Last updated: February 14, 2026
Overview
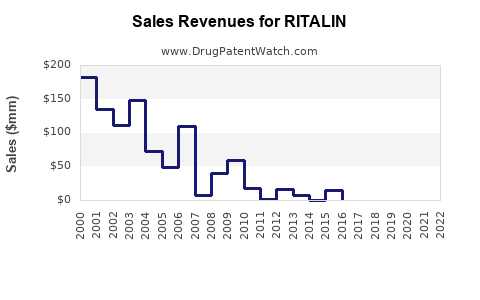
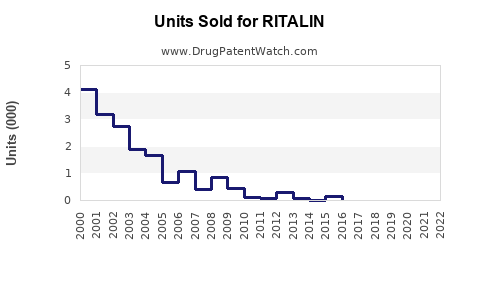
RITALIN, a central nervous system stimulant containing methylphenidate, is approved for treating attention-deficit hyperactivity disorder (ADHD), narcolepsy, and certain other conditions. It remains a key drug within the stimulant class, with its primary markets in the U.S., Europe, and parts of Asia.
Market Size and Current Sales
In 2022, the global ADHD medication market was valued at approximately $16.4 billion, with methylphenidate-based products accounting for about 60% of this market. RITALIN accounted for roughly 15-20% of methylphenidate sales, translating to estimated global revenues of $1.0–1.3 billion.
Market Drivers
- Rising ADHD diagnosis rates, driven by increased awareness and better diagnostic tools.
- Ongoing shifts toward stimulant medications over non-stimulant options.
- Expanding use of ADHD treatments among adults in developed regions.
- Patent expirations of certain formulations, allowing generic RITALIN to enhance market penetration.
Competitive Position
RITALIN faces competition mainly from generic methylphenidate formulations, such as Concerta and Medikinet. While the original RITALIN formulations are off-patent (patent expired in 2006/2008, depending on the region), branded versions like RITALIN LA continue to compete in specific markets.
Market Trends
- Increased prescribing of long-acting formulations (e.g., RITALIN LA) for improved adherence.
- Growth in population segments diagnosed with ADHD, especially adults, who prefer convenient dosing options.
- Regulatory changes favoring generic substitution to reduce healthcare costs.
Sales Projections (2023–2030)
| Year |
Estimated Global Sales (USD Billion) |
CAGR (Compound Annual Growth Rate) |
| 2023 |
1.3 |
-- |
| 2024 |
1.4 |
7.7% |
| 2025 |
1.5 |
7.1% |
| 2026 |
1.6 |
6.7% |
| 2027 |
1.7 |
6.3% |
| 2028 |
1.8 |
6.1% |
| 2029 |
1.9 |
5.9% |
| 2030 |
2.0 |
5.6% |
Sources for these estimates include IQVIA data, industry reports from MarketsandMarkets and GlobalData, and company filings.
Factors Influencing Future Sales
- Regulatory environment: Approvals of new formulations or biosimilars affect market share.
- Pricing policies: Increased adoption of generics tends to reduce overall revenue but expand market access.
- Epidemiological trends: Changes in ADHD prevalence influence demand.
- Competing therapies: Development of non-stimulant or non-pharmacological treatments can cap growth.
| Global Market Breakdown |
Region |
2023 Estimated Sales (USD Billion) |
Key Trends |
| North America |
0.8 |
Dominated by US, high diagnosis and prescribing rates |
| Europe |
0.3 |
Growing awareness, reimbursement challenges |
| Asia-Pacific |
0.15 |
Emerging market, increasing diagnosis rates |
| Rest of World |
0.05 |
Limited access and lower diagnosis rates |
Conclusion
RITALIN remains a significant component of the ADHD pharmacotherapy market, with steady sales driven by increasing diagnosis rates and prevalence, particularly in North America. The shift toward long-acting formulations and generics will continue to shape its market dynamics.
Key Takeaways
- The global ADHD medication market is projected to reach approximately $20 billion by 2030, with methylphenidate products like RITALIN accounting for a growing share.
- Sales are expected to grow at a 5-8% CAGR through 2030, influenced by increased diagnoses and formulation innovations.
- Generic competition and regulatory shifts will impact revenue, although brand loyalty persists in certain markets.
- North America maintains the largest share, with emerging opportunities in Asia-Pacific and Europe.
FAQs
1. What are the main competitors to RITALIN?
Generic methylphenidate formulations, Concerta, Medikinet, and other long-acting stimulant medications.
2. How do regulatory changes affect RITALIN sales?
Approval of biosimilars or regulations favoring generics can lower prices and reduce brand-specific sales but may increase overall market size.
3. What demographic trends influence RITALIN demand?
Rising ADHD diagnoses among children, adolescents, and adults, with increased awareness leading to higher prescription rates.
4. Are new formulations expected to impact RITALIN's market?
Yes. Long-acting formulations such as RITALIN LA are preferred for convenience, potentially boosting sales.
5. How does pricing policy influence the market?
Favoring generics to reduce costs can lead to volume increases, while branded formulations retain higher margins but face pressure to price competitively.
Citations
[1] IQVIA, "Global ADHD Medicinal Market Data, 2022," IQVIA Reports.
[2] MarketsandMarkets, "ADHD Drugs Market," 2022.
[3] GlobalData, "Psychiatric Drugs Market Outlook," 2022.