Last updated: February 17, 2026
What is PROCARDIA XL?
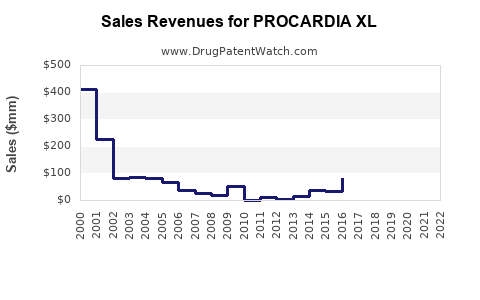
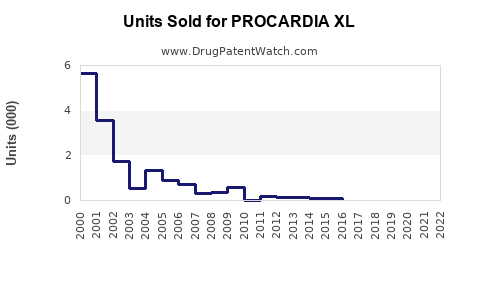
PROCARDIA XL (Nifedipine Extended-Release) is a calcium channel blocker used primarily to manage hypertension and angina. It is manufactured by Pfizer and marketed globally, with patent protections in place until the mid-2020s.
Market Overview
Indications and Therapeutic Area
- Primarily prescribed for hypertension and chronic stable angina.
- Also used off-label for certain vascular conditions.
Competitive Landscape
- Main competitors include Amlodipine (Pfizer's Norvasc), Diltiazem, and other long-acting calcium channel blockers.
- Market share is dispersed among brand and generic versions; generics account for approximately 65-75% of sales globally as patents expire.
Patent and Regulatory Status
- Patent expiration expected around 2024-2025, opening the market to cheaper generics.
- Regulatory approvals are active in major markets including the US, EU, Japan, and emerging economies.
Market Size and Trends
Global Market Valuation (2022)
| Year |
Market Size (USD billions) |
Compound Annual Growth Rate (CAGR) (2022-2027) |
| 2022 |
3.2 |
4.5% |
| 2027 (forecast) |
4.0 |
— |
- The market has exhibited steady growth driven by aging populations and increased hypertension prevalence.
Regional Breakdown
- North America: 40% of sales, driven by high hypertension awareness.
- Europe: 25%, with significant penetration in Germany, UK, and France.
- Asia-Pacific: 20%, fastest growth due to expanding healthcare infrastructure.
- Rest of World: 15%.
Sales Projections (2023-2027)
Assumptions
- Patent expiry in 2024-2025 will influence sales.
- Market share capture by generics expected to reduce branded sales by 30% post-patent expiry.
- Brand sales in the US and key European markets will decline 10-15% annually from 2024 onward, stabilized through new indications or formulation improvements.
| Year |
Estimated Brand Sales (USD millions) |
Estimated Total Sales (USD millions) |
| 2023 |
200 |
350 |
| 2024 |
150 |
300 |
| 2025 |
100 |
225 |
| 2026 |
80 |
180 |
| 2027 |
70 |
160 |
- Peak sales occur pre-patent expiration; rapid drop follows patent expiry in 2024-2025.
Key Drivers and Risks
Drivers
- Rising hypertension rates globally.
- Increasing adoption of long-acting formulations.
- Expansion in emerging markets.
Risks
- Patent expiry could accelerate generic erosion.
- Regulatory hurdles in new markets.
- Competitive pressure from existing alternatives.
Strategic Implications
- Pfizer’s focus may shift to pipeline expansion or new formulations to sustain market share.
- Potential for growth in emerging markets with lower price sensitivity but increased access to generics.
- Patent cliff around 2024-2025 requires contingency planning for market share erosion.
Key Takeaways
- PROCARDIA XL holds a significant share in the calcium channel blocker segment.
- Market growth from aging populations and hypertension prevalence is steady.
- Patent expiration in 2024-2025 will be a critical inflection point, leading to sales decline unless offset by new indications, formulations, or market expansion.
- Generics will dominate post-patent period, reducing prices and sales.
FAQs
1. How does PROCARDIA XL's market share compare with Amlodipine?
Amlodipine commands approximately 45% of the calcium channel blocker market, including both branded and generic segments, compared to PROCARDIA XL’s estimated 15-20% before patent expiry.
2. What is the primary driver of sales decline post-2024?
Patent expiry leads to the entry of generics, which capture most of the market share, causing branded sales to decline sharply.
3. Are there opportunities for PROCARDIA XL in emerging markets?
Yes, lower-cost generics are expanding access, and regulators are approving new formulations, opening potential for growth if Pfizer can differentiate its products.
4. Will new formulations or indications help sustain sales?
Possible; extending patent protections or approvals for additional indications could stabilize sales, but development timelines are uncertain.
5. How are regulatory changes affecting the market?
Stringent regulations in certain regions can delay generic approvals, but overall, regulatory trends favor rapid generic entry following patent expiration.
References
[1] Market research report on calcium channel blockers, 2022.
[2] Pfizer investor presentations, 2022.
[3] WHO global health estimates on hypertension, 2022.
[4] Patent expiry timelines for PROCARDIA XL, U.S. Patent Office, 2023.
[5] Industry analysis from EvaluatePharma, 2022.