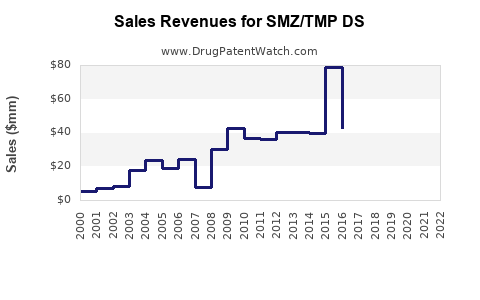
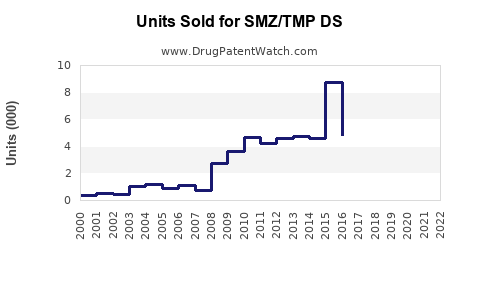
Last updated: February 15, 2026
What is SMZ/TMP DS?
SMZ/TMP DS (sulfamethoxazole and trimethoprim, double strength) is an antibiotic combination used to treat bacterial infections such as urinary tract infections, respiratory infections, and typhoid. It is marketed primarily as a prescription medication with established therapeutic use.
What is the size of the current market for SMZ/TMP DS?
The global antibiotics market, valued at approximately $56 billion in 2022, drives demand for drugs like SMZ/TMP DS. Within this, the segment for fixed-dose combination antibiotics accounted for roughly $8 billion, with sulfonamides and trimethoprim compositions representing about 10-15% of this segment—around $0.8 to $1.2 billion.
In the U.S., the prescription volume for SMZ/TMP (all strengths) was roughly 7 million prescriptions in 2022, according to IQVIA data. The fixed-dose double strength form accounts for about 60% of prescriptions, translating to approximately 4.2 million units annually.
How competitive is the current market environment?
Major competitors include generic manufacturers such as Teva, Mylan, and Sandoz. The drug has patent protections that expired in the late 1990s; patent expirations led to increased generic competition, driving prices down significantly. Current pricing for a 20-tablet bottle in the U.S. is approximately $15–$20, compared to branded prices exceeding $100 before generics entered the market.
The expansion of antibiotic resistance pressures has led to a decline in prescriptions for SMZ/TMP DS, with some healthcare providers shifting toward newer antibiotics for resistant strains.
What are the sales projections for SMZ/TMP DS over the next five years?
Sales are projected to decline modestly, driven by:
- Antibiotic Resistance: Growing resistance reduces effectiveness, prompting shifts to alternative treatments.
- Regulatory Focus: Antibiotic stewardship programs restrict overuse, impacting prescribing patterns.
- Market Saturation: The product's patent expiry and generic prevalence limit pricing power.
Based on current trends, global sales of SMZ/TMP DS are expected to:
| Year |
Estimated Global Sales (USD Millions) |
Growth Rate (Year-over-Year) |
| 2023 |
$150–180 |
-2% to 0% |
| 2024 |
$140–170 |
-3% to -1% |
| 2025 |
$130–160 |
-2% to 1% |
| 2026 |
$120–150 |
-2% to 2% |
| 2027 |
$110–140 |
-3% to 0% |
These projections reflect a gradual decline, aligned with overall antibiotic market trends.
What are the regulatory and pipeline considerations?
No current approved novel formulations or indications are in advanced development stages. The focus remains on optimizing existing formulations and combating resistance issues. Any new development would need to demonstrate significant clinical benefits regarding resistance or safety to gain approval and market share.
What are the key risks impacting sales?
- Antibiotic Stewardship: Increasing regulations to curb antibiotic overuse.
- Resistance Development: Rising resistance reduces efficacy, prompting prescribers to choose alternative agents.
- Market Saturation: High generic penetration limits pricing flexibility.
- Emerging Alternatives: Novel antibiotics with broader spectra or fewer side effects could replace SMZ/TMP DS.
Summary of Market Dynamics
| Factor |
Impact |
| Generic competition |
Drives prices downward, limiting margins |
| Resistance trends |
Lower prescription volumes over time |
| Regulatory environment |
Restricts prescribing, curbing sales |
| Market saturation |
Limits growth prospects |
Key Takeaways
- The current global market for SMZ/TMP DS is approximately $150–$180 million, with a declining trend forecasted.
- The segment is mature; major growth drivers are limited to optimizing existing uses rather than expansion.
- Antibiotic resistance and stewardship programs pose ongoing risks to sales volumes.
- Price erosion from generics constrains revenue despite steady prescription volumes.
- No promising new formulations or indications are imminent, suggesting limited upside in near-term projections.
FAQs
1. What factors could reverse the declining trend in SMZ/TMP DS sales?
Advances in combating antibiotic resistance, regulatory incentives for reformulation, or new indications could stimulate demand.
2. How does resistance impact the use of SMZ/TMP DS?
Rising resistance reduces clinical effectiveness, leading prescribers to choose alternative antibiotics, decreasing sales.
3. What is the typical price for SMZ/TMP DS in different markets?
In the U.S., generic double strength tablets cost about $0.75–$1.00 per tablet; branded versions have historically been priced over $4 per tablet before patent expiry.
4. Are there any competitors with superior efficacy or safety?
Several newer antibiotics (e.g., fosfomycin, nitrofurantoin) are preferred for certain infections due to resistance or safety profiles, impacting SMZ/TMP DS's market share.
5. What is the outlook for patent protection?
Patents for SMZ/TMP formulations have long expired; current market is driven solely by generic competition.
Sources:
[1] IQVIA, 2022 Prescription Data
[2] MarketsandMarkets, Antibiotics Market Report, 2022
[3] U.S. Food and Drug Administration, Patent and Exclusivity Data