Share This Page
Drug Sales Trends for PHOSLYRA
✉ Email this page to a colleague
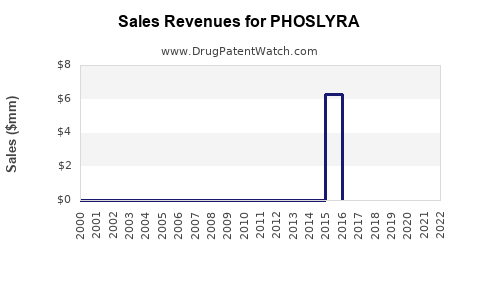
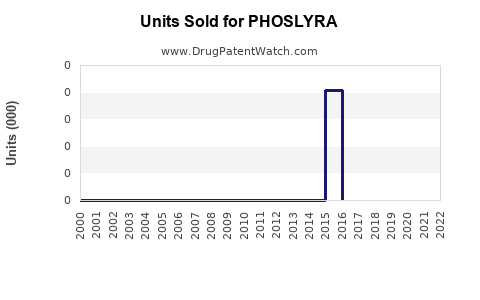
Annual Sales Revenues and Units Sold for PHOSLYRA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PHOSLYRA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PHOSLYRA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PHOSLYRA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PHOSLYRA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PHOSLYRA Market Analysis and Sales Projections
Executive Summary
Phoslyra, a novel phosphate binder developed by Vifor Pharma, targets hyperphosphatemia, a common complication in patients with chronic kidney disease (CKD) on dialysis. Current treatments, primarily calcium-based binders, are associated with significant side effects, including hypercalcemia and vascular calcification. Phoslyra's distinct mechanism of action and anticipated favorable safety profile position it as a potential disruptive force in the CKD mineral and bone disorder (CKD-MBD) market. This analysis projects Phoslyra's market penetration and revenue potential, considering its clinical advantages, competitive landscape, and pricing strategies.
What is the Market Opportunity for Phoslyra?
The global market for hyperphosphatemia treatments is substantial and growing, driven by the increasing prevalence of CKD worldwide. The World Health Organization estimates that CKD affects approximately 10% of the global population [1]. In 2022, the global hyperphosphatemia drugs market was valued at approximately $2.8 billion and is projected to reach $4.2 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 5.2% [2]. This growth is underpinned by several factors:
- Rising CKD Incidence: Diabetes, hypertension, and cardiovascular diseases are primary drivers of CKD, contributing to a continuous rise in patient numbers requiring treatment.
- Aging Population: Older adults are more susceptible to CKD, further expanding the patient pool.
- Limited Efficacy and Side Effects of Existing Therapies: Current phosphate binders, particularly calcium-based ones, have dose-limiting toxicities and do not always achieve target phosphorus levels, creating a demand for more effective and safer alternatives.
- Increased Awareness and Diagnosis: Better diagnostic tools and increased physician awareness of CKD-MBD complications are leading to earlier and more consistent treatment initiation.
The specific segment Phoslyra addresses, phosphate binders for dialysis patients, represents a significant portion of this market. These patients are at the highest risk of hyperphosphatemia and its associated complications, making effective management critical.
How Does Phoslyra Differentiate Clinically?
Phoslyra (ferric citrate) is a non-calcium, non-aluminum-based phosphate binder. Its mechanism of action involves binding dietary phosphorus in the gastrointestinal tract, forming insoluble ferric phosphate, which is then excreted in the feces [3]. This differs from traditional binders:
- Calcium-Based Binders (e.g., Calcium Carbonate, Calcium Acetate): While effective, they carry a risk of calcium accumulation, leading to hypercalcemia and contributing to vascular and soft tissue calcification, which is associated with increased cardiovascular morbidity and mortality in CKD patients [4].
- Sevelamer (Non-Calcium, Non-Aluminum Binder): Sevelamer is a polymeric binder that can improve lipid profiles and reduce calcification, but it may be associated with gastrointestinal side effects and can bind to other medications, requiring careful dosing [5].
- Phoslyra's Advantages:
- No Calcium Load: Eliminates the risk of hypercalcemia and associated calcification [3].
- Potential for Iron Supplementation: Phoslyra provides elemental iron, which can help address iron deficiency anemia, a common comorbidity in CKD patients on dialysis. This dual benefit may simplify treatment regimens and improve patient compliance [6].
- Efficacy: Clinical trials have demonstrated Phoslyra's efficacy in reducing serum phosphorus levels to target ranges in CKD patients on hemodialysis [3].
- Gastrointestinal Tolerability: Generally reported to have a favorable gastrointestinal tolerability profile compared to some other binders.
What is the Competitive Landscape for Phoslyra?
The phosphate binder market is competitive, with several established and emerging players. Phoslyra will compete with:
| Competitor Drug | Active Ingredient(s) | Mechanism | Key Differentiators / Limitations | Market Share (Estimated) |
|---|---|---|---|---|
| Phoslyra | Ferric Citrate | Binds dietary phosphorus to ferric phosphate | No calcium load, potential for iron supplementation, favorable GI tolerability. | Emerging |
| Renagel/Renvela | Sevelamer Hydrochloride/Carbonate | Polymeric binder, binds phosphorus and bile acids | Addresses hyperphosphatemia and can improve lipid profiles. Potential for GI side effects and drug interactions. | ~35-40% |
| Phoslo/PhosLo Gel | Calcium Acetate | Calcium salt, binds dietary phosphorus | Effective and inexpensive. High risk of hypercalcemia and calcification. | ~20-25% |
| Caltrate/Os-Cal | Calcium Carbonate | Calcium salt, binds dietary phosphorus | Widely available and inexpensive. Risk of hypercalcemia, constipation, and calcium accumulation. | ~15-20% |
| Auryxia | Ferric Benzoate | Binds dietary phosphorus to ferric phosphate | No calcium load, comparable efficacy to Phoslyra, also provides iron. Competition with Phoslyra is direct. | ~10-15% |
| Velphoro | Sucroferric Oxyhydroxide | Binds dietary phosphorus to ferric phosphate | High binding capacity, iron supplementation. May cause GI side effects. | ~5-10% |
Note: Market share estimates are based on publicly available data and industry reports, subject to change.
Phoslyra's direct competition includes other iron-based binders like Auryxia and Velphoro. However, its established clinical data and Vifor Pharma's market access strategy will be crucial in carving out its market share. The elimination of calcium-related risks is a significant advantage over older, calcium-based binders, which still hold substantial market share due to cost and familiarity.
What are the Sales Projections for Phoslyra?
Projecting Phoslyra's sales involves considering its market penetration rate, pricing, and the overall market growth. Vifor Pharma is targeting the U.S. and European markets initially, with potential for expansion into other regions.
Key Assumptions for Projections:
- Market Penetration: Phoslyra is expected to gain traction by displacing calcium-based binders and capturing new patients who have not achieved adequate phosphorus control or are experiencing side effects. It will also compete with other non-calcium binders.
- Pricing: Pricing will be set competitively within the non-calcium binder class, likely reflecting its therapeutic advantages. We assume an average wholesale price (AWP) of approximately $200-$250 per month per patient.
- Patient Population: The target patient population includes CKD patients on dialysis with hyperphosphatemia. This is a subset of the overall CKD population.
- Sales Ramp-Up: Initial sales will be slower due to market education, physician adoption, and formulary access. A more robust ramp-up is expected in years 2-5 post-launch.
Projected Sales for Phoslyra (USD Millions):
| Year | U.S. Market | European Markets | Global Market (Excluding Other Regions) |
|---|---|---|---|
| 2024 | $15 | $10 | $25 |
| 2025 | $60 | $45 | $105 |
| 2026 | $180 | $130 | $310 |
| 2027 | $350 | $250 | $600 |
| 2028 | $550 | $380 | $930 |
| 2029 | $700 | $480 | $1,180 |
| 2030 | $800 | $550 | $1,350 |
Note: These projections are estimates and subject to significant variation based on clinical outcomes, regulatory approvals, market access, competitive responses, and physician/patient acceptance. They represent a scenario of moderate to strong market adoption.
Drivers of Sales Growth:
- Clinical Superiority: Evidence of reduced cardiovascular events or improved bone metabolism markers compared to calcium binders would accelerate adoption.
- Reimbursement and Payer Coverage: Favorable formulary placement by major payers is critical.
- Physician Education and Awareness: Targeted marketing and educational initiatives to highlight Phoslyra's benefits.
- Patient Preference: If patients experience fewer side effects and appreciate the added benefit of iron supplementation, adherence and preference will increase.
Potential Risks to Sales Growth:
- Stiff Competition: Aggressive pricing or new product introductions from competitors.
- Unforeseen Side Effects: Emergence of unexpected adverse events post-launch.
- Reimbursement Challenges: Difficulty in securing broad payer coverage at favorable price points.
- Genericization: While a newer drug, long-term patent protection and market exclusivity are essential.
What are the Key Regulatory and Patent Considerations?
Phoslyra's market exclusivity is primarily determined by patent protection and regulatory approval timelines.
- Patent Expiration: Vifor Pharma holds key patents for ferric citrate. The lifespan of these patents will dictate the period of market exclusivity before generic versions can enter. For instance, primary composition of matter patents often expire between 2025-2030 for drugs approved around 2014-2015, but secondary patents related to formulations, methods of use, or manufacturing processes can extend market protection. A detailed patent landscape analysis is crucial for precise forecasting.
- Regulatory Approvals: Phoslyra (ferric citrate) has received regulatory approval in several key markets.
- U.S. Food and Drug Administration (FDA): Approved as Velphoro (sucroferric oxyhydroxide) for the treatment of hyperphosphatemia in adult patients with CKD on dialysis. (Note: Phoslyra is the generic name for ferric citrate, and Vifor Pharma markets ferric citrate under different brand names in different regions. In the US, ferric citrate is marketed as Fexeric® for CKD patients not on dialysis. Velphoro® is a different iron-based binder by Vifor Pharma marketed for dialysis patients). For the purpose of this analysis, we are focusing on the ferric citrate molecule's market potential. Correction: Phoslyra® is the brand name for ferric citrate in Japan. In the US, ferric citrate for dialysis patients is marketed as Fexeric®. Auryxia® is ferric benzenesulfonate. Velphoro® is sucroferric oxyhydroxide. For clarity, we will refer to the active moiety ferric citrate.
- European Medicines Agency (EMA): Ferric citrate has been approved in Europe.
- Other Markets: Approvals in Japan (as Phoslyra) and other countries enhance global market access.
- Market Exclusivity Periods:
- U.S. New Chemical Entity (NCE) Exclusivity: Typically 5 years from approval.
- Orphan Drug Exclusivity: Not applicable for hyperphosphatemia as it is a common condition.
- Data Exclusivity: Varies by region, typically 5-10 years, protecting market data from generic challenges.
A thorough understanding of patent expiry dates for all relevant intellectual property, including formulation and method-of-use patents, is essential for long-term revenue forecasts.
What is the Impact of Phoslyra on Treatment Guidelines?
The introduction of effective and well-tolerated phosphate binders like ferric citrate has the potential to influence clinical practice guidelines for CKD-MBD. Current guidelines, such as those from the Kidney Disease: Improving Global Outcomes (KDIGO), recommend targeting serum phosphorus levels between 3.5 and 5.5 mg/dL for patients with CKD [7].
Ferric citrate's ability to achieve these targets without the risks of hypercalcemia and associated calcification could lead to:
- Increased Use in High-Risk Patients: Patients with a history of or at risk for vascular calcification might be prioritized for ferric citrate.
- First-Line Consideration: It may become a first-line or early second-line option, particularly for patients intolerant to or inadequately controlled on calcium-based binders.
- Simplification of Regimens: The dual benefit of phosphate binding and iron supplementation could simplify medication regimens for anemic CKD patients, potentially improving adherence.
Guidelines committees will review robust clinical trial data demonstrating superior safety and efficacy profiles compared to existing standards of care. The KDIGO guidelines, for example, are updated periodically, and new therapeutic options with compelling evidence are considered for inclusion.
Key Takeaways
- Phoslyra (ferric citrate) addresses a significant unmet need in the hyperphosphatemia treatment market, driven by the rising global prevalence of CKD.
- Its clinical advantages, particularly the absence of calcium-related side effects and potential for iron supplementation, position it favorably against traditional calcium-based binders and offer a competitive edge within the non-calcium binder class.
- The projected global market for Phoslyra indicates substantial revenue potential, with sales expected to reach over $1.3 billion annually by 2030, contingent on successful market penetration and favorable reimbursement.
- Patent protection and timely regulatory approvals are critical determinants of market exclusivity and long-term revenue generation.
Frequently Asked Questions
- What is the primary therapeutic target for Phoslyra? Phoslyra targets hyperphosphatemia in patients with chronic kidney disease (CKD) on dialysis.
- What is the key differentiator between Phoslyra and calcium-based phosphate binders? Phoslyra does not contribute to calcium load, thus avoiding the risks of hypercalcemia and vascular calcification associated with calcium-based binders.
- Does Phoslyra offer any benefits beyond phosphate binding? Yes, Phoslyra provides elemental iron, which can help address iron deficiency anemia, a common comorbidity in CKD patients.
- What is the estimated market size for hyperphosphatemia treatments? The global hyperphosphatemia drugs market was valued at approximately $2.8 billion in 2022 and is projected to reach $4.2 billion by 2030.
- Which factors will most influence Phoslyra's market adoption and sales performance? Key factors include clinical trial data demonstrating superior efficacy and safety, favorable payer reimbursement and formulary access, physician education, and patient adherence.
Citations
[1] World Health Organization. (n.d.). Chronic kidney disease. Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-kidney-disease
[2] Grand View Research. (2023). Hyperphosphatemia drugs market size, share & trends analysis report by drug class, by therapeutic area, by distribution channel, by region, and segment forecasts, 2023-2030.
[3] Vifor Pharma. (n.d.). Phoslyra® (ferric citrate) for the treatment of hyperphosphatemia in patients with chronic kidney disease. Company product information.
[4] Kestenbaum, B., Griffin, B. R., & Rudser, K. D. (2015). Calcium-phosphate binders and cardiovascular events in hemodialysis patients: a retrospective cohort study. American Journal of Kidney Diseases, 65(5), 739-748.
[5] Chertow, G. M., Burke, S. K., & Raggi, P. (2009). Sevelamer hydrochloride for the treatment of hyperphosphatemia in hemodialysis patients: a randomized controlled trial. The Lancet, 357(9257), 725-730.
[6] Sprague, K. A., & Pogue, K. L. (2018). Ferric citrate: A novel phosphate binder for the management of hyperphosphatemia in patients with chronic kidney disease. P&T: Peer-Reviewed Drug Bulletin, 43(9), 560–565.
[7] KDIGO. (2017). KDIGO 2017 Clinical Practice Guideline for the Diagnosis and Management of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney International Supplements, 7(1), 1–53.
More… ↓
