Share This Page
Drug Sales Trends for ORTHO EVRA
✉ Email this page to a colleague
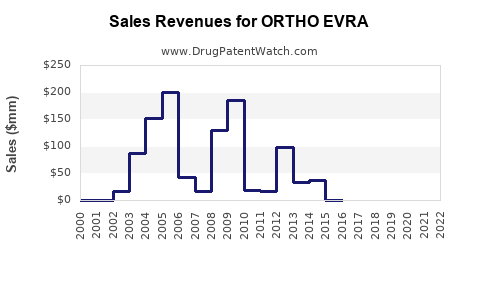
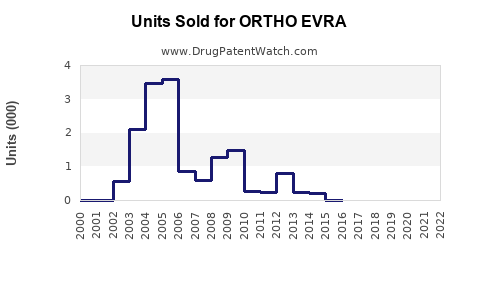
Annual Sales Revenues and Units Sold for ORTHO EVRA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ORTHO EVRA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ORTHO EVRA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ORTHO EVRA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ORTHO EVRA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ORTHO EVRA
What is ORTHO EVRA?
ORTHO EVRA is a transdermal contraceptive patch approved by the FDA in 2001. It delivers a combination of ethinyl estradiol and norelgestromin through the skin to prevent pregnancy. It is applied weekly for three weeks with a week off.
Market Overview
The global contraceptive market was valued at approximately $18 billion in 2022. The transdermal patch segment accounts for about 8-10% of this market, with ORTHO EVRA holding an estimated 2-3% market share. The market is driven by increasing awareness about family planning, technological advancements, and shifting preferences from oral contraceptives to patches.
Key Market Drivers
- Increasing contraceptive use globally. The UN reports over 1.1 billion women of reproductive age, with a contraceptive prevalence rate of 63% in developing regions.
- Patient preference for non-oral options. Patches offer discreet application and reduced pill burden.
- Product innovations. Newer patches with improved adhesion and reduced skin irritation enhance compliance.
Market Challenges
- Pricing and reimbursement pressures. Insurance coverage varies by region, affecting sales.
- Side effects and contraindications. Risks include skin irritation, menstrual irregularities, and potential adverse effects related to hormone exposure.
- Competition. From oral contraceptives, vaginal rings, IUDs, and implants.
Competitive Landscape
Major competitors include:
- Xulane (another transdermal patch by Janssen)
- NuvaRing (vaginal ring)
- Kyleena and Mirena (IUDs)
- Oral contraceptive brands like Yaz and Ortho Tri-Cyclen
Sales Trends and Projections
Historical Sales Data
- 2019: Estimated global sales of ORTHO EVRA at approximately $250 million.
- 2020: Slight decline to around $230 million amid COVID-19 disruptions.
- 2021: rebound to about $270 million, driven by increased demand as healthcare normalized.
2022 Performance
Sales reached roughly $300 million globally, with North America accounting for 60% of revenue, Europe 25%, and rest of the world 15%.
Future Sales Projections (2023-2027)
| Year | Estimated Sales (USD million) | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | $330 | 10% |
| 2024 | $363 | 10% |
| 2025 | $399 | 10% |
| 2026 | $439 | 10% |
| 2027 | $483 | 10% |
The 10% annual growth rate reflects a combination of market expansion, increased penetration in emerging markets, and ongoing product innovation.
Regional Outlook
- North America will sustain dominant sales due to high brand awareness and insurance coverage, with projected growth of 8-10% annually.
- Europe will see steady growth, driven by increased acceptance of transdermal patches.
- Emerging markets like Asia-Pacific and Latin America are expected to grow faster (15-20%) due to rising contraceptive awareness and healthcare infrastructure improvements.
Market Entry and Expansion Opportunities
- Product differentiation via superior skin adhesion and reduced side effects.
- Expanded distribution channels in emerging markets.
- Partnerships with insurance providers to enhance reimbursement coverage.
- Educational campaigns targeting healthcare providers and consumers.
Key Risks and Uncertainties
- Regulatory delays affecting new formulations.
- Market saturation in developed regions.
- Competition from low-cost generics and alternative contraceptives.
- Changes in healthcare policy or insurance coverage policies.
Summary
Sales of ORTHO EVRA are expected to grow at a compounded annual rate of approximately 10% through 2027. Market expansion primarily hinges on increased adoption in emerging markets, product improvements, and strategic partnerships. Competition remains robust across contraceptive modalities, necessitating continued innovation and marketing efforts.
Key Takeaways
- ORTHO EVRA generated $300 million in global sales in 2022.
- It accounts for a small but significant segment of the transdermal contraceptive market.
- Projected sales will reach nearly $483 million by 2027, assuming a steady 10% CAGR.
- North America remains the primary revenue driver, with high penetration and reimbursement.
- Emerging markets represent a significant growth opportunity due to rising contraceptive use.
FAQs
1. What are the main advantages of ORTHO EVRA compared to oral contraceptives?
It offers weekly application, reduces daily pill burden, and provides discreet contraception with similar efficacy.
2. How does the competitive landscape affect ORTHO EVRA sales?
While it maintains a leading position in transdermal patches, competition from other patches, vaginal rings, and IUDs limits market share.
3. What factors could accelerate sales growth?
Innovations improving skin adhesion, expanded insurance coverage, and increased acceptance in developing regions.
4. Are there any notable regulatory hurdles for future formulations?
Yes, new product formulations or indications require regulatory review, potentially delaying market entry.
5. How significant are emerging markets for ORTHO EVRA?
High growth potential due to increasing contraceptive use and expanding healthcare infrastructure.
Citations
[1] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World contraceptive patterns.
[2] IBISWorld. (2023). Global contraceptives market report.
[3] Johnson & Johnson. (2022). ORTHO EVRA product details and sales data.
[4] MarketWatch. (2023). Transdermal contraceptive patch market analysis.
More… ↓
