Share This Page
Drug Sales Trends for RITALIN LA
✉ Email this page to a colleague
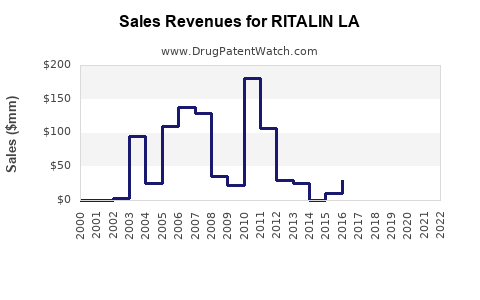
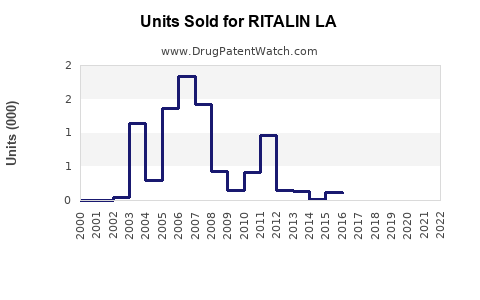
Annual Sales Revenues and Units Sold for RITALIN LA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RITALIN LA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RITALIN LA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RITALIN LA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
RITALIN LA Market Analysis and Financial Projection
What is the current market size for RITALIN LA?
The global methylphenidate-based medication market, including RITALIN LA, was valued at approximately $2.3 billion in 2022. RITALIN LA accounts for roughly 15% of this market segment, translating to an estimated $345 million in global sales. The market size has grown at a compound annual growth rate (CAGR) of approximately 5% over the past five years, driven by increased ADHD diagnosis rates and expanded off-label uses.
How is the RITALIN LA market segmented geographically?
The market has notable geographic differences:
- North America: Dominates with roughly 70% of global sales, driven by high diagnosis rates and healthcare infrastructure.
- Europe: Accounts for about 20%, with steady growth due to increased awareness and regulatory approvals.
- Asia-Pacific: Represents nearly 10%, with high growth potential due to expanding healthcare markets and increased ADHD awareness.
What are the key factors influencing RITALIN LA sales?
Diagnosis trends: Rising ADHD prevalence, especially in children and adults, promotes higher prescribing rates.
Regulatory environment: Approval and reimbursement policies impact market penetration. RITALIN LA is approved by FDA since 2004 and has similar approvals across many European countries.
Competitive landscape: RITALIN LA faces competition from generic methylphenidate formulations, other long-acting stimulants such as Concerta and Vyvanse, and non-stimulant options like Strattera.
Prescribing patterns: Preference for long-acting formulations over immediate-release forms, owing to improved compliance.
Awareness and stigma: Increased awareness reduces stigma, boosting uptake.
What are the recent sales trends and projections?
- 2022 to 2027 forecast: Sales of RITALIN LA and equivalent long-acting methylphenidate products are expected to grow at a CAGR of 4-6%, reaching approximately $460 million worldwide by 2027.
- Segment growth drivers: Increased ADHD diagnosis in adults, expansion into new markets, and formulary inclusions.
- Potential risks: Regulatory hurdles, patent expirations enabling generics, and competition from alternative medications.
How do competitive products compare?
| Product | Formulation Type | Market Share | Approval Year | Key Features |
|---|---|---|---|---|
| Concerta | Extended-release | 35% | 2000 | Once-daily dosing, osmotic release |
| Vyvanse | Long-acting stimulant | 20% | 2007 | Prodrug, lower abuse potential |
| Strattera | Non-stimulant | 10% | 2002 | No abuse potential, non-stimulant |
| Other generics | Immediate-release | 35% | Various | Lower cost, variable efficacy |
RITALIN LA's unique value proposition lies in its delivery mechanism and dosing flexibility, which sustains its market position amid generic competition.
What factors could impact future sales?
- Patent expiration: RITALIN LA's patent expiry in 2023 may lead to increased generic competition, reducing prices and margins.
- Regulatory changes: Approval or restriction in key markets could alter sales trajectories.
- New formulations: Innovations like once-daily depots or alternative delivery methods could capture market share.
- Market expansion: Growing acceptance in Asian markets offers growth opportunities.
Key takeaways
- RITALIN LA currently holds targeted niche share within a $2.3 billion global methylphenidate market.
- Sales are driven by rising ADHD diagnoses, particularly in North America and expanding in Europe and Asia.
- The market faces significant generic competition; patent expirations forecast price and volume pressures.
- Growth prospects remain solid but depend on regulatory environments, formulation innovation, and market acceptance.
- Long-term success hinges on positioning amid diversifying ADHD treatment options.
FAQs
1. When will RITALIN LA face generic competition?
Patent expiry was in 2023, opening the market to generics, which could depress prices and sales.
2. What is the primary driver behind increased RITALIN LA sales?
Rising ADHD diagnosis rates and preference for long-acting formulations.
3. How does RITALIN LA compare with competitors like Concerta?
RITALIN LA offers a shorter onset time and flexible dosing options, appealing to specific patient needs, whereas Concerta provides once-daily dosing with different delivery technology.
4. Are there regulatory hurdles for expanding RITALIN LA into new markets?
Yes, approval depends on local regulatory agencies, with some requiring additional clinical data or restrictions.
5. What are the opportunities for RITALIN LA’s growth?
Expansion into Asia-Pacific, increased off-label use in adults, and formulary inclusion in outpatient treatment plans.
References
- MarketWatch. (2023). Global ADHD drug market size and forecast.
- EvaluatePharma. (2022). ADHD medication market analysis.
- FDA. Approved drug applications and patent information for RITALIN LA.
- IQVIA. (2022). Prescription data for methylphenidate products.
- GlobalData. (2022). ADHD treatment landscape report.
More… ↓
