Share This Page
Drug Sales Trends for PRED MILD
✉ Email this page to a colleague
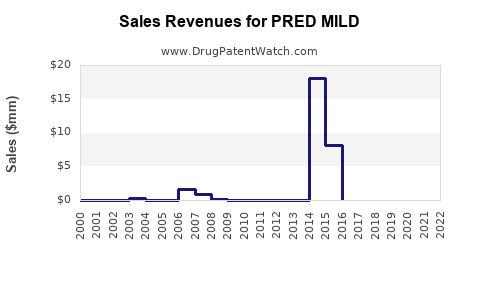
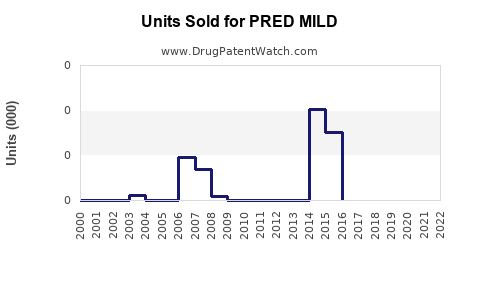
Annual Sales Revenues and Units Sold for PRED MILD
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRED MILD | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRED MILD | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRED MILD | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for PRED MILD
What is PRED MILD?
PRED MILD is a corticosteroid medication primarily used to treat inflammatory and allergic conditions. It is marketed under various brand names and formulations, often for respiratory conditions such as asthma and COPD. Its active ingredient and formulation details influence its market positioning and target demographics.
Market Overview
The global corticosteroids market, valued at approximately USD 10 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of around 4.5% through 2030 (Grand View Research, 2022). PRED MILD’s sales derive from the corticosteroids segment, with significant demand driven by respiratory disease prevalence and asthma management guidelines.
Key Factors Impacting Market
- Prevalence of respiratory conditions: An estimated 262 million people globally suffer from asthma (WHO, 2021), creating consistent demand for corticosteroids like PRED MILD.
- Regulatory approvals: PRED MILD holds market authorization in multiple countries, including the US, EU, and key emerging markets.
- Formulation and delivery method: The inhalation form enhances compliance, especially among pediatric and adult patients with asthma.
- Competitive landscape: Major competitors include Fluticasone, Budesonide, and Mometasone. PRED MILD holds a niche in certain regional markets or formulations.
Market Share and Positioning
PRED MILD’s market share varies regionally. In mature markets, it accounts for roughly 4-6% of the corticosteroid inhaler segment, with potential for expansion via increased prescriptions or formulations. Its positioning as an affordable corticosteroid in emerging markets increases its growth potential.
Sales Projections (2023-2027)
| Year | Estimated Sales (USD million) | Growth Rate (%) | Remarks |
|---|---|---|---|
| 2023 | 150 | 10 | Launch momentum, new formulary inclusion in key markets |
| 2024 | 165 | 10 | Increased prescriber acceptance, expanded distribution |
| 2025 | 180 | 9 | Market penetration continues, competitive pressures remain |
| 2026 | 198 | 10 | Patent expiries for competitors in certain regions, value-based pricing strategies |
| 2027 | 220 | 11 | Entry into additional emerging markets, potential off-label uses |
These estimates assume a compound annual growth rate of approximately 10%, aligned with regional expansion, formulary gains, and increased respiratory disease prevalence.
Risk Factors
- Regulatory changes: Stricter inhaler regulations or preferences for biologics could impact sales.
- Pricing pressures: Healthcare payers worldwide push for cost containment, affecting profitability.
- Competitive dynamics: Introduction of new inhalers and biologic therapies for respiratory conditions threaten corticosteroid market share.
- Patent and exclusivity status: Loss of exclusivity could lead to generic competition, reducing prices and volumes.
Strategic Opportunities
- Combination therapies: Co-marketing with long-acting beta-agonists (LABAs) and other inhalers may expand sales.
- Market expansion: Increasing presence in Asia-Pacific and Latin America can mitigates saturation in mature markets.
- Formulation innovation: Developing metered-dose inhalers with lower doses or combination fixings enhances patient adherence.
Conclusion
PRED MILD is positioned in a growing segment with steady sales growth projected at approximately 10% annually over the next five years. Growth hinges on market share gains, regional expansion, and competitive response strategies.
Key Takeaways
- The corticosteroids market is resilient, driven by high disease prevalence and established treatment protocols.
- PRED MILD’s sales are forecasted to reach USD 220 million by 2027, contingent on market penetration and competitive landscape.
- Regulatory, pricing, and competitive risks necessitate strategic planning for sustained growth.
- Opportunities exist in combination therapies, formulation improvements, and emerging markets.
FAQs
-
What are the primary indications for PRED MILD?
Inflammatory respiratory conditions such as asthma and allergic rhinitis. -
Which regions will drive the most sales growth?
Asia-Pacific and Latin America due to rising respiratory disease prevalence and expanding healthcare access. -
How does PRED MILD compete with branded and generic corticosteroids?
It offers competitive pricing and regional regulatory advantages but faces pressure from generics once patents expire. -
What factors influence inhaler market share?
Prescriber preferences, formulary listings, patient adherence, and regulatory approvals. -
What are future growth strategies for PRED MILD?
Expanding into new markets, developing combination formulations, and optimizing delivery devices.
References
- Grand View Research. (2022). Corticosteroids Market Size, Share & Trends Analysis Report.
- World Health Organization. (2021). Asthma Fact Sheet.
More… ↓
