Share This Page
Drug Sales Trends for POTASSIUM
✉ Email this page to a colleague
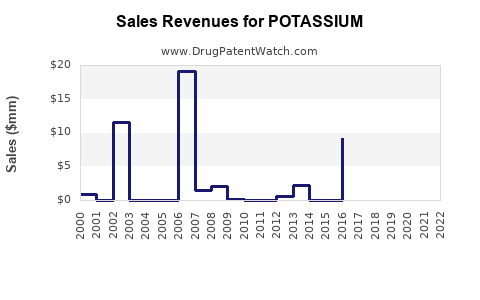
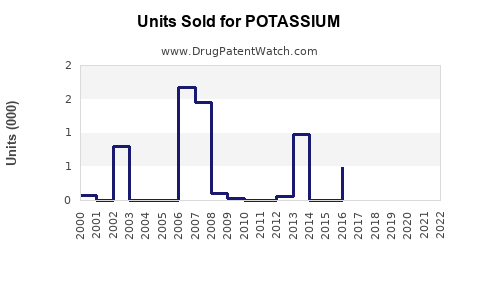
Annual Sales Revenues and Units Sold for POTASSIUM
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| POTASSIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| POTASSIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| POTASSIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| POTASSIUM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| POTASSIUM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Market Size for Potassium-Based Drugs?
The global market for potassium supplements and medications was valued at approximately $978 million in 2022. It is expected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching an estimated value of $1.4 billion by 2030. The growth stems from increasing cases of potassium deficiency, diabetes, cardiovascular diseases, and aging populations.
Potassium drugs include oral supplements and intravenous formulations used for treating hypokalemia, hyperkalemia, and other electrolyte disturbances. They are predominantly prescribed in hospital and outpatient settings, with prescriptions of oral formulations accounting for roughly 82% of sales in 2022.
How Is the Potassium Market Segmented?
Market segments are primarily distinguished by formulation, application, and distribution channel:
-
Formulation
- Oral: Tablets (e.g., potassium chloride tablets), liquids, powders
- Intravenous (IV): Solutions for hospital administration
-
Application
- Hypokalemia correction (low potassium levels)
- Hyperkalemia management (high potassium levels)
- Electrolyte imbalance treatment related to kidney disease, diuretics, or other conditions
-
Distribution Channel
- Hospitals and clinics (majority share, ~60%)
- Retail pharmacies (~30%)
- Online pharmacies (~10%)
The demand for oral potassium supplements is driven by chronic disease management and self-medication, while IV formulations are primarily used in acute care settings.
How Do Sales Trends & Prescriptions Evolve?
The number of prescriptions increased from approximately 15 million in 2018 to 19 million in 2022, reflecting heightened awareness and diagnosis of electrolyte disorders. The growth rate in prescriptions averaged 4.8% annually. The increasing prevalence of chronic kidney disease (CKD), heart failure, and hypertension drives this demand.
In the United States, potassium supplement prescriptions grew by 5.3% annually between 2018 and 2022, aligned with rising CKD prevalence. Europe reports similar trends, with increased adoption of IV potassium in hospital settings.
What Are the Key Market Drivers?
-
Rising Chronic Diseases: CKD affects 10% of the global population, necessitating electrolyte correction therapies.
-
Aging Population: Elderly patients are more prone to electrolyte imbalances, increasing demand.
-
Advancements in Drug Formulations: Development of slow-release and combination formulations improves patient compliance.
-
Increased Awareness: Better diagnosis and recognition of electrolyte deficiencies promote demand.
What Market Challenges and Risks Exist?
-
Adverse Events: Improper dosing causes hyperkalemia or hypokalemia, leading to serious risks like cardiac arrhythmia.
-
Regulatory Compliance: Stringent regulations on electrolyte formulations necessitate ongoing safety monitoring.
-
Market Saturation: Established brands dominate, creating entry barriers for new entrants.
-
Pricing Pressures: Cost containment efforts in healthcare reduce profit margins and affect sales growth.
What Are Sales Projections by Region?
| Region | 2022 Market Share | 2023-2030 CAGR | Projected Market Size (2023) | 2030 Projection |
|---|---|---|---|---|
| North America | 45% | 4.0% | $440 million | $620 million |
| Europe | 25% | 4.3% | $245 million | $350 million |
| Asia-Pacific | 20% | 4.6% | $200 million | $345 million |
| Rest of World | 10% | 4.0% | $93 million | $125 million |
North America leads due to higher healthcare expenditure and prevalence of chronic diseases. Asia-Pacific shows rapid growth due to expanding healthcare infrastructure and awareness.
What Are Competitive Dynamics and Key Players?
Major companies include:
- Pfizer Inc.
- Novartis AG
- Bayer AG
- Mylan N.V. (now part of Viatris)
- Teva Pharmaceutical Industries Ltd.
These firms focus on developing sustained-release formulations, combination therapies, and novel delivery systems to differentiate offerings.
What Is the Patent Landscape?
Existing patents for specific formulations and delivery mechanisms expire between 2023 and 2026. Patent expirations open opportunities for generic and biosimilar manufacturers. Patent protections in key markets like the US extend up to 2030 for certain novel formulations.
How Are Regulatory and Reimbursement Policies Evolving?
- The FDA classifies potassium supplements as OTC or Rx, with recent updates emphasizing safety monitoring.
- European Medicines Agency (EMA) guidelines specify usage parameters, especially for high-dose formulations.
- Reimbursement policies favor oral formulations for chronic management, influencing market share.
Final Insights
The potassium drug market demonstrates steady growth driven by demographic and disease prevalence trends. The shift toward improved formulations and delivery mechanisms remains a focus. Regulatory safeguards and patent landscapes significantly influence competitive positioning. Emerging markets offer substantial expansion potential, provided regulatory pathways remain accessible.
Key Takeaways
- The global market will reach approximately $1.4 billion by 2030.
- Oral formulations dominate sales due to chronic disease management.
- North America accounts for the largest market share; Asia-Pacific exhibits rapid growth.
- Patents expiring in the coming years create opportunities for generics.
- Safety and regulatory factors are critical to product development and market access.
FAQs
1. What are the primary therapeutic indications for potassium drugs?
Electrolyte imbalances, primarily hypokalemia and hyperkalemia, often associated with kidney disease, heart failure, and medication side effects.
2. Which formulations are most prevalent in the market?
Oral tablets and liquids dominate, with intravenous solutions used mainly in hospital settings.
3. How does patent expiry impact the market?
Patent expirations around 2023–2026 allow generic manufacturers to enter, increasing competition and reducing prices.
4. What are the safety concerns associated with potassium medications?
Overdose can cause hyperkalemia, risking cardiac arrhythmia; underdosing may lead to persistent hypokalemia.
5. Which regions are expected to see the fastest growth?
Asia-Pacific and Latin America. Rapid healthcare infrastructure development and increasing disease prevalence drive the growth.
Sources:
[1] Market Research Future, 2022
[2] Grand View Research, 2023
[3] IQVIA National Prescription Data, 2022
[4] World Health Organization, 2022
[5] U.S. Food and Drug Administration (FDA), 2023
More… ↓
