Share This Page
Drug Sales Trends for POLYETHYLENE GLYCOL 3350
✉ Email this page to a colleague
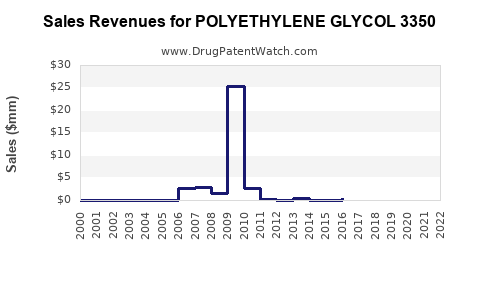
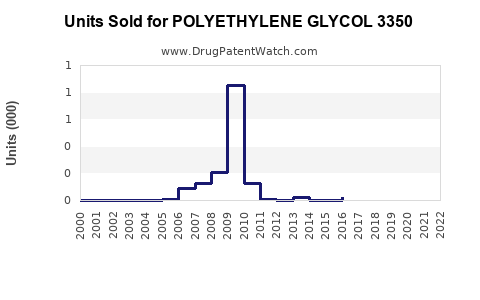
Annual Sales Revenues and Units Sold for POLYETHYLENE GLYCOL 3350
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| POLYETHYLENE GLYCOL 3350 | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| POLYETHYLENE GLYCOL 3350 | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| POLYETHYLENE GLYCOL 3350 | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| POLYETHYLENE GLYCOL 3350 | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
POLYETHYLENE GLYCOL 3350 Market Analysis and Financial Projection
What Is the Current Market Size for Polyethylene Glycol 3350 (PEG 3350)?
Polyethylene glycol 3350 (PEG 3350) is a laxative primarily used for short-term treatment of constipation. The global market for PEG 3350 was valued at approximately $1.4 billion in 2022. The compound is available under various brand names, such as MiraLAX in the United States and Molaxole in Europe. The market is driven by the prevalence of gastrointestinal disorders, aging populations, and the acceptance of PEG 3350 as a first-line treatment.
Regional markets differ substantially: North America accounts for around 40% of sales, driven by high healthcare expenditure and awareness. Europe contributes approximately 25%, with steady growth due to aging demographics. Asia-Pacific represents about 20%, with rapid expansion expected due to increasing healthcare access and urbanization. Latin America and Middle East/Africa together account for the remaining 15%.
What Are the Key Market Drivers and Constraints?
Drivers
- Rising incidence of constipation linked to aging populations and lifestyle factors.
- Favorable safety profile of PEG 3350 compared to stimulant laxatives.
- Preference for OTC remedies, reducing healthcare consultation costs.
- Ongoing research supporting PEG 3350's efficacy and tolerability.
Constraints
- Availability of alternative treatments like fiber supplements and other laxatives.
- Regulatory concerns over manufacturing standards in certain countries.
- Patent expiries leading to generic competition, impacting pricing and margins.
- Limited use in pediatric populations due to safety concerns, constraining market expansion.
How Do Sales Trends Evolve in the Near Term?
Sales are projected to grow at a compound annual growth rate (CAGR) of 3-4% from 2023 to 2028. Growth is expected to be steady in North America and Europe, driven by marketed drugs' existing presence. The Asia-Pacific region will see increased adoption owing to improved healthcare access, with projections indicating a CAGR of 5%.
Market expansion is also supported by the development of formulations with improved dosing schedules and flavor masking, increasing patient adherence. Additionally, the entry of generic PEG 3350 products post-patent expiration around 2020 has intensified price competition, keeping average selling prices stable or slightly declining.
What Is the Competitive Landscape in PEG 3350 Market?
Major companies include:
- BASF: Produces PEG 3350 as an excipient, with a significant share in the OTC laxative segment.
- Fresenius Kabi: Manufactures branded PEG 3350 powders and solutions.
- Hadvak (subsidiary of Bausch Health): Offers MiraLAX, a leading OTC product.
- Sandoz (Novartis division): Supplies generic PEG 3350 formulations.
Market dynamics are characterized by:
- High reliance on branded formulations in North America.
- Growing market penetration of generics in Europe and Asia-Pacific.
- R&D activities aimed at new delivery systems, such as liquid suspensions and ready-to-drink formats.
What Are Future Opportunities and Challenges?
Opportunities
- Development of pediatric-specific PEG 3350 formulations.
- Expansion into emerging markets with improvements in distribution infrastructure.
- Novel formulations with enhanced palatability and dosing flexibility.
- Potential for combination therapies addressing related gastrointestinal disorders.
Challenges
- Competition from newer laxatives, fiber-based products, and digital therapeutics.
- Stringent regulatory standards impacting manufacturing.
- Price erosion from increasing generic competition.
What Are Key Data Points in Sales Projections?
| Year | Global Market Size (USD billion) | Growth Rate (CAGR) | Key Regions |
|---|---|---|---|
| 2022 | 1.4 | — | North America, Europe, Asia-Pacific |
| 2023 | 1.45 | 3.6% | |
| 2024 | 1.52 | 4.8% | |
| 2025 | 1.58 | 4.2% | |
| 2026 | 1.65 | 4.2% | |
| 2027 | 1.72 | 4.2% | |
| 2028 | 1.78 | 3.5% |
(Note: Projections assume steady demand growth, with increased generic penetration post-patent expiry and ongoing development efforts.)
What Are Critical Regulatory Considerations?
Regulatory bodies such as the FDA, EMA, and China NMPA require compliance with Good Manufacturing Practices (GMP). Labeling updates, safety data, and manufacturing standards influence market access. Regulatory delays can hamper product launches, especially in emerging markets.
Final Summary
The PEG 3350 market demonstrates stable growth driven by aging populations and the product's safety profile. Market expansion will depend on improved formulations and entering emerging markets, but faced with challenges from competition and regulation. Companies focusing on innovation, supply chain optimization, and regional expansion will position best for sustained growth.
Key Takeaways
- The global PEG 3350 market was valued at $1.4 billion in 2022.
- Expected CAGR from 2023–2028 is approximately 3.5–4.2%.
- North America leads the market, with Europe and Asia-Pacific showing significant growth potential.
- Generic competition and regulatory compliance are primary factors influencing pricing and availability.
- Opportunities lie in pediatric formulations, emerging markets, and novel delivery systems.
FAQs
1. What are the primary markets for PEG 3350?
North America, Europe, and Asia-Pacific.
2. When do patent expirations influence the market?
Most key patents expired around 2020, leading to increased generic products.
3. How does PEG 3350 compare to other laxatives?
It has a favorable safety profile and is preferred for chronic use over stimulant laxatives.
4. What regulatory challenges exist for PEG 3350?
Manufacturing standards, safety labeling, and regional approval processes.
5. Is there potential for new PEG 3350 formulations?
Yes; reformulations improving taste, dosing, and pediatric suitability are being developed.
References
[1] Market research reports, 2022.
[2] FDA and EMA regulatory guidelines.
[3] Company filings and press releases.
More… ↓
