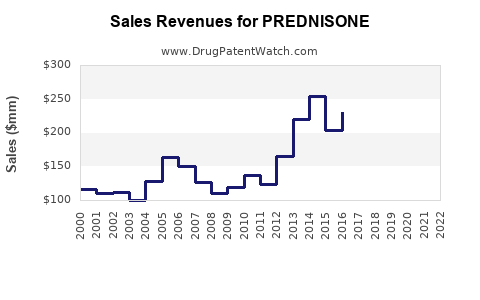
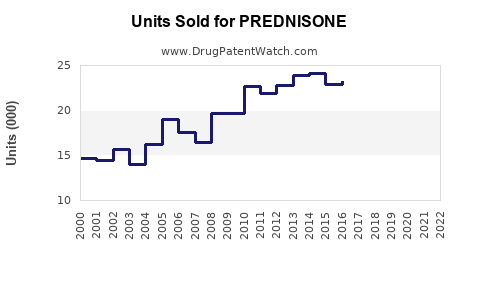
Last updated: February 20, 2026
What Is the Current Market Size for Prednisone?
Prednisone is a synthetic corticosteroid used primarily for its anti-inflammatory and immunosuppressive properties. It is prescribed for conditions such as asthma, allergies, autoimmune diseases, and certain cancers. The global corticosteroids market was valued at approximately USD 7.3 billion in 2022 and is expected to grow at a CAGR of 4.2% through 2030 (Grand View Research, 2023).
Prednisone accounts for the largest share of this market, driven by its widespread approval, generic availability, and diverse therapeutic uses.
How Is Prednisone Positioned in the Global Market?
Pricing and Competitive Landscape
- Prednisone is marketed primarily as a generic drug, with oral tablets available from multiple manufacturers.
- Average retail price per 20mg tablet in the US is approximately USD 0.10–0.20.
- Major companies manufacturing Prednisone include Upsher-Smith Laboratories, Amneal Pharmaceuticals, and Mylan.
Regulatory Environment
- Prednisone approval exists in markets across North America, Europe, Asia Pacific, and Latin America.
- Regulatory approval processes are streamlined for generics in the US (FDA), Europe (EMA), and elsewhere, facilitating continued market entry.
Market Penetration
- Prednisone has high penetration in developed markets due to established prescribing patterns.
- Growth in emerging markets is driven by increasing healthcare infrastructure and prevalence of autoimmune conditions.
What Are the Key Drivers and Barriers for Prednisone Sales?
Drivers
- Expanding indications: Off-label uses and new clinical evidence support broader prescribing.
- Cost-effectiveness: Lower price compared to biologics and newer therapies sustains demand.
- Aging populations: Increased autoimmune and inflammatory disease prevalence.
Barriers
- Side effect profile: Long-term use linked with osteoporosis, hyperglycemia, and immune suppression.
- Availability of newer agents: Biologics and targeted therapies can replace Prednisone in some indications.
- Regulatory restrictions: Restrictions on long-term corticosteroid use due to safety concerns.
How Will Sales Evolve Through 2030?
Projected Sales Growth
| Year |
Estimated Global Sales (USD Billion) |
Growth Rate (CAGR) |
| 2022 |
1.6 |
-- |
| 2025 |
2.0 |
6.2% |
| 2030 |
2.5 |
4.8% |
Assumptions: Growth driven by increasing demand in emerging markets, expanding indications, and continued generic competition.
Regional Growth Trends
- North America: Largest market, USD 0.8–1.0 billion in 2022, expected CAGR of 3.5–4% due to high penetration.
- Europe: Similar growth dynamics; sales projected to reach USD 0.5 billion by 2030.
- Asia Pacific: Fastest growth, CAGR approximately 6.5%, driven by healthcare infrastructure expansion and rising autoimmune disease prevalence.
Impact of Patent and Regulatory Changes
- As patents expire (many in the late 2010s), new generic entries will sustain price competition.
- Regulatory policies favor generics, minimizing barriers and supporting sales volume.
What Are the Key Risks and Opportunities?
Risks
- Safety concerns and side effects could limit long-term use.
- Competition from newer, targeted therapies, especially for autoimmune conditions.
- Pricing pressures, especially in price-sensitive markets.
Opportunities
- Development of fixed-dose combination products.
- Expansion into new indications backed by clinical trials.
- Entry into emerging markets with expanding healthcare access.
Summary
Prednisone retains a dominant position in corticosteroid therapy segments, with steady sales influenced by its cost advantage, generic availability, and broad prescribing indications. Market growth is driven by demographic trends and expanding indications but limited by safety concerns and competition from newer therapies.
Key Takeaways
- Global Prednisone sales were approximately USD 1.6 billion in 2022, with a forecast to reach USD 2.5 billion by 2030.
- The drug's market is mature in developed regions, with rapid growth in emerging markets.
- Price competition and safety issues are primary barriers; innovation and market access strategies are essential for future growth.
FAQs
1. How do patent expiries affect Prednisone sales?
Patent expiries lead to increased generic competition, decreasing prices and potentially increasing overall sales volume. It sustains market entry and reduces barriers for new manufacturers.
2. What are major therapeutic indications for Prednisone?
Prednisone is used for inflammatory conditions (asthma, dermatitis), autoimmune diseases (Lupus, rheumatoid arthritis), and certain cancers, among others.
3. In which regions does Prednisone have the highest market growth?
Emerging markets in Asia Pacific and Latin America show the highest growth potential due to increasing healthcare infrastructure and disease prevalence.
4. What safety concerns impact Prednisone usage?
Long-term use is associated with osteoporosis, hyperglycemia, fluid retention, and immune suppression, which can limit indications and duration of therapy.
5. How competitive is the market for Prednisone?
High. Multiple generics are available, with low prices and wide distribution channels. Competition pressures prices but maintains volume.
References
[1] Grand View Research. (2023). Corticosteroids Market Size, Share & Trends Analysis Report.
[2] U.S. Food and Drug Administration. (2022). Approved Drug Products.
[3] EMA. (2022). Market authorization of corticosteroids in Europe.