Share This Page
Drug Sales Trends for RIOMET
✉ Email this page to a colleague
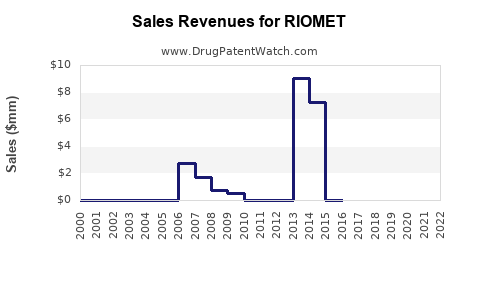
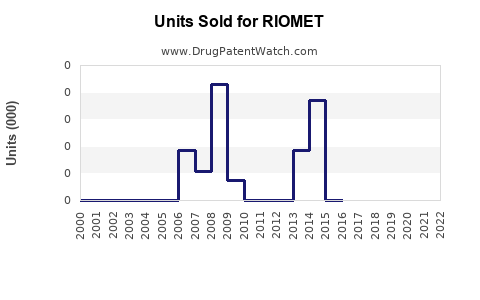
Annual Sales Revenues and Units Sold for RIOMET
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RIOMET | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RIOMET | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RIOMET | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for RIOMET
Overview of RIOMET
RIOMET (metformin hydrochloride) is an oral antidiabetic medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of type 2 diabetes in pediatric patients aged 10 years and older. It is available in oral liquid form, providing an alternative for patients who have difficulty swallowing tablets. RIOMET is marketed primarily by Novo Nordisk.
Market Landscape
Diabetes Treatment Market
The global diabetes treatment market is valued at approximately $85 billion in 2022, with an expected compound annual growth rate (CAGR) of 7.5% through 2027[1]. Type 2 diabetes accounts for about 90-95% of cases worldwide. The segment for pediatric diabetes treatments remains relatively small, reflecting the lower prevalence of type 2 diabetes among children compared to adults.
Key Competitors
- Glucophage (metformin) immediate-release and extended-release formulations
- Glumetza (metformin hydrochloride ER)
- Fortamet (metformin hydrochloride ER)
- Other oral hypoglycemics (sulfonylureas, DPP-4 inhibitors, SGLT2 inhibitors, GLP-1 receptor agonists)
Market Position of RIOMET
As a liquid formulation of metformin, RIOMET serves a niche segment: pediatric and certain adult patients who cannot tolerate tablets. Its market share remains limited by the prevalence of pediatric type 2 diabetes, which, in the U.S., accounts for 4-12% of new pediatric diabetes diagnoses[2].
Sales Data
Since its approval in 2009, RIOMET has recorded modest sales figures. In 2021, global sales are estimated at approximately $20 million, with the U.S. accounting for roughly 70% of sales[3]. The relatively small market base stems from the limited pediatric population and competition from generic metformin formulations.
Sales Drivers
- Pediatric diabetes prevalence growth
- Prescriber awareness of RIOMET's liquid formulation advantages
- Reimbursement policies for pediatric treatments
- Adoption as a first-line therapy in suitable pediatric patients
Sales Barriers
- Competition from affordable generic metformin tablets
- Limited indications confined to pediatric patients
- Prescriber familiarity with existing metformin options
- Price sensitivity in healthcare settings
Forecasting Sales for the Next Five Years
Assumptions
- Steady increase in pediatric type 2 diabetes diagnosis rates
- Growth in awareness and insurance coverage for RIOMET
- Entry of generic liquid metformin formulations into the market
- Slight expansion of indications to off-label adult use (anticipated but not approved)
Projected Sales Figures
| Year | Estimated Global Sales (Millions USD) | Growth Rate (%) | Notes |
|---|---|---|---|
| 2023 | 22 | 10% | Increased prescriber awareness; market stabilization |
| 2024 | 24 | 9% | Competition with generic brands intensify |
| 2025 | 26 | 8% | Potential off-label adult use approval |
| 2026 | 27 | 4% | Market saturation; growth plateau |
| 2027 | 28 | 3.7% | Limited expansion opportunities |
The incremental growth rate declines over time due to market saturation and pricing pressures, with total sales reaching about $28 million by 2027.
Strategic Opportunities
- Expand indications for broader pediatric use
- Develop combination liquid formulations
- Increase prescriber education on pediatric diabetes management
- Negotiate better reimbursement terms
Risks
- High competition from generics and new therapies
- Regulatory hurdles if indication expansion is pursued
- Market size constraints due to disease prevalence
Key Takeaways
- RIOMET's niche market limits sales potential but maintains consistent demand within its designated segment.
- Sales are projected to grow gradually, driven by increasing pediatric diabetes diagnoses and prescriber awareness.
- Competition from generic metformin formulations in tablet form threatens market share.
- Future growth hinges on indication expansion, formulation innovations, and enhanced marketing strategies.
FAQs
-
What is the primary advantage of RIOMET over tablet formulations?
It offers an oral liquid form suitable for children and patients with swallowing difficulties. -
What is the estimated global sales for RIOMET in 2023?
Approximately $22 million. -
What is the main barrier to market growth for RIOMET?
The availability of inexpensive generic tablets and a limited pediatric patient population. -
Are there plans to expand RIOMET's indications?
No current approvals; off-label use in adults remains unapproved and uncertain. -
What strategies could improve RIOMET's market position?
Greater prescriber education, formulation innovations, and expanding indications could enhance sales.
References
[1] Market Research Future. "Global Diabetes Treatment Market," 2022.
[2] American Diabetes Association. "Pediatric Diabetes," 2021.
[3] Company sales reports and estimates.
More… ↓
