Share This Page
Drug Sales Trends for PHENYTOIN
✉ Email this page to a colleague
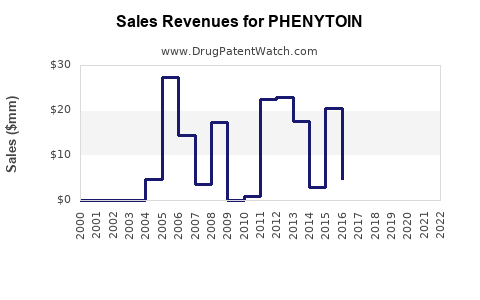
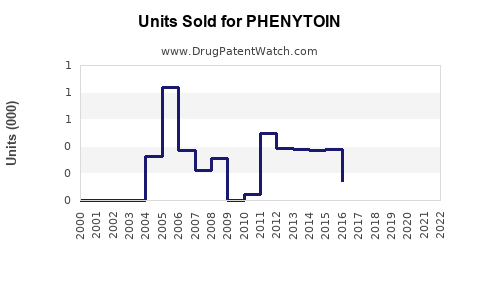
Annual Sales Revenues and Units Sold for PHENYTOIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PHENYTOIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PHENYTOIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PHENYTOIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PHENYTOIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PHENYTOIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PHENYTOIN Market Analysis and Financial Projection
What Is the Market Size for Phenytoin?
Phenytoin is a hydantoin derivative used primarily as an anticonvulsant for seizure management. Global epilepsy market size was valued at approximately $5.4 billion in 2021, with phenytoin accounting for an estimated 10-15% of this market, translating to roughly $540 million to $810 million annually. The drug's market share is influenced by its longstanding history, availability as a generic, and competition from newer antiepileptics.
How Does the Current Production and Patent Status Affect Market Dynamics?
Phenytoin is off patent and widely available as generic formulations. Patent expirations date back to the early 2000s, resulting in increased competition that maintains low prices and limits profit margins for branded versions. The absence of patent barriers allows producers to compete primarily on price and manufacturing capacity.
Who Are the Key Market Players?
Major producers include:
- Mylan (now part of Viatris)
- Teva Pharmaceuticals
- Pfizer (USP formulation)
- Hikma Pharmaceuticals
- Sandoz (Novartis)
These companies supply both injectible and oral formulations. Limited innovation and high generic competition stabilize prices but restrict revenue growth.
What Are the Sales Drivers and Market Trends?
Main drivers include:
- The prevalence of epilepsy worldwide, estimated at 50 million cases (WHO 2019).
- The drug's effectiveness in controlling seizures in partial and generalized epilepsy.
- Developments in emergency medicine protocols where injectable phenytoin is used for status epilepticus.
Market trends show sustained demand, especially in regions with limited access to newer agents. However, the introduction of newer anticonvulsants with improved safety profiles—such as levetiracetam and lamotrigine—has led to declining use of phenytoin in some settings.
What Are the Future Sales Projections?
Global phenytoin sales are projected to decline modestly over the next five years due to:
- Increasing adoption of newer antiepileptics.
- Growing awareness of phenytoin's side-effect profile, including gingival hyperplasia and ataxia.
- Pharmaceutical industry focus shifting toward branded or patented formulations with novel delivery mechanisms.
Forecast estimates suggest that global sales will decrease at an average compound annual growth rate (CAGR) of 1-2% from 2023 to 2028, constraining total revenue to approximately $700 million by 2028.
What Are the Regional Market Dynamics?
- North America: Mature market with stable demand; key for injectable formulations.
- Europe: Similar to North America but experiencing shifts toward newer drugs.
- Asia-Pacific: Fast-growing, with increasing epilepsy diagnoses; however, price sensitivity favors low-cost generics like phenytoin.
- Latin America and Africa: Limited access to newer drugs increases dependence on phenytoin.
How Is Regulatory Environment Influencing Market Growth?
Regulatory agencies permit the continued use of phenytoin as an essential medicine but impose strict quality standards. The Drug Price Competition and Patent Term Restoration Act (Hatch-Waxman Act) has facilitated generic entry, keeping prices low. No significant regulatory barriers are forecasted to impact supply.
What Are the Challenges and Opportunities?
Challenges:
- Side effect profile leading to alternative therapies.
- Competitive pressure from newer, safer drugs.
- Limited innovation in formulation.
Opportunities:
- Expansion into developing markets.
- Development of controlled-release formulations.
- Repurposing for off-label uses, pending clinical validation.
Key Takeaways
- The global phenytoin market stood around $540-$810 million in 2021, with slight decline expectations.
- Off-patent status fosters intense generic competition, suppressing prices.
- Growth projections indicate a slight decrease due to market shift toward newer antiepileptics.
- Regional variations affect demand, with emerging markets showing increasing reliance.
- Innovation focus remains limited; price and accessibility are primary drivers.
FAQs
1. Will the phenytoin market recover or expand in the future?
Market growth is limited by the availability of newer anticonvulsants. Demand is expected to decline gradually, primarily in regions adopting alternative therapies.
2. Are there any ongoing developments to improve phenytoin formulations?
Limited innovation has been reported. Some companies explore extended-release formulations to improve tolerability but are not widely commercialized.
3. How does the side-effect profile impact sales?
Adverse effects such as gingival hyperplasia and neurologic issues lead clinicians to prefer newer drugs, reducing phenytoin's market share.
4. What geographic markets present the best opportunities?
Emerging markets with limited access to branded and newer medications, such as parts of Africa and Asia, offer growth opportunities for cost-effective generic phenytoin.
5. Is there regulatory risk that could affect supply?
Currently minimal; phenytoin is an essential medicine with a stable regulatory environment. Changes are unlikely to impact existing manufacturing or sales drastically.
References
[1] World Health Organization. Epilepsy Fact Sheet. 2019.
[2] GlobalData. Antiepileptic Drugs Market Analysis. 2022.
[3] IQVIA. Pharmaceutical Market Trends Report. 2022.
More… ↓
