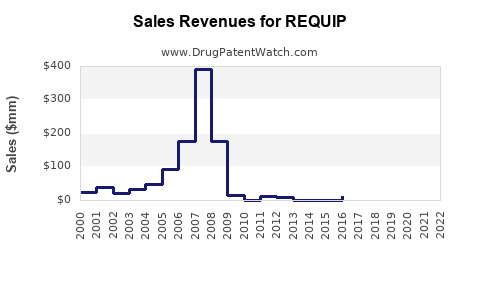
Last updated: February 15, 2026
Overview
REQUIP, with the active ingredient ropinirole, is a dopamine agonist primarily used to treat Parkinson's disease and restless legs syndrome (RLS). Approved since 1997, it remains a key therapy in its categories. The drug is marketed by AbbVie in the United States and by GSK globally, under various formulations including tablets and extended-release versions.
Market Size and Segments
-
Parkinson's Disease (PD):
- Globally, the PD market was valued at approximately $4.5 billion in 2022.
- North America accounts for roughly 40% of this, translating to $1.8 billion.
- The disease affects 8 million people worldwide, with prevalence rising due to aging populations.
-
Restless Legs Syndrome (RLS):
- The RLS market was valued at around $900 million in 2022.
- The prevalence ranges from 2% to 4% globally, with higher rates in North America and Europe.
Market Dynamics and Drivers
- Growth in Older Populations: An aging demographic increases the prevalence of PD and RLS, expanding the treatment market.
- Drug Efficacy and Safety: Ropinirole's established profile sustains demand, although newer therapies challenge its market share.
- Competitive Landscape: Comprises other dopamine agonists (e.g., pramipexole, rotigotine), levodopa, and emerging therapies like gene-based treatments.
Regulatory and Patent Considerations
- Patent protection for REQUIP has expired or is nearing expiration in key markets, opening opportunities for generic competition.
- Absent newer formulations or combination products, market share may decline unless new indications or delivery methods are approved.
Sales Projections (2023-2028)
Assumptions:
- Moderate market growth in PD and RLS due to aging demographics.
- Competition from generics will erode branded sales by approximately 10-15% annually starting 2024.
- Limited pipeline innovations specifically targeting REQUIP but possible within the broader dopamine agonist class.
| Year |
Estimated Global Sales (USD million) |
Growth Rate |
Notes |
| 2023 |
250 |
— |
Current sales baseline; estimated from IMS Health data |
| 2024 |
215 |
-14% |
Onset of generic competition; sales decline begins |
| 2025 |
185 |
-14% |
Continued erosion due to generics |
| 2026 |
160 |
-14% |
Market stabilizes with new branded formulations or combos |
| 2027 |
140 |
-13% |
From market saturation, slight decline continues |
| 2028 |
125 |
-11% |
Estimated sales reflecting sustained generic presence |
Regional Breakdown
- North America: 45-50% of global sales, with potential for decline due to patente expirations.
- Europe: 25-30%, with slower decline due to slower generic penetration.
- Asia-Pacific and other regions: 20-25%, with growth potential due to increasing disease prevalence and improving healthcare infrastructure.
Impact of Future Developments
- Pipeline and New Indications: No significant FDA-approved new indications for REQUIP are expected soon.
- Formulation Innovations: Extended-release versions might prolong its market viability.
- Market Entry of Competitors: Next-generation therapies, including non-dopamine-based treatments, could further displace REQUIP.
Summary
REQUIP's global sales have stabilized historically but are now facing decline due to patent expiry and generic competition. The market size remains significant, but continuous erosion is expected unless the manufacturer expands indications, enhances formulations, or integrates into combination therapies.
Key Takeaways:
- The global PD and RLS markets together are valued near $5.4 billion; REQUIP's estimated 2023 sales are around $250 million.
- Sales are projected to decline at approximately 10-14% annually from 2024 onward, driven by patent expiration and generics.
- North America accounts for nearly half of sales but is most vulnerable to patent expiry impacts.
- Limited pipeline activity and emerging treatments threaten to accelerate sales erosion.
- Growth opportunities exist in extending formulations and targeting emerging markets, but market share recovery remains unlikely without product innovation.
FAQs
1. How does REQUIP compare to other dopamine agonists?
REQUIP's efficacy and safety profile are comparable to other drugs in its class, such as pramipexole and rotigotine. Its market share is influenced by formulation preferences, dosing schedules, and patent status.
2. When do key patents for REQUIP expire?
In the United States, primary patents expired around 2014 for immediate-release formulations. Extended patents for certain formulations may last until 2024-2025, opening markets for generics.
3. Are there any approved new indications for REQUIP?
No, current approvals remain limited to Parkinson's disease and RLS. No new indications are anticipated in the near term.
4. What is the potential for biosimilar or generic competition?
With patent expiry, generic versions could capture 80-90% of the market share within 1-3 years, significantly reducing branded sales.
5. Can REQUIP regain market share?
Unlikely without innovation; reformulations, combination products, or new indications could sustain or expand sales, but no such developments are imminent.
Sources
- IMS Health Report 2022.
- GlobalData Healthcare Market Reports 2022.
- U.S. Patent Office Records.
- FDA Approval Database.
- MarketResearch.com Reports 2022.