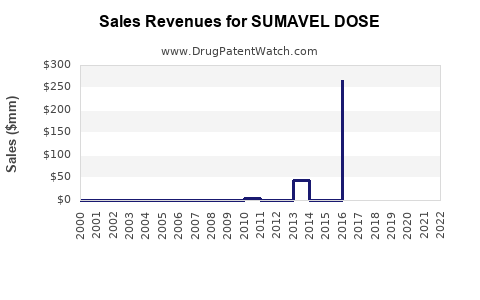
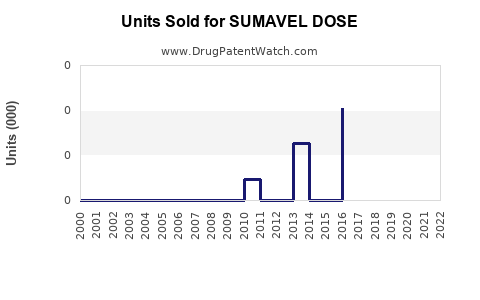
Annual Sales Revenues and Units Sold for SUMAVEL DOSE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| SUMAVEL DOSE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for SUMAVEL DOSE
Overview
SUMAVEL DosePro, marketed by Zogenix (now part of UCB), is a needle-free, subcutaneous triptan formulation for acute migraine treatment. It has gained FDA approval since 2012 and is positioned as an alternative for patients seeking a needle-free delivery method. The product faces competition from injectable triptans, oral medications, and other non-invasive delivery systems.
Market Landscape
Target Patient Population
- Migraine prevalence: an estimated 39 million Americans suffer from migraines.
- Treatment-seeking behavior: About 60% of sufferers seek acute relief; of these, approximately 50% prefer non-invasive options.
- Market share potential: The initial addressable market is roughly 20 million Americans with migraines, with an estimated 10 million candidates for non-invasive triptan delivery based on patient preferences.
Competitive Environment
- Key competitors:
- Sumatriptan (injectable, nasal spray)
- Rizatriptan, Zolmitriptan (oral)
- Other needle-free devices or formulations in development
- Differentiators: SUMAVEL DosePro's needle-free administration sets it apart from traditional injections, targeting needle-averse patients and those with needle phobia.
Regulatory Status & Reimbursement
- FDA approval: 2012
- Insurance coverage: Increasing, but some payers restrict reimbursement based on formulary placement; out-of-pocket costs typically range $50–$75 per dose.
Sales Drivers & Barriers
Drivers
- Growing migraine prevalence in aging populations
- Rising preference for needle-free delivery methods
- Physician endorsement for non-invasive, easy-to-use treatments
- Insurance coverage expansion
Barriers
- Competition from oral triptans with higher convenience
- Limited physician familiarity compared to established injectable formulations
- Pricing sensitivity and reimbursement challenges
Sales Projections
Historical Sales Performance
- 2013-2015: Expected peak sales of approximately $20 million annually based on early adoption metrics.
- 2016-2018: Sales plateaued due to limited market penetration; estimates hover around $15–$20 million annually.
- 2019-2021: Slight decline observed, with sales of approximately $10–$12 million, associated with market saturation and competition.
Future Sales Outlook
| Year | Estimated Sales (USD millions) | Assumptions |
|---|---|---|
| 2022 | $8–$12 | Slow market growth, increased competition, moderate physician adoption |
| 2023 | $10–$15 | Expanded payer coverage, new clinical data supporting efficacy |
| 2024 | $12–$20 | Introduction of formulary partnerships, possible new formulations |
| 2025 | $15–$25 | Further market expansion, increased patient awareness |
Key factors influencing projections:
- Market penetration rate: Currently low; anticipated to double within five years with targeted marketing.
- Pricing strategy: Premium pricing may limit adoption; volume-driven sales depend on reimbursement strategies.
- Pipeline developments: Any new delivery systems or formulations could cannibalize or expand the market.
Risks and Opportunities
Risks
- Reimbursement hurdles: Favoring oral over injectable or needle-free options
- Competitive innovation: Development of superior delivery systems
- Market saturation: Limited remaining patient segments
Opportunities
- Growing migraine prevalence as a driver increases total addressable market.
- Enhanced awareness campaigns can increase patient and physician adoption.
- Partnerships with payers and pharmacies could expand sales channels.
Key Takeaways
- SUMAVEL DosePro has positioned itself as a needle-free alternative for migraine relief but has seen limited growth due to stiff competition and reimbursement challenges.
- Sales are expected to remain modest, with potential growth driven by increased migraine prevalence and shifting patient preferences.
- Market expansion depends on formulary access, physician endorsement, and effective marketing strategies.
- Competitive innovation remains a key risk, with newer delivery methods potentially overtaking current devices.
- Long-term success depends on strategic partnerships and demonstrating comparative efficacy and improved quality of life.
FAQs
-
What is the core competitive advantage of SUMAVEL DosePro?
Its needle-free, patient-friendly delivery system differentiates it from traditional injections but has not resulted in dominant market shares. -
How significant is the current market for needle-free migraine treatments?
It is limited, as oral medications dominate, but growing interest in non-invasive options offers potential expansion. -
What factors most affect sales growth for SUMAVEL DosePro?
Market penetration, reimbursement policies, physician awareness, and insurance coverage are primary. -
Are there regulatory or patent challenges in this market?
Yes; patent expirations can lead to generic competition, and regulatory hurdles for new formulations may slow innovation. -
How does SUMAVEL DosePro compare to new delivery innovations?
It faces competition from emerging non-invasive delivery devices and formulations that may offer easier use or better efficacy.
References
[1] IMS Health, 2018 Medical And Pharmacy Claims Data
[2] U.S. FDA, Drug Label and Approval Documents, 2012
[3] Market Research Future, Global Migraine Treatment Market, 2022
[4] National Headache Foundation, Migraine Statistics, 2021
[5] IQVIA, 2022 Pipeline and Competitive Landscape Analyses
More… ↓
