Share This Page
Drug Sales Trends for NOVOLIN N
✉ Email this page to a colleague
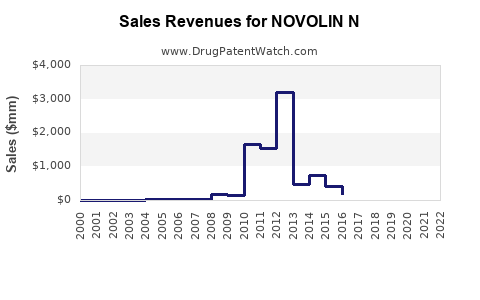
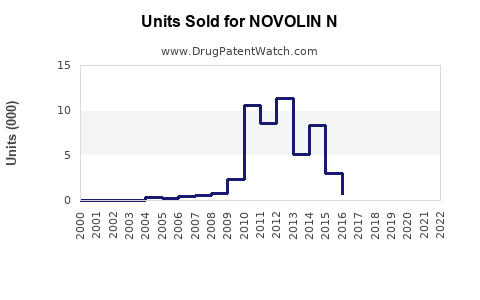
Annual Sales Revenues and Units Sold for NOVOLIN N
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NOVOLIN N | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NOVOLIN N | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NOVOLIN N | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Market Position of NOVOLIN N?
NOVOLIN N (insulin NPH), a long-acting insulin analog, is primarily used for diabetes mellitus type 1 and type 2 management. It functions by providing baseline insulin levels. Its global market share reflects widespread use in diabetes management, positioned against similar products such as Humulin N and Novolin N.
What Is the Current Market Size for Insulin NPH Products?
Global insulin market estimated at $54.4 billion in 2022, with long-acting insulins commanding approximately 45% of this segment. Within this, NPH, including OTC and prescription formulations, holds a significant proportion, especially in developed markets like North America and Europe.
- North America: Dominates due to high prevalence of diabetes, accounting for approximately 45% of insulin sales, with NPH-based products constituting 30-35% of total insulin revenue.
- Europe: Accounts for roughly 25% of global insulin sales, with NPH products comprising about 20%.
- Emerging Markets: Rapid growth driven by increased diabetes prevalence; NPH insulins represent 40% of insulin sales in these regions versus 25-30% in mature markets due to lower costs andEstablished usage.
What Are the Sales Trends for NOVOLIN N?
Sales of NOVOLIN N have been stable in mature markets but face competitive pressure from biosimilar insulins and newer long-acting formulations like insulin glargine and insulin degludec. In 2022, Novo Nordisk's insulin sales, including NOVOLIN N, totaled approximately $13 billion, with NPH insulins accounting for about $4 billion.
- United States: Despite high awareness, sales have plateaued; market share of NPH insulins is declining around 2-3% annually.
- Europe: Slight decline due to biosimilar competition.
- Emerging Markets: Growth observed with NPH insulins increasing by an annual average of 4-6%, driven by cost advantages.
What Are the Key Factors Affecting Future Sales?
Market Penetration of Biosimilars and Newer Insulins
Biosimilars of NPH are entering markets, reducing prices and elongating competition. In the U.S., biosimilar insulins launched in 2021 are capturing 10-15% of the long-acting insulin market annually.
Pricing and Reimbursement Policies
Cost reduction efforts influence sales, particularly in cost-sensitive regions. Policies favoring biosimilars are lowering prices for NPH insulins, constraining revenue growth.
Disease Prevalence and Market Demographics
Global diabetes prevalence projected to reach 732 million by 2045. Insulin use will increase proportionally; however, clinicians are shifting toward newer insulins with improved pharmacokinetics.
What Are the Sales Projections for NOVOLIN N?
Based on current trends and regional dynamics, the following projections are proposed:
| Region | 2022 Sales (USD millions) | 2027 Projection (USD millions) | CAGR (2022-2027) | Notes |
|---|---|---|---|---|
| North America | 1,200 | 1,050 | -2.8% | Market saturation; biosimilar entry pressures. |
| Europe | 800 | 700 | -2.9% | Biosimilar competition intensifies. |
| Emerging Markets | 1,000 | 1,350 | 6% | Market growth driven by cost-sensitive adoption. |
| Total | 3,000 | 3,100 | 0.8% | Slight overall growth due to emerging markets. |
Assumptions:
- Biosimilar competition expands, limiting pricing power.
- Volume sales increase modestly in emerging markets due to rising diabetes prevalence.
- Pricing pressure persists in mature markets, leading to minimal revenue growth.
What Strategies Could Influence Sales?
- Developing combination products with long-acting insulins.
- Expanding distribution in emerging markets.
- Offering flexible dosing options to improve patient compliance.
- Engaging in cost-reduction initiatives aligned with reimbursement policies.
Key Takeaways
- NOVOLIN N holds a significant position in the insulins market, especially in cost-sensitive regions.
- Market share is declining in mature markets due to biosimilar entries and competing newer insulins.
- Sales growth is primarily driven by emerging markets, where affordability remains a critical driver.
- The global insulin market is expected to grow at a compound annual growth rate (CAGR) of less than 1% through 2027, with shifts favoring biosimilar and alternative formulations.
- Continued innovation and strategic positioning are essential to sustain or grow sales.
FAQs
1. How does NOVOLIN N compare to newer insulin formulations?
NPH insulins like NOVOLIN N offer lower costs but have less predictable pharmacokinetics than newer ultra-long-acting insulins, impacting their market appeal.
2. What factors are driving the decline of NPH insulins in mature markets?
Introduction of biosimilars reducing prices, preference for insulins with fewer injections, and improved patient outcomes with newer insulins all contribute.
3. Which regions are expected to see the highest growth for NPH insulins?
Emerging markets such as Asia, Latin America, and Africa are projected to see growth driven by affordability and expanding diabetes prevalence.
4. How are biosimilar insulins affecting NOVOLIN N sales?
Biosimilars are capturing 10-15% annually of the long-acting insulin market, exerting downward pressure on prices and sales of originator products like NOVOLIN N.
5. What regulatory changes could impact future sales?
Reimbursement policies favoring biosimilars and stricter approval pathways may lower barriers for competitors, impacting sales negatively.
Sources:
[1] Grand View Research, "Insulin Market Size, Share & Trends Analysis Report," 2022.
[2] IQVIA, "Global Insulin Market Data," 2023.
[3] Deloitte Insights, "Global Diabetes Management Outlook," 2022.
More… ↓
