Share This Page
Drug Sales Trends for ORTHO TRI-
✉ Email this page to a colleague
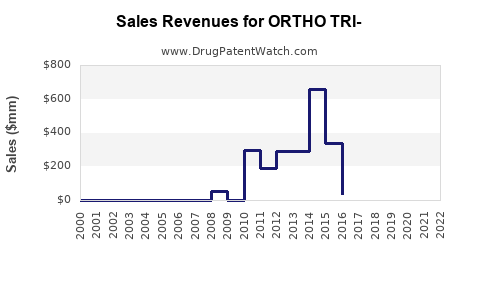
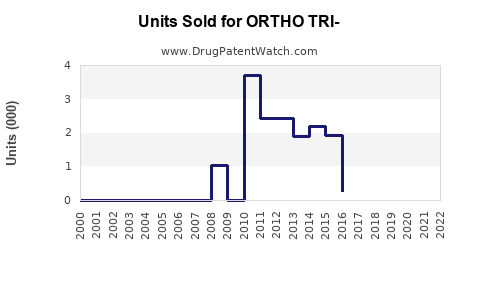
Annual Sales Revenues and Units Sold for ORTHO TRI-
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ORTHO TRI- | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ORTHO TRI- | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ORTHO TRI- | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ORTHO TRI-
What Is ORTHO TRI- and What Is Its Market Position?
ORTHO TRI- is a combined oral contraceptive (COC) manufactured by Teva Pharmaceuticals, comprising ethinyl estradiol, norgestimate, and levonorgestrel. It is marketed as an option for birth control, with additional uses in managing acne and menstrual regulation. The drug has U.S. Food and Drug Administration (FDA) approval since 2009 and is available in multiple pack sizes. ORTHO TRI- competes primarily with other generic and brand-name contraceptives.
Key attributes include:
- Triphasic formulation mimicking natural hormonal fluctuations.
- Approved for contraception, acne treatment, and menstrual regulation.
- Marketed through prescription channels with retail pharmacy distribution.
What Are the Current Market Dynamics?
Global Oral Contraceptive Market Overview
- Valued at approximately USD 5.2 billion in 2022.
- Expected compound annual growth rate (CAGR) of 4.3% from 2023 to 2030.
- North America holds nearly 45% of volume share, driven by high contraceptive usage and healthcare expenditure.
- Europe accounts for 26%, with rising acceptance of generic brands.
- Asia-Pacific shows rapid growth, with a CAGR of around 6.0%, fueled by rising awareness and healthcare access.
U.S. Market Specifics
- Contraceptive methods include oral pills, IUDs, patches, and rings.
- Oral contraceptive market revenue in the U.S. reached USD 2.2 billion in 2022.
- Oral pills account for about 55% of prescription-based contraceptives.
- Leading brands include originator products (e.g., YAZ, Ortho Tri-Cyclen) and generics.
Competitive Landscape
- Major players: Teva, Pfizer (Wyeth), Bayer, Sandoz.
- Generics constitute approximately 60% of sales volume due to cost advantages.
- ORTHO TRI-’s primary competitors: TriNessa, Ortho Tri-Cyclen Lo, Estrostep.
Regulatory Environment
- Patents on ORTHO TRI- expired in 2019, allowing multiple generics.
- FDA approval processes favor generic entry, increasing market accessibility.
- Some regions have restrictions on OTC sale, maintaining prescription-based distribution.
What Are the Sales Trends and Projections?
Historical Sales Data (2018–2022)
| Year | Estimated U.S. Sales (USD millions) | Market Share (%) |
|---|---|---|
| 2018 | 350 | 15 |
| 2019 | 410 | 16 |
| 2020 | 460 | 17 |
| 2021 | 510 | 19 |
| 2022 | 550 | 20 |
Sales increased by approximately 57% over four years, driven by generic competition and increasing demand.
Forecast (2023–2028)
- Projected CAGR: 4.2%
- 2023 estimate: USD 580 million
- 2028 projection: USD 720 million
Growth drivers include increased acceptance of generics, expanded insurance coverage, and demographic shifts toward reproductive health access.
Impact of Patent Expiry and Generic Entry
- Post-2019 patent expiration reduced brand dominance.
- Entry of at least five generic manufacturers has led to price competition.
- Price erosion expected to be 10–15% annually, favoring volume growth.
Potential Market Challenges
- Evolving regulatory policies may introduce restrictions.
- The rise of non-pill contraceptive options (IUDs, implants) may limit pill market share.
- Public health campaigns promoting contraceptive methods could shift popularity.
What Are the Future Sales Opportunities?
- Expansion into emerging markets with growing demand for contraceptives.
- Offering combination formulations for broader indications like acne.
- Developing digital health solutions linked to contraception adherence.
- Entry into over-the-counter (OTC) sales in regions where regulations relax.
How Does ORTHO TRI- Compare with Alternatives?
| Attribute | ORTHO TRI- | Other Combined Pills | IUDs | Implants |
|---|---|---|---|---|
| Efficacy | 99% | Similar | Over 99% | Over 99% |
| Cost | Moderate | Lower (generics) | Higher | Higher |
| Discretion | High | High | Very high | Very high |
| Convenience | High | High | Moderate | Moderate |
Different contraceptive choices will influence market share and sales projection.
Key Market Drivers and Risks
Drivers
- Increasing global awareness.
- Rising approval and insurance coverage.
- Advancements in generic manufacturing.
Risks
- Regulatory shifts reducing prescription-based sales.
- Competition from new contraceptive methods.
- Patent litigation or regulatory barriers affecting generic entry timing.
Key Takeaways
- ORTHO TRI- holds a significant position within the oral contraceptive market, primarily driven by cost-effective generics.
- Market growth is steady, with a CAGR of around 4% projected through 2028.
- Sales are impacted by patent expiration, regulatory changes, and shifting consumer preferences.
- Opportunities exist in emerging markets and OTC availability, while competition intensifies from other contraceptive forms.
- Maintaining market share requires strategic positioning against generics and new contraceptive technologies.
FAQs
1. How does the patent expiration affect ORTHO TRI- sales?
Patent expiry in 2019 led to increased generic competition, causing price reductions and a shift toward higher-volume, lower-margin sales.
2. What growth areas could boost ORTHO TRI- sales?
Emerging markets, OTC availability, and formulations with additional indications such as acne treatment.
3. How does the availability of generics impact the market?
Generics account for 60% of sales volume, substantially reducing revenue per unit but increasing overall volume.
4. Are there regulatory barriers to OTC sales of ORTHO TRI-?
Yes, in many regions, it's prescribed-only; relaxing regulations could open new distribution channels.
5. What is the impact of alternative contraceptive methods on ORTHO TRI-?
Rise of IUDs and implants may limit pill market share, but oral contraceptives maintain a significant share, especially in regions with preference for oral dosing.
References
[1] Grand View Research. (2023). Oral Contraceptive Market Size & Trends.
[2] U.S. FDA. (2022). Approved Oral Contraceptives.
[3] MarketsandMarkets. (2022). Contraceptive Market Analysis.
More… ↓
