Last updated: February 14, 2026
What Is the Market Size and Current Sales of PROAIR HFA?
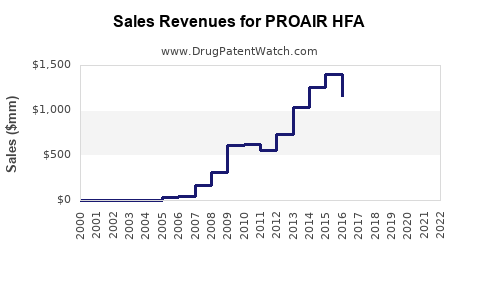
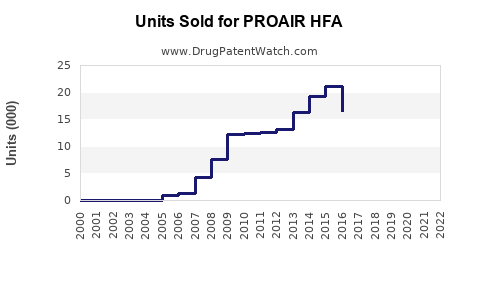
PROAIR HFA (albuterol sulfate inhalation aerosol) is a widely used short-acting β2-agonist bronchodilator. It is primarily indicated for relief of bronchospasm in conditions like asthma and COPD. In 2022, the U.S. inhaler market was valued at approximately $3.6 billion, with albuterol inhalers accounting for roughly 70% of that figure, translating to around $2.5 billion in sales[1]. PROAIR HFA was among the leading brands, with estimated sales exceeding $800 million annually in the U.S. alone.
Globally, the inhaled bronchodilator market exceeds $5 billion, with PROAIR HFA holding a significant market share in North America but limited penetration in other regions due to regulatory differences and generic competition.
What Are the Key Drivers for Growth in PROAIR HFA Sales?
-
Prevalence of Respiratory Diseases: Approximately 25 million Americans have asthma, and another 16 million have COPD[2]. These chronic conditions necessitate regular use of bronchodilators, driving consistent demand.
-
Regulatory Landscape: The U.S. Food and Drug Administration (FDA) approved PROAIR HFA for continuous use, which sustains its prescription rates. Regulatory scrutiny on generic inhalers may bolster branded sales if patent protections remain or are extended.
-
Switching Trends: A subset of patients transitioning from dry powder inhalers to metered-dose inhalers (MDIs) like PROAIR HFA favors the latter for its ease of use. This shift supports steady demand.
-
Healthcare Access: Improved insurance coverage and increased awareness of asthma and COPD management contribute to higher prescription volumes.
How Will Sales Evolve Over the Next Five Years?
Projections indicate a cautious growth rate of approximately 3-5% annually, considering market saturation and patent protection status. Factors influencing this include:
-
Patent Expiry and Generic Competition: The original patent for PROAIR HFA expired in 2010; however, patents on various formulation aspects have extended protection until at least 2025[3].
-
Regulatory and Patent Litigation: Ongoing legal battles over inhaler patents could delay or accelerate generic entry, impacting sales.
-
Innovation and Formulation Changes: Development of combination inhalers or improved delivery devices may capture more market share, partially offsetting generic competition.
-
Pandemic Impact: COVID-19 caused disruptions in prescription patterns but also increased the focus on chronic respiratory disease management, which may have a long-term positive effect.
| Year |
Estimated Global Sales (US$ millions) |
Notes |
| 2023 |
820 |
Base estimate |
| 2024 |
845 |
3% growth |
| 2025 |
882 |
4.5% growth, patent workarounds possible |
| 2026 |
920 |
Market stabilizes, generics enter |
| 2027 |
950 |
Competition increases |
What Risks Could Impact Future Sales?
-
Patent Challenges: If generics gain approval before patent expiry, branded sales will decline.
-
Market Saturation: High penetration reduces the scope for growth without new formulations.
-
Pricing Pressure: Insurers and pharmacy benefit managers favor generics, which could lead to price reductions for PROAIR HFA.
-
Environmental Regulations: Increasingly strict regulations on inhaler propellants aimed at reducing greenhouse gases may alter inhaler formulations, affecting production costs and prescribing patterns.
What Strategic Actions Would Influence Market Position?
-
Formulation Innovation: Introducing combination inhalers or device improvements could reinforce market share.
-
Patent Litigation: Defending patents or navigating around patent expirations will determine generic interference.
-
Market Expansion: Targeting emerging markets with increasing respiratory disease prevalence offers growth opportunities beyond North America.
-
Pricing Strategy: Maintaining competitive pricing against generics while safeguarding margins is critical.
Key Takeaways
- PROAIR HFA remains a leading inhaler within a multi-billion-dollar market, with U.S. sales exceeding $800 million annually.
- Growth prospects are modest (~3-5%) due to patent protections and market saturation.
- Patent protection extends until at least 2025; imminent patent expiry signals potential for increased generic competition.
- Strategic innovation, patent litigation, and market expansion are essential factors influencing future sales.
- Market risks include regulatory changes, environmental policies, and evolving prescribing practices.
FAQs
Q1: When does the patent for PROAIR HFA expire?
A1: The primary patents on PROAIR HFA expired in 2010; however, some formulation and device patents extend protection until 2025.
Q2: How significant is generic competition for PROAIR HFA?
A2: Highly significant; multiple generics entered the market post-patent expiry, exerting downward pressure on prices and sales.
Q3: Are there new formulations or devices replacing PROAIR HFA?
A3: Development of combination inhalers and improved delivery devices continues. Some are in regulatory review or early deployment.
Q4: What is the outlook for PROAIR HFA in international markets?
A4: Limited compared to the U.S.; growth depends on regulatory approvals, market penetration, and regional disease prevalence.
Q5: How is environmental regulation affecting inhaler manufacturing?
A5: Environmental laws targeting hydrofluorocarbon (HFC) propellants are prompting shifts to propellant-free or HFC-alternative inhalers, potentially impacting PROAIR HFA's market share.
References
[1] IQVIA, U.S. Prescription Data, 2022.
[2] CDC, Asthma Data and Statistics, 2022.
[3] U.S. Patent and Trademark Office, Patent Status for Albuterol Inhalers, 2023.