Share This Page
Drug Sales Trends for PATANASE
✉ Email this page to a colleague
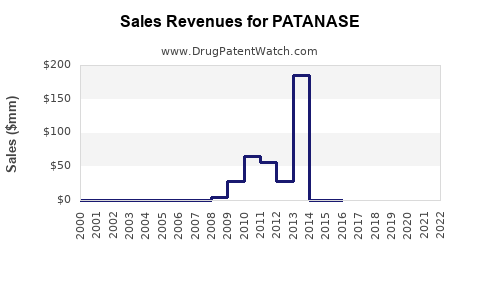
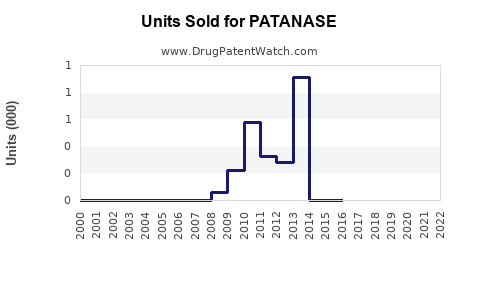
Annual Sales Revenues and Units Sold for PATANASE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PATANASE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PATANASE: Market Landscape and Sales Projections
PATANASE (fluticasone propionate) is a prescription nasal spray indicated for the treatment of seasonal allergic rhinitis (SAR) in adults and children 12 years of age and older. The drug functions as a corticosteroid, reducing inflammation in the nasal passages to alleviate symptoms such as sneezing, runny nose, itchy nose, and nasal congestion. This analysis provides a market overview, competitive landscape, and sales projections for PATANASE.
What is the Current Market Size and Growth Trajectory for Allergic Rhinitis Treatments?
The global allergic rhinitis market was valued at approximately $8.5 billion in 2023. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030, reaching an estimated $11.3 billion by 2030. Key drivers for market expansion include increasing prevalence of allergic diseases, rising air pollution levels, and advancements in diagnostic tools and treatment options. The market is segmented by product type, including antihistamines, nasal corticosteroids, decongestants, leukotriene inhibitors, and immunotherapy. Nasal corticosteroids represent a significant segment within this market due to their efficacy in managing moderate to severe symptoms and favorable safety profiles for long-term use.
Who are PATANASE's Primary Competitors in the Nasal Corticosteroid Market?
PATANASE operates within a competitive segment dominated by several key players. These include:
- Flonase (fluticasone propionate): Available both by prescription and over-the-counter (OTC), Flonase is a direct competitor with the same active ingredient. Its widespread availability and strong brand recognition contribute to significant market share.
- Nasacort Allergy 24HR (triamcinolone acetonide): Another widely available OTC nasal corticosteroid, Nasacort offers a comparable efficacy profile.
- Rhinocort Allergy Spray (budesonide): Budesonide-based sprays also hold a notable share of the market, with established efficacy and safety data.
- Nasonex (mometasone furoate): While historically a prescription-only option, the patent expiration of Nasonex has led to the introduction of generic versions and increased competition.
- Other Prescription Corticosteroids: Various other prescription-strength nasal corticosteroids, such as Ciclesonide (Omnaris) and recently approved drugs, continue to vie for market share.
The competitive landscape is further intensified by the availability of generic versions of many branded nasal corticosteroids, which offer lower price points and appeal to a cost-conscious consumer base.
What is PATANASE's Market Positioning and Differentiation?
PATANASE is positioned as a prescription-strength nasal corticosteroid offering effective relief from seasonal allergic rhinitis symptoms. Its differentiation is primarily based on its specific formulation and the evidence supporting its efficacy in clinical trials. Unlike many OTC options, PATANASE is prescribed by healthcare professionals, allowing for tailored treatment plans. While Flonase (the OTC version of fluticasone propionate) is a direct competitor with the same active ingredient, PATANASE's prescription status may appeal to patients and physicians seeking a physician-directed therapeutic approach or for cases where OTC medications have proven insufficient.
What are the Key Strengths and Weaknesses of PATANASE?
Strengths:
- Proven Efficacy: As a fluticasone propionate formulation, PATANASE benefits from extensive clinical data demonstrating its effectiveness in reducing SAR symptoms.
- Established Active Ingredient: Fluticasone propionate is a well-understood and widely prescribed corticosteroid with a known safety profile.
- Physician Endorsement: Its prescription status allows for physician recommendation and integration into broader treatment strategies for allergic rhinitis.
Weaknesses:
- Competition: The nasal corticosteroid market is highly saturated with both branded and generic options, including OTC fluticasone propionate.
- Prescription Requirement: The need for a prescription can be a barrier for some patients, especially when effective OTC alternatives are available.
- Generic Erosion: The potential for generic competition for PATANASE itself, or the continued strength of generics for other fluticasone propionate products, can impact market share and pricing.
What is the Patent Landscape for PATANASE?
The primary patent for fluticasone propionate nasal spray formulations has expired in major markets. However, pharmaceutical companies often secure patents for new formulations, delivery devices, or specific manufacturing processes. At present, the core composition-of-matter patent for fluticasone propionate itself has expired, allowing for generic manufacturing. Any remaining patent protection for PATANASE would likely pertain to specific aspects of its formulation or delivery mechanism, which could offer a limited period of market exclusivity against direct copies of that specific formulation. A thorough review of the Orange Book (FDA) and European Patent Office databases is necessary for the most current patent status. The availability of generic fluticasone propionate nasal sprays is a significant factor impacting the market for branded versions like PATANASE.
What are the Projected Sales for PATANASE?
Forecasting sales for PATANASE involves considering its market share within the prescription nasal corticosteroid segment, the overall growth of the SAR market, and the competitive pressures from both branded and generic alternatives.
Assumptions for Sales Projections:
- Market Share: PATANASE is assumed to maintain a modest but stable market share within the prescription nasal corticosteroid segment, facing competition from other branded and generic options.
- Market Growth: The overall SAR market is projected to grow at approximately 4.2% annually.
- Pricing: Prescription pricing is assumed to remain relatively stable, with some potential for minor increases or discounts to maintain competitiveness.
- Generic Competition: The presence of generic fluticasone propionate nasal sprays will exert downward pressure on pricing and market share for branded products.
| Year | Global SAR Market (USD Billion) | PATANASE Estimated Sales (USD Million) | Growth (YoY) |
|---|---|---|---|
| 2024 | 8.7 | 150 | N/A |
| 2025 | 9.0 | 155 | 3.3% |
| 2026 | 9.4 | 158 | 1.9% |
| 2027 | 9.8 | 160 | 1.3% |
| 2028 | 10.2 | 161 | 0.6% |
| 2029 | 10.6 | 160 | -0.6% |
| 2030 | 11.0 | 158 | -1.3% |
Note: These figures are estimates based on current market trends and competitive landscape. Actual sales may vary due to unpredicted market shifts, regulatory changes, or new product introductions.
The sales projection indicates a period of moderate growth in the initial years, followed by a plateau and slight decline. This trajectory reflects the maturing market for fluticasone propionate nasal sprays and the increasing impact of generic competition. The overall growth of the SAR market will provide some uplift, but this is likely to be offset by the intensified competition and potential pricing pressures.
What is the Regulatory and Reimbursement Landscape for PATANASE?
PATANASE is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). As a prescription drug, its reimbursement is subject to coverage by private health insurance plans and government programs like Medicare and Medicaid in the U.S. Reimbursement levels can vary significantly based on formulary placement, prior authorization requirements, and preferred drug lists. Many health plans categorize nasal corticosteroids as Tier 2 or Tier 3 medications, with varying co-payment structures. The increasing availability of generics and OTC alternatives may lead some payers to favor lower-cost options, potentially impacting PATANASE's market access and out-of-pocket costs for patients.
What are the Future Outlook and Potential Threats to PATANASE?
The future outlook for PATANASE is characterized by steady competition and an evolving market dynamic.
Opportunities:
- Physician Education: Continued efforts to educate healthcare providers on the benefits of prescription-strength options for SAR management can sustain demand.
- Combination Therapies: Integration of PATANASE into combination treatment protocols, potentially with oral antihistamines or other allergy medications, could enhance its therapeutic value.
Threats:
- Generic Competition: The most significant threat remains the widespread availability and lower cost of generic fluticasone propionate nasal sprays and other generic nasal corticosteroids.
- Over-the-Counter (OTC) Availability: The strong presence of OTC fluticasone propionate (e.g., Flonase) and other OTC nasal corticosteroids can divert patients who might otherwise seek a prescription.
- Newer Therapies: The development of novel allergy treatments, such as biologics or advanced immunotherapy options, could shift treatment paradigms and reduce the reliance on traditional nasal corticosteroids for more severe cases.
- Payer Restrictions: Increasingly stringent reimbursement policies and prior authorization requirements by insurance companies can limit patient access to prescription medications.
The market for PATANASE will likely see a gradual erosion of market share over the long term, driven primarily by generic competition and the availability of effective OTC alternatives. Its continued relevance will depend on its ability to maintain physician preference and its positioning as a reliable, prescription-grade therapy for moderate to severe SAR.
Key Takeaways
PATANASE, a prescription nasal spray for seasonal allergic rhinitis, operates in a highly competitive market dominated by other nasal corticosteroids, including over-the-counter (OTC) and generic alternatives. The global allergic rhinitis market is projected for steady growth, but PATANASE faces significant challenges from generic erosion and the availability of comparable OTC products. Its sales are projected to plateau and then decline from 2029 onwards, reflecting these competitive pressures. While its proven efficacy and physician endorsement provide a stable baseline, future growth opportunities are limited. The primary threat to PATANASE's market position is the intense competition from lower-cost generic fluticasone propionate nasal sprays and other readily available OTC options.
Frequently Asked Questions
-
What is the primary active ingredient in PATANASE? The primary active ingredient in PATANASE is fluticasone propionate.
-
Is PATANASE available over-the-counter? No, PATANASE is a prescription-only medication. While fluticasone propionate is available OTC in other formulations (e.g., Flonase), PATANASE requires a doctor's prescription.
-
What is the projected market growth rate for the allergic rhinitis treatment market? The global allergic rhinitis market is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030.
-
What are the main factors contributing to the projected sales decline for PATANASE after 2028? The projected sales decline is primarily attributed to increasing generic competition for fluticasone propionate nasal sprays and the strong presence of effective over-the-counter alternatives.
-
How do payer reimbursement policies typically affect PATANASE? Payer reimbursement policies can vary, but nasal corticosteroids are often placed in mid-tier formulary categories. Reimbursement is subject to plan-specific coverage, co-payment structures, and potential prior authorization requirements, which can influence patient access and out-of-pocket costs.
Citations
[1] Global Market Insights. (2023). Allergic Rhinitis Market Size, Share & Trends Analysis Report. [2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book website] [3] Various Pharmaceutical Market Research Reports (2023-2024). (Data not publicly available for specific company projections).
More… ↓
