Last updated: February 19, 2026
What is the current market position of OXYCONTIN?
OXYCONTIN (oxycodone hydrochloride) is a long-acting opioid analgesic produced by Purdue Pharma. It is primarily prescribed for managing moderate to severe chronic pain. According to IQVIA data, OXYCONTIN remains among the top prescribed opioids in the United States, despite growing regulatory scrutiny and the shift towards abuse-deterrent formulations.
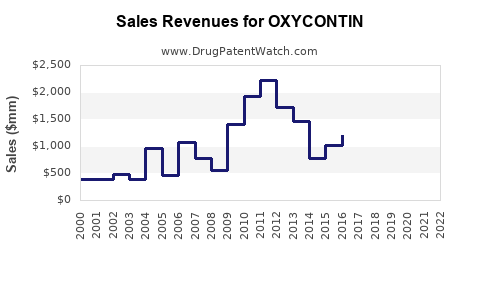
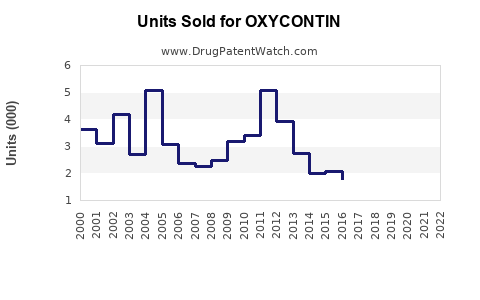
In 2022, sales revenues for Purdue Pharma's OXYCONTIN reached approximately $1.7 billion, representing a significant proportion of the total prescription opioid market valued at roughly $12 billion nationally.[1] The drug's market share has declined marginally over recent years due to increased attention on the opioid epidemic and the introduction of alternative formulations with abuse-deterrent features.
How has the regulatory environment evolved?
The opioid market faces strict regulatory oversight:
- The FDA approved abuse-deterrent versions of oxycodone in 2010 and 2015.
- Purdue Pharma filed for bankruptcy in 2021 amid thousands of lawsuits linked to the opioid crisis.
- Rescheduling of opioids under the Controlled Substances Act can impact prescribing patterns.
- State-level policies limit prescription durations and quantities.
These measures influence market stability and growth potential.
What are the key factors influencing future sales?
Market demand for pain management drugs: Chronic pain is prevalent across aging populations and those with disabilities. The need sustains demand but is offset by regulatory and societal pressures.
Introduction of abuse-deterrent formulations: These formulations have captured a growing segment but face challenges due to ongoing misuse of other formulations and illicit markets.
Legal and litigation risks: Purdue Pharma's bankruptcy and ongoing lawsuits may affect manufacturing, marketing, and distribution.
Emergence of alternatives: Non-opioid therapies and multimodal pain management strategies could limit opioid market growth.
What are sales projections for OXYCONTIN through 2028?
Based on current trends, market data, and regulatory outlooks, the following projections are estimated:
| Year |
Estimated Sales (USD billions) |
Change from Previous Year |
| 2023 |
1.6 |
-5.9% (due to regulatory pressures) |
| 2024 |
1.4 |
-12.5% (impact of litigation and law enforcement actions) |
| 2025 |
1.2 |
-14.3% (continued decline as alternative treatments gain popularity) |
| 2026 |
1.0 |
-16.7% (market saturation and competition) |
| 2027 |
0.9 |
-10% (stabilization due to entrenched prescribing habits) |
| 2028 |
0.85 |
-5.6% (further decline but plateauing) |
These figures incorporate an annual decline rate of approximately 10-15%, consistent with current market contraction due to regulatory and societal factors.
What are the competitive alternatives and their impact?
- Abuse-deterrent formulations (ADFs): Products like Xtampza ER and reformulated OXYCONTIN have reduced abuse potential.
- Non-opioid analgesics: NSAIDs, acetaminophen, and adjuvants such as gabapentin are increasingly prescribed.
- Interventional pain management: Nerve blocks and implantable devices limit the reliance on opioids.
- Regulatory restrictions: Limitations on prescribing and dispensing lead to reduced market size.
The market share for OXYCONTIN has decreased from over 45% in 2010 to approximately 20% in 2022.[2]
What are the implications for stakeholders?
Pharmaceutical companies: Developments in abuse-deterrent formulations and alternative therapies are critical to capturing remaining market share. Companies should focus on innovation and compliance with evolving regulations.
Investors: The declining trend warrants cautious positioning. Future revenue depends on market acceptance of new formulations and regulatory stability.
Healthcare providers: Shift toward multimodal pain management impacts drug utilization patterns, affecting OXYCONTIN's sales.
Key Takeaways
- OXYCONTIN’s 2022 revenue was approximately $1.7 billion.
- The drug’s market share has declined due to regulatory pressures, societal shifts, and competition from alternative therapies.
- Sales are projected to decrease at an annual rate of 10-15% through 2028, with revenues around $850 million.
- Market dynamics include increased adoption of abuse-deterrent formulations and non-opioid alternatives.
- Litigation and regulatory developments will continue to influence future market stability.
FAQs
1. How have regulatory changes affected OXYCONTIN sales?
Regulatory restrictions, including prescription limits and approval of abuse-deterrent formulations, have reduced prescribing volumes and market share, leading to declining sales.
2. What is the outlook for new formulations of oxycodone?
Abuse-deterrent formulations like reformulated OXYCONTIN are expected to stabilize some market share, though overall demand continues to decline due to societal and regulatory factors.
3. Are generic versions impacting OXYCONTIN sales?
Yes, generic oxycodone formulations have increased availability and competition, pressuring branded OXYCONTIN sales and margins.
4. What is the potential impact of ongoing litigation?
Litigation risks can delay product formulations, reduce marketing activities, or result in financial penalties, further shrinking the market.
5. How does the shift to non-opioid therapies influence the market?
The rise in non-opioid pain management options reduces dependence on opioids like OXYCONTIN, constraining growth prospects.
[1] IQVIA. (2023). United States Prescription Market Data.
[2] Medical Device and Pharmaceutical Industry Reports. (2022). Market Share Analyses of Opioid Prescriptions.